Introduction
Materials and Methods
Plant material and cultural practice
Light treatments
Plant growth measurements
Determination of pigments and secondary metabolites
Determination of photosynthetic capacity, ChlF variables, the Chl content and the spectral reflectance
Insect feeding test
Statistical analysis
Results
Phenology, leaf traits, and pigment and phytochemical contents
Physiological responses of FR-treated plants
Insect feeding test of herbivore performance
Discussion
Introduction
As an important environmental signal, light regulates plant physiological responses through photoreceptors (Casson and Hetherington 2014), showing important functions that help shape the action spectra for the energy sources involved in photosynthetic CO2 assimilation, with significant impacts on plant growth (Kasajima et al. 2008). Far-red light (FR) induces shade-avoidance reaction and senescence of plants (Martinez-Garcia et al. 2014). In natural canopies, the ratio of red light (R) to FR is approximately 1.2 in daylight and is reduced to 0.2 in shade. The alteration in spectral quality acts as a vital cue for light competition among nearby plants (Heraut-Bron et al. 1999; Smith 2000; Gommers et al. 2013). A low R-to-FR ratio induces changes in physiological responses, which regulate a range of morphological changes in plants to promote their light-capturing capability (Keuskamp et al. 2010). As a plant adaptation mechanism, photo-morphogenesis enables plants to adapt to their surroundings and is influenced by phytochromes that are highly sensitive to R and FR (Jeong et al. 2024).
Prescriptive adjustments of the light spectra improve plant cultivation by controlling plant growth and development. Light-emitting diodes (LED) can produce a variety of wavelengths and offer superior energy efficiency and an economical approach compared to high-pressure sodium lamps, providing potential solutions for more sustainable greenhouse production (Yang et al. 2017; Bian et al. 2018; Lee et al. 2020; Choi et al. 2022; Vitale et al. 2022). Previously, we reported that an R:G:B ratio of 4:1:1 had numerous beneficial effects on the growth performance of basil plants compared to treatments of 2:1:1 or 1:1:1 (Lin et al. 2021). Lin et al. (2022) also indicated positive effects in coriander (Coriandrum sativum) plants on their growth, development, and appearance, including increases in chlorophyll (Chl), carotenoids (Car), and anthocyanins (Ant), under LED treatments involving flux density levels of 50 photosynthetic photon (PPFD) of R, B, and G. Furthermore, growing Rosa plants under R and RBW (white diodes) light and FR at a high R-to-FR ratio stimulates flower bud formation (Matysiak 2021). Jeong et al. (2024) demonstrates that as the FR percentage and temperature increase, the leaf length and plant height increase in basil and lettuce (Lactuca sativa L.). Legendre and van Iersel (2021) reported that supplemental FR light increases leaf expansion and the canopy size in lettuce due to increases in the cumulative incident light and biomass. Wang et al. (2021) revealed that a low R-to-FR ratio enhances the integrity and stability of the chloroplast structure in tomato plants under salinity conditions. This effect is achieved by maintaining a high activity level of the antioxidant defense system and mitigating the decreased levels of photosynthetic pigments caused by reactive oxygen species. Gerhardt et al. (2008) found that the production of flavonoids (Fla) in Brassica napus is repressed by increasing the FR intensity with ultraviolet B mediation in the spectrum.
The impact of FR light on leaves significantly influences the photosynthetic capacity of plants by modulating photosynthetic electron transport and the direction of light energy, resulting in enhanced crop yields (Tan et al. 2022). The chlorophyll fluorescence (ChlF) value, a non-invasive metric, reflects the photo-synthetic potential for photochemical dissipation and is widely used to determine plant growth and resistance in response to environmental conditions (Chen et al. 2024). The chlorophyll fluorescence indicator Fv/Fv ratio of lettuce leaves exhibited a declining trend with the supplementation of FR (Bi et al. 2024). The low quantum yield of photosynthesis under FR is primarily caused by an imbalance in excitation between photosystem I (PSI) and photosystem II (PSII) (Zhen et al. 2019). Therefore, ChlF measurements are essential for investigating plant responses to FR. In addition to ChlF, reflectance spectroscopy represents another non-invasive method by which to analyze plant physiological responses, such as the normalized difference vegetation index (NDVI) and photochemical reflectance index (PRI). These alterations in reflectance spectra have also been linked to the efficiency of photosynthetic light utilization (Gamon et al. 2015). Hence, the aforementioned ChlF and spectral reflectance indices can prove valuable in estimating leaf pigments and assessing plant growth non-destructively when constructing estimation indices.
Previous studies have demonstrated that FR can be effectively applied in vertical farming systems to improve crop productivity and herbivore resistance. Meijer et al. (2022) reported that exposure to a low R:FR ratio increases plant biomass but simultaneously represses plant defensive responses against insect herbivory. Interestingly, in contrast to most studies reporting that FR light weakens defense mechanisms (Ballaré 2014; Cortes et al. 2016; Meijer et al. 2022), FR-enriched environments can cause maize plants to respond more strongly to biotic stress by triggering the emission of the green leaf volatile (Z)-3-hexenyl acetate (Escobar-Bravo et al. 2024). In Asia, green leafy vegetables are grown in controlled environments to maximize productivity. Despite the implementation of exclusion strategies to reduce pest outbreaks, indoor vegetable cultivation remains susceptible to various herbivore pests (Nguyen et al. 2024). Thus, understanding the role of FR in regulating plant growth performance and plant defense against herbivory is crucial for optimizing these vegetable cultivation systems.
Bok choy, a rapidly growing and nutrient-dense vegetable containing vitamins, minerals, and dietary fiber as well as non-nutritive bioactive compounds, including Fla and polyphenols (PP), serves as the focal point of a number of studies investigating the impact of vertical farming systems on its productivity in greenhouse environments (Alcalde et al. 2022; Naseri 2024; Kang et al. 2025). Thus far, several studies have reported the effects of different light spectra and intensity levels on bok choy production (Manawasinghe et al. 2020; Alcalde et al. 2022), while the physiological responses and herbivore resistance capabilities of bok choy under varying FR intensities remain unclear. In the present study, we analyzed the effects of various intensity levels of FR light supplemented with R, B, and G light on the growth performance and physiological responses of bok choy in an effort to identify the optimal FR light conditions for maximizing crop productivity, phytochemical contents, metabolite contents, and the antioxidant capacity. Ultimately, our goal is to a pioneer year-round cultivation method of fresh, nutrient-rich, economically viable bok choy plants, ideally suited for controlled environments. This endeavor stands as a sustainable alternative to the reliance on chemical growth regulators. Developing an optimal approach for controlling both light quality and intensity can facilitate the design of artificial lighting environments tailored to maximize the economic advantages of bok choy production.
Materials and Methods
Plant material and cultural practice
Bok choy (Brassica rapa subsp. chinensis cv. ‘Pure Show’) seeds were sown in plug trays and grown in a fully controlled indoor plant cultivation room with a 12 h photoperiod (25°C day/night temperature and 70% relative humidity) under 60 µmol m-2 s-1 PPFD. After two weeks, the seedlings were transplanted into three-inch plastic pots. We used a commercial potting mix of peat moss, perlite, and vermiculite (3:1:1 v/v/v). The study applied a slow-release compound fertilizer (N-P2O5-K2O, 13–11–12), which was added to each pot (5 g per pot) every two weeks. The plants were watered every two days for six weeks after sowing (WAS). Bok choy plants with uniform growth at 2 WAS were raised and maintained in the controlled growth room under the following light treatments at a constant temperature of 25°C and with a 12/12 h light/dark photoperiod for another four weeks.
Light treatments
Bok choy seedlings were initially grown for two weeks under LED lighting composed of red (R, 650 nm, 40 µmol m-2 s-1), blue (B, 450 nm, 10 µmol m-2 s-1), and green (G, 550 nm, 10 µmol m-2 s-1). The light quality was characterized by an R:B:G ratio of 4:1:1. For the subsequent four weeks, three different FR treatments were additionally applied: (1) 0-FR (control, 0 µmol m-2 s-1 FR), (2) 1-FR (40 µmol m-2 s-1 FR), and (3) 2-FR (80 µmol m-2 s-1 FR). The spectrum was recorded at the youngest leaf of the bok choy plants. Each lighting experiment was conducted in a growth room maintained at a constant temperature of 25°C with a 12/12 h light/dark photoperiod. Light intensity above the bok choy canopy was measured daily and maintained by adjusting the distance of the LEDs. For each light treatment, 24 bok choy plants were grown in a completely randomized design, and the experiment was independently repeated three times. To minimize positional effects, replicates were randomly arranged among the light treatments. From each repeat, six plants were randomly selected for the subsequent growth and physiological analyses.
Plant growth measurements
Plants were harvested and morphology parameters were measured at 6 WAS. During each measurement session, the height, leaf length, and leaf width of the most recently fully expanded leaf were recorded for each plant. Plant height was detected using a Vernier caliper, with measurements taken from the base of the shoot to the top of the stem within the plant’s canopy. At harvest, shoot and root fresh weights (FW) were recorded. Samples were subsequently dried at 60°C for five days to determine the dry weights (DW). The water content (WC) was calculated using the following equation: WC = (FW − DW)/FW × 100%. For determination of the specific leaf weight (SLW), leaf discs were obtained using a circular punch with a diameter of 4 cm. The discs were dried and weighed, and the SLW (g/mm2) was calculated by dividing the leaf dry weight by the disc area (12.56 cm2), yielding an estimate of the leaf mass per unit area.
Determination of pigments and secondary metabolites
The Chl, Car, and Ant contents in plant leaves were analyzed as described by Chen et al. (2024). Briefly, the most recently fully expanded leaf of each treated plant was harvested fresh and immediately ground individually in a mortar with liquid nitrogen. For chlorophyll determination, 0.1 g of leaf powder was mixed with 5 mL of 80% acetone and incubated at 4°C overnight. After centrifugation, the supernatants were transferred to new tubes, and chlorophyll a (Chl a) and chlorophyll b (Chl b) absorbance levels were measured at 663 nm and 646 nm, respectively, using a spectrophotometer (Hitachi U-2000, Tokyo, Japan). For the Car analysis, the leaf powder was extracted with methanol. The Car content was determined at absorbance levels of 470 nm 652 nm, and 665 nm by using an Infinite M200 plate reader (Tecan, Switzerland). For the Ant measurement, leaf powder was extracted in 99% methanol containing 1% HCl and incubated at 25°C for 1 h. The extract was then centrifuged at 3,000 rpm for 5 min at 4°C, and the absorbance of the supernatant was measured at 530 nm and 657 nm. For the total Fla analysis, the Fla content was assessed according to Lin et al. (2021) by mixing 1 mL of methanol extract with 1 mL of 2% aluminum chloride solution, agitating at room temperature for 15 minutes, and measuring the absorbance at 430 nm. Total PP contents were determined using a modified method based on Bantis et al. (2016). Gallic acid and a portion of the acidic methanolic extract were diluted with an acidified methanol solution containing 1% HCl. Subsequently, 100 µL of each sample was mixed with 2 mL of Folin-Ciocalteu reagent and then vortexed for 20–30 s. After 1 min, 2 mL of 7.5% sodium carbonate solution was added, and the mixture was vortexed again. Absorbance was then measured at 750 nm using a Varioskan flash multimode reader (Thermo Scientific, Rockford, IL, USA).
Determination of photosynthetic capacity, ChlF variables, the Chl content and the spectral reflectance
Throughout the entire experiment conducted between 10:00 and 16:00, a portable photosynthesis analysis system (GFS-3000, gas exchange and fluorescence system, Walz, Germany) was employed to measure gas exchange, the photosynthesis characteristics, and the ChlF parameters. The CO2 concentration entering the leaf chamber was 450 µmol m-2 s-1, and the leaf clip temperature was 25°C during the data collection process. For each light treatment, six plants bearing the most recently fully expanded leaves were used for the gas exchange and ChlF measurements. Leaf surfaces (approximately 6 cm2 per leaf) were illuminated with PPFD levels of 2,000, 1,600, 1,200, 800, 400, 200, 100, 50, 20, and 0 µmol m-2 s-1. Plants that had been treated in the dark overnight were gradually exposed to light conditions, starting with low levels and increasing incrementally to higher levels of PPFD. Gas exchange and ChlF parameters were analyzed after dark adaptation for 30 min. Data points for each individual were recorded at 1 min intervals over a span of 60 min. The ChlF and gas exchange indicators were determined as previously described in Chen et al. (2024).
The relative chlorophyll content was assessed in the most recently fully expanded mature leaves using a soil and plant analysis development analyzer (SPAD-502 Chlorophyll Meter, Konica Minolta, Tokyo, Japan). Spectral reflectance indices were detected at wavelengths in a range of 380–790 nm using a scanning spectrophotometer (Poly PlantPen RP 400, Photon Systems Instruments, Prague, Czech Republic). The formulas of the normalized difference vegetation index (NDVI) and photochemical reflectance index (PRI) were calculated as previously described by Song et al. (2023), as follows: (R750 ‒ R705)/(R750 + R705 ‒ 2 × R445) and (R531 227 ‒ R570)/ (R531 + R570), respectively. The carotenoid reflectance indices (CRI) were calculated as 228 (1/R510) ‒ (1/R550), and the anthocyanin reflectance indices (ARI) were calculated as (1/R550) ‒ 229 (1/R700). R represents reflectance at a specific wavelength (nm).
Insect feeding test
Spodoptera litura egg masses were collected in the field in Taichung County, Taiwan, and the colony was maintained in a growth chamber (25 ± 2°C) in a laboratory. Artificial diets were supplied for hatching neonates (Tan et al. 2011), and larvae were reared individually in plastic cups after their third instar. Pupae were collected, placed in a container, and supplied with a 10% sugar solution as food once adults emerged (Tan et al. 2019). Egg masses were collected daily, and the colony was kept in the lab for an insect bioassay. To determine how the light wavelength influences plant suitability for insect herbivores, larval relative growth rate (RGR) bioassays were conducted. Six-week-old bok choy plants were planted as described above and the sixth leaf (from the bottom, 9-10 leaves in total) was collected from each plant for three treatments for the bioassay. Fourth-instar S. litura larvae were used for the RGR bioassay, and each larva was weighed and then fed one of the three treated leaves in a plastic cup lined with 2% agar to keep the leaves moist (Tan et al. 2019). Forty-eight hours later, each larva was reweighted (n = 40/each treatment).
Statistical analysis
Twenty-four bok choy plants were grown under each light condition, and all analyses were conducted throughout the experimental duration. The morphological and physiological parameters and ChlF values were subjected to a completely randomized analysis of variance (ANOVA) using PC SAS9.4 (SAS Institute, Cary, NC, USA). Mean values were distinguished using the least significant difference (LSD) test at p < 0.05, with data presented as the mean ± standard error (SE).
Results
Phenology, leaf traits, and pigment and phytochemical contents
All plants grew well during the cultivation period. In particular, the 2-FR-treated leaves of bok choy plants showed signs of a taller plant size and longer leaf length, width, and area compared to the 0-FR control plants at 6 WAS (Fig. 1). During the 28-day exposure to the FR treatments, both the 1-FR and 2-FR treatments significantly increased the plant height, leaf length, leaf width, leaf fresh weight, and leaf water content compared to controls (Fig. 2A-2C, 2D, 2E, 2G, 2I, and 2J). However, only the 2-FR-treated plants showed significantly higher root fresh weights, root dry weights, and root water contents. In addition, there were significant increases in these traits under increasing FR intensity levels, indicating that FR significantly increased the plant height, leaf length, leaf width, leaf fresh weight, root fresh weight, dry weight, root water content, and leaf water content. In contrast, specific leaf weights showed significant decreases among the treatments and controls (Fig. 2F). After 28 days of exposure, the leaf dry weight did not show any significant differences among the treatments and controls (Fig. 2H).

Fig. 1.
Representative views of bok choy plants grown under three different FR intensity levels of 0-FR (R+ B+ G, control), 1-FR (R+ B+ G+ 40 µmol m-2 s-1 FR) and 2-FR (R+ B+ G+ 80 µmol m-2 s-1 FR) for four weeks. The experiment was repeated three times with comparable results. The scale bar is 5 cm.
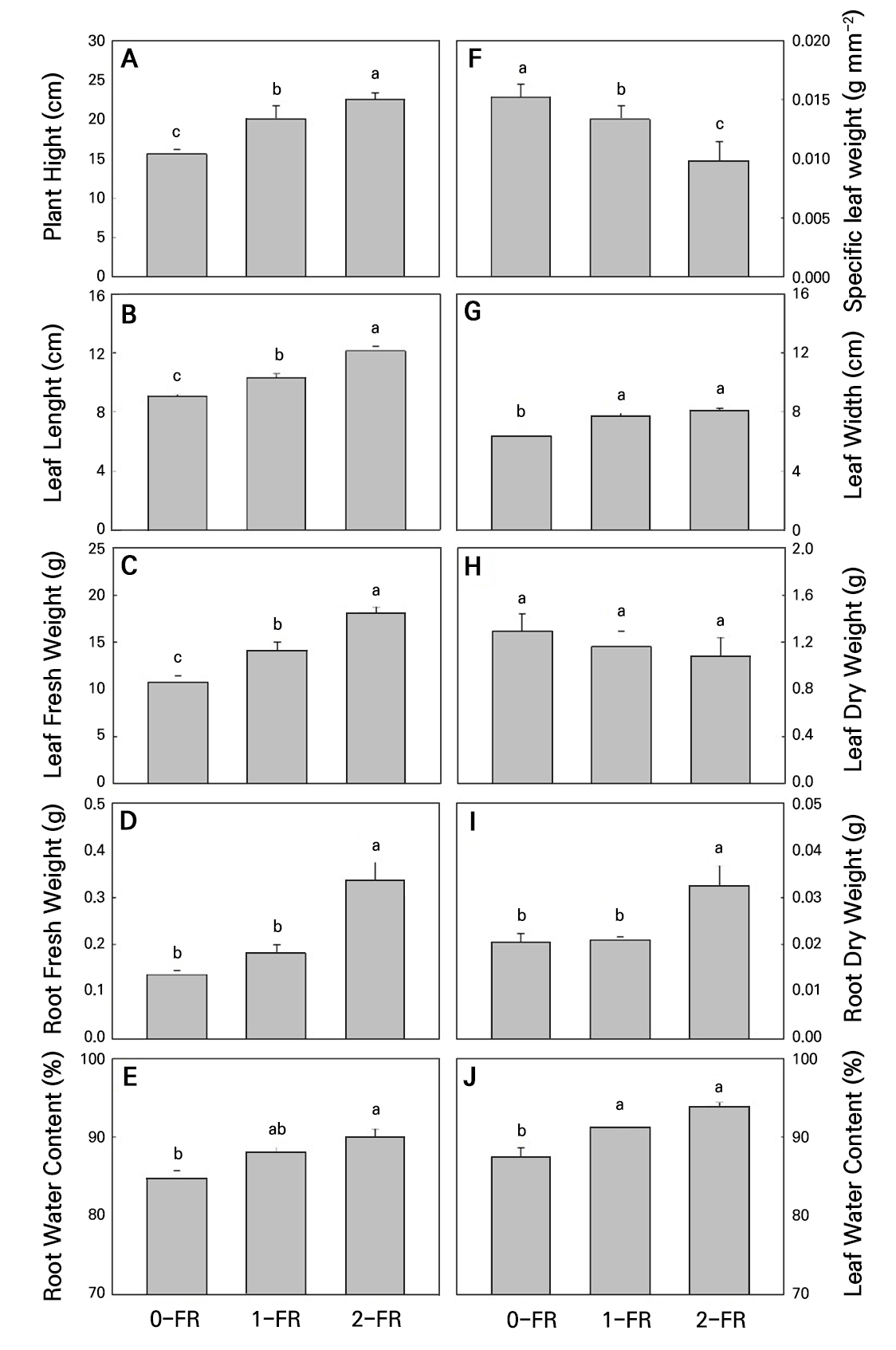
Fig. 2.
Plant height (A), leaf length (B), leaf fresh weight (C), root fresh weight (D), root water content (E), specific leaf weight (F), leaf width (G), leaf dry weight (H), root dry weight (I), and leaf water content (J) of bok choy plants exposed to supplemental 0-FR, 1-FR, and 2-FR treatments for four weeks. Error bars represent the standard error of three biological replications (six plants per replication). Different letters indicate significant differences as determined by LSD tests (α = 0.05).
No significant effects among FR and the control treatments were observed with regard to the Chl a content (Fig. 3A), whereas the FR-treated plants exhibited significantly lower Chl b levels compared to the controls (Fig. 3B). Both Car and Ant contents were significantly lower in the FR treatments compared to the controls, and these decreased with an increase in the FR intensity (Fig. 3D and 3E). Conversely, we observed that the 2-FR-treated plants had significantly higher Fla and PP contents compared to those treated with 1-FR and the controls (Fig. 3C and 3F).
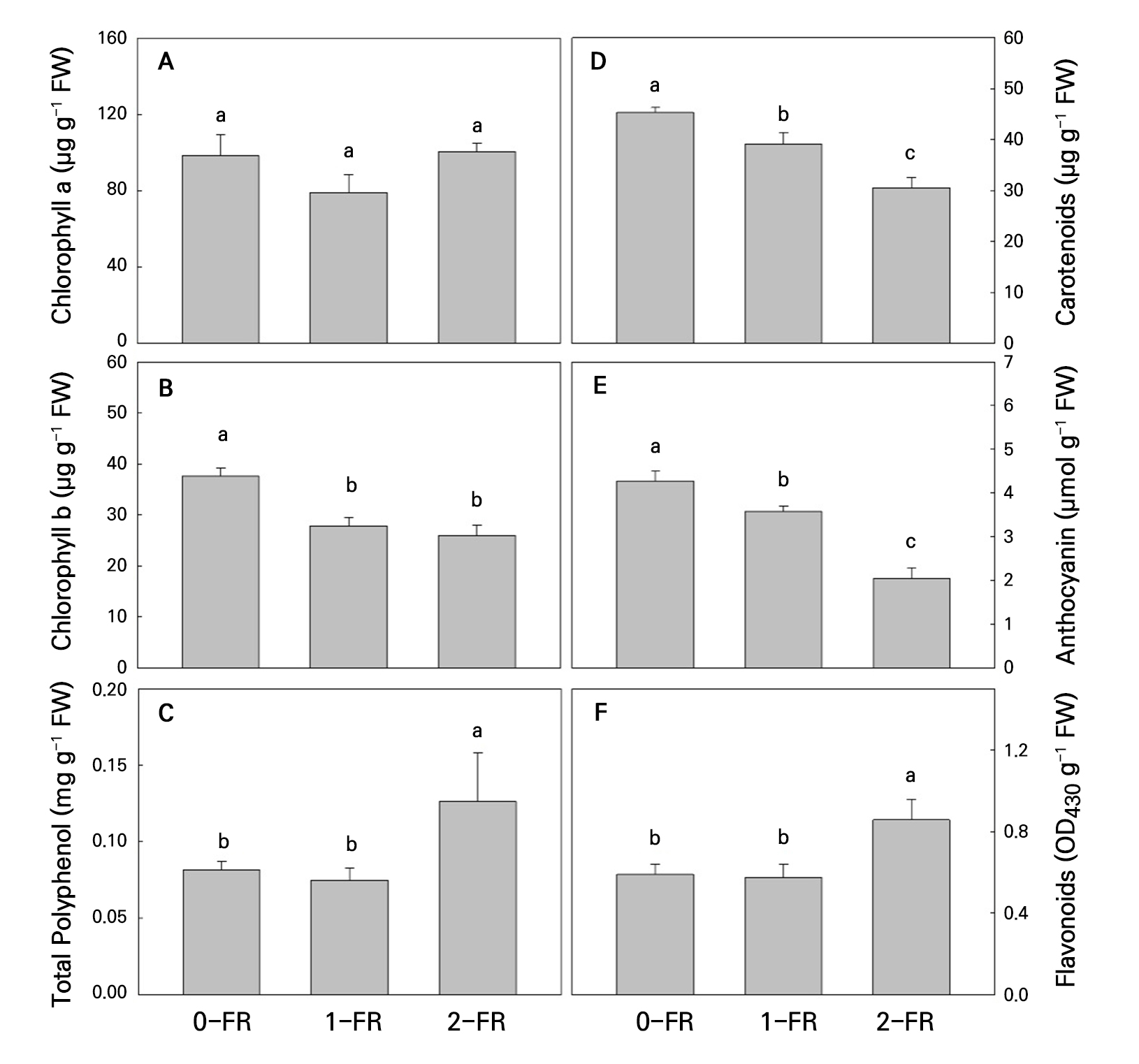
Fig. 3.
Chlorophyll a (A), chlorophyll b (B), polyphenols (C), carotenoids (D), anthocyanin (E), and flavonoids (F) of bok choy plants exposed to supplemental 0-FR, 1-FR, and 2-FR treatments for four weeks. Error bars represent the standard error of three replications (six plants per replication). Different letters indicate significant differences as determined by LSD tests (α = 0.05).
Physiological responses of FR-treated plants
Non-destructive indices were determined to study the FR treatment effects on the plants. As shown in Fig. 4, SPAD, NDVI, PRI, Fv/Fm, CRI, and ARI levels of all 2-FR-treated plants were significantly lower than those in the 1-FR-treated and control plants, and all of these indices decreased as the FR intensity was increased. Additionally, no significant differences in PRI, CRI, NDVI, or ARI were found between the 1-FR-treated and control plants (Fig. 4B-4D and 4F).
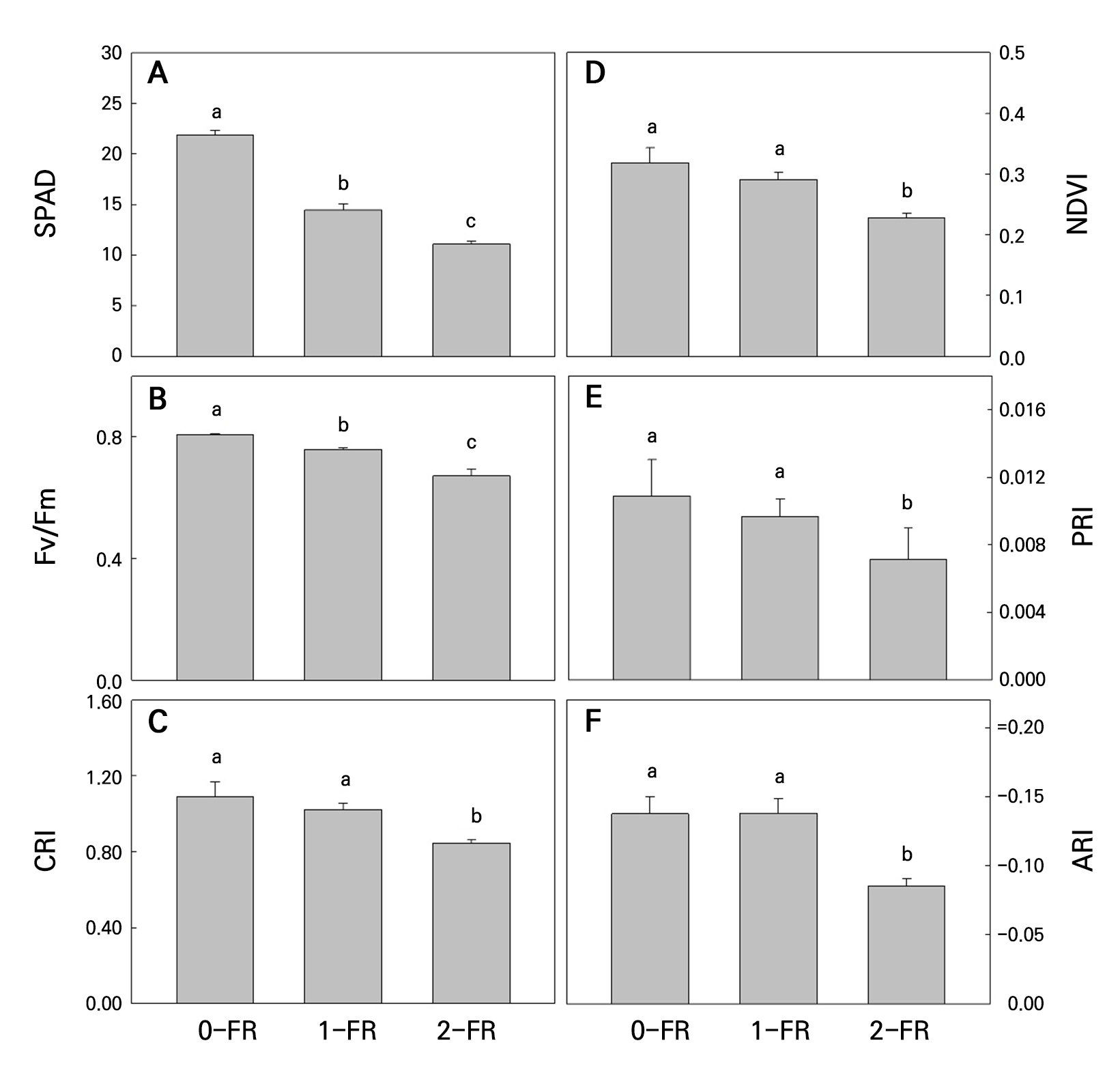
Fig. 4.
SPAD (A), Fv/Fm (B), CRI (C), NDVI (D), PRI (E), and ARI (F) values in bok choy leaves subjected to supplemental 0-FR, 1-FR, and 2-FR treatments for four weeks. Error bars represent the standard error of three replications (six plants per replication). Different letters indicate significant differences as determined by LSD tests (α = 0.05).
Figure 5 presents the LI changes in the Gs and Pn levels from the FR treatments of bok choy under PPFD levels of 2,000, 1,600, 1,200, 800, 400, 200, 100, 50, 20, and 0 µmol m-2 s-1. Both Gs and Pn from the control and 1-FR treatment in all LI treatments increased rapidly to peak at 2,000 PPFD, whereas Gs and Pn in the 2-FR treatment rapidly increased and then remained stable after 800 and 400 PPFD, respectively. Notably, Pn in the 2-FR treatment was significantly lower than those in the controls and 1-FR treatment, ranging from 200 to 2,000 PPFD.
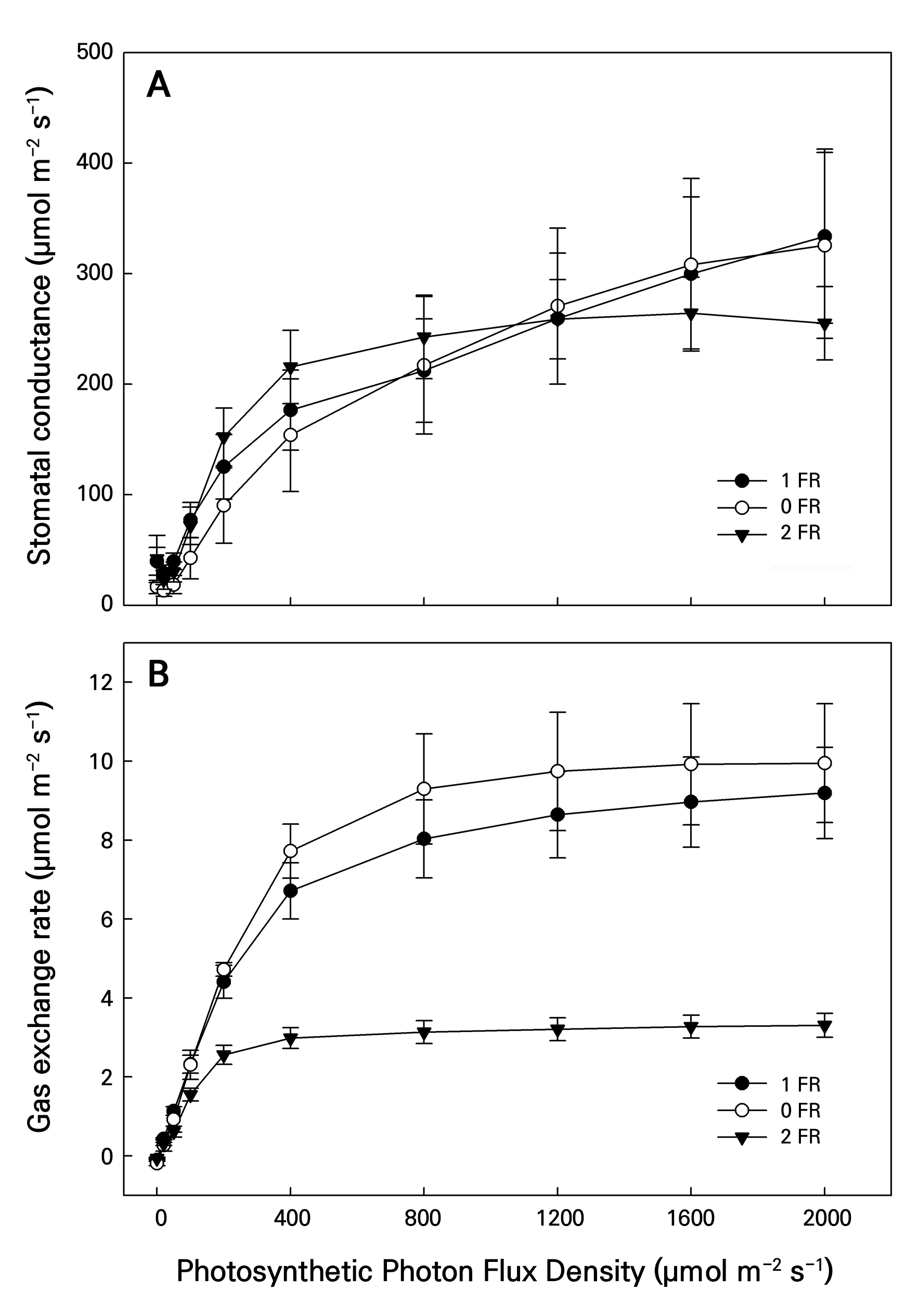
Fig. 5.
Changes in stomatal conductance (Gs, µmol H2O m–2 s–1, panel A) and net photosynthetic rate for the gas exchange rate (Pn, µmol CO2 m–2 s–1, panel B) of bok choy plants exposed to supplemental 0-FR, 1-FR, and 2-FR treatments for four weeks. White circles, black circles, and black triangles represent the 0-FR, 1-FR, and 2-FR treatments, respectively. During the 60-min light induction period, measurements were taken at 25°C under 0, 20, 50, 100, 200, 400, 800, 1,200, 1,600, and 2,000 PPFD. Error bars indicate the standard error (n = 6).
Figure 6 shows the PPFD changes in the photosynthetic light induction periods of Y(II), ETR, and NPQ. All LI measurements were initially obtained in the dark followed by exposure to PPFD levels of 2,000, 1,600, 1,200, 800, 400, 200, 100, 50, 20, and 0 µmol m-2 s-1. As the PPFD level was increased, the Y(II) values of the leaves in all treatments gradually reduced, while the NPQ values gradually increased (Fig. 6A and 6C). However, the ETR values increased sharply as the PPFD treatment levels increased, while the ETR levels remained stable after 800 PPFD (Fig. 6B). In addition, when these dark-adapted plants were treated under conditions ranging from 200 to 2,000 PPFD, the Y(II) and ETR values of the controls were significantly higher than those in all FR treatments (Fig. 6A and 6B), whereas the NPQ values in the FR-treated leaves were significantly higher than those in the controls under conditions ranging from 200 to 2,000 PPFD (Fig. 6C).
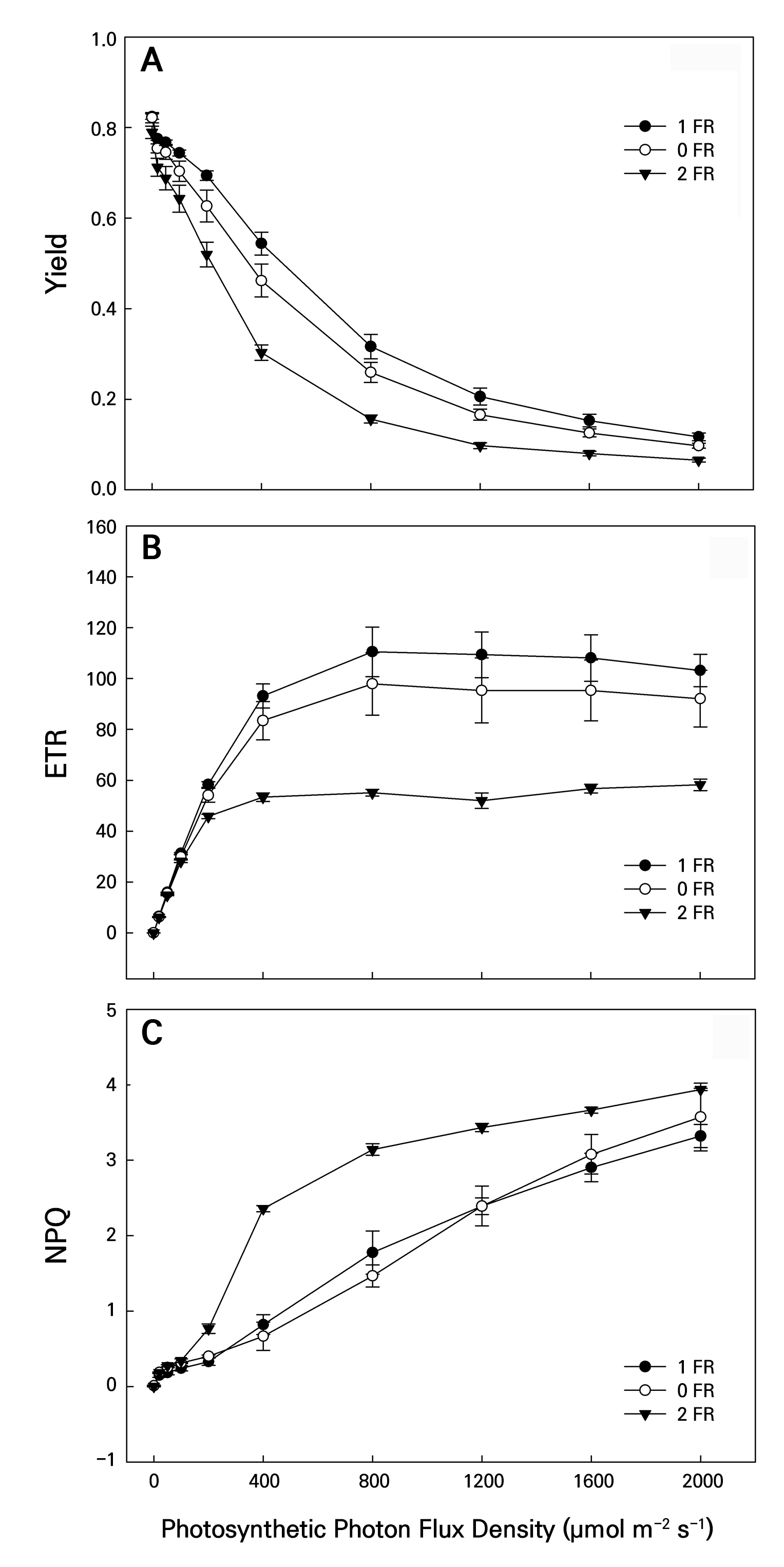
Fig. 6.
Changes in the YII (A), ETR (B), and NPQ (C) values of bok choy plants exposed to supplemental 0-FR, 1-FR, and 2-FR treatments for four weeks. White circles, black circles, and black triangles represent the 0-FR, 1-FR, and 2-FR treatments, respectively. During the 60-min light induction period, measurements were taken at 25°C under 0, 20, 50, 100, 200, 400, 800, 1200, 1600, and 2000 PPFD. Error bars indicate the standard error (n = 6).
Insect feeding test of herbivore performance
With regard to foliar herbivore performance, we evaluated the RGR of a generalist herbivore on FR-treated plants. Figure 7 shows that S. litura caterpillars showed significantly lower body weight gains when fed on 2-FR-treated plants as compared to 0-FR-treated and 1-FR-treated plants.
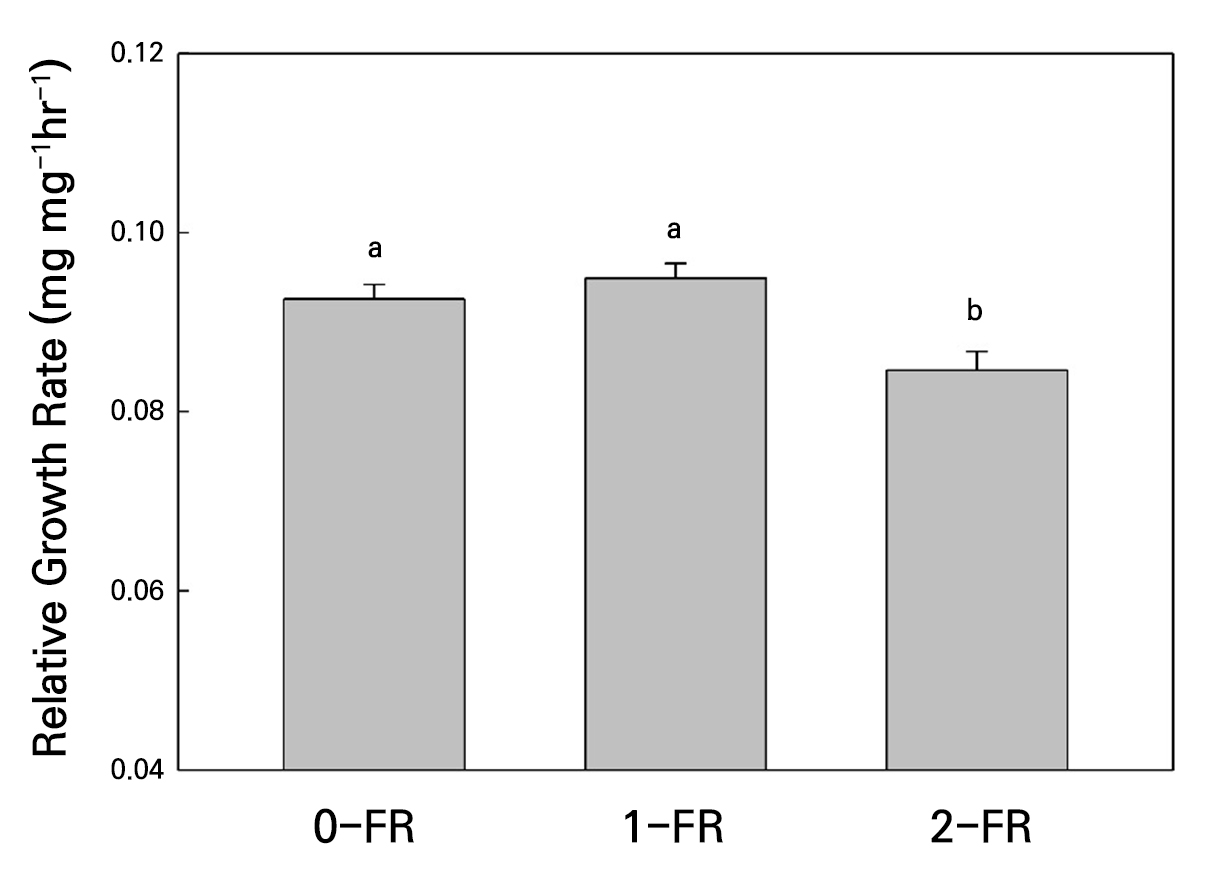
Fig. 7.
Insect herbivore Spodoptera litura relative growth rate (RGR) of bok choy plants under different far-red light treatments (0-FR, 1-FR, or 2-FR) for four weeks. Error bars represent the standard error of 40 caterpillars for each treatment. Different letters indicate significant differences as determined by LSD tests (α = 0.05).
Discussion
In this study, FR treatments increased the plant height, leaf length, leaf width, leaf area, fresh weight, Fla, and PP but decreased Chl b, Ant, and Car contents, the photosynthetic efficiency, and the performance of S. litura herbivores in bok choy plants. The 2-FR treatment significantly decreased Fv/Fm, Gs, Pn, Y(II), and ETR levels in bok choy plants compared to those in the 1-FR-treated and control plants. Previously, Zou et al. (2019) reported that an adjustment of plant growth induced by supplemental FR light is crucial for enhancing indoor lettuce cultivation. Additionally, the increased yield from FR light supplementation cannot be replicated by adding equivalent amounts of red and blue light, thereby emphasizing its critical contribution to optimizing growth and productivity in indoor vegetable production systems.
The photosynthetic pigment content represents the photosynthetic ability of plants. The Car content and CRI values of bok choy leaves decreased when the FR intensity was increased. Car can transfer energy to Chl for photosynthesis and can protect Chl from photo-oxidation (Tan et al. 2022). The high-FR treatment induced low levels of Fv/Fm, Pn, Y(II), and ETR, possibly stemming from the low Chl and Car values. A decrease in photosynthetic capacity can lead to reduced light energy harvesting, conversion, accumulation, and fixation levels in chloroplasts (Lemeille and Rochaix 2010; Zou et al. 2019; Pozo et al. 2020). Consequently, these non-destructive index values can be useful when measuring FR-treated plants and controls. Different FR treatments at varying LI levels showed variable ChlF and photosynthesis values. At a specific LI level, plants can be distinguished based on their Gs, Pn, ETR, NPQ, and Y(II) outcomes. Generally, as LI was increased from 20 to 2,000 PPFD, increasing Gs, Pn, and NPQ and decreasing ETR and Y(II) values were observed in all plants. Franklin and Quail (2010) reported that after prolonged exposure to FR light, plants perceive it as a signal of low light and adjust their morphology to capture more photons by elongating their petioles and elevating their leaf tips. Supplemental FR light induces stomatal closure by modulating the redox state of plastoquinone in guard cells, thereby impacting the efficiency of CO2 fixation (Busch 2014). Bok choy plants exhibited a swift rise in the NPQ value alongside a decline in the Y(II) value, which was a manifestation of photo-protective ability, and thus these plants exhibited a considerable decrease in their PSII efficiency (Lai et al. 2022). As FR was increased, NPQ increased but Y(II) decreased, implying that high-FR light increased the heat dissipation of PSII and reduced the light energy available for photosynthesis in the leaves. When bok choy plants were subjected to high LI levels, the surplus energy in PSII heightened, resulting in elevated NPQ values. In parallel, an elevated level of NPQ indicated a light-suppressing effect, resulting in a decrease in the Y(II) value. The NPQ value in 2-FR-treated plants was notably higher compared to those in the 1-FR and control plants under conditions ranging from 200 to 2,000 PPFD (Fig. 6), indicating that 2-FR-treated leaves may have more NPQ with a higher damage level mediated by photo-oxidation. Furthermore, FR-treated leaves tended to divert photosynthetic ETR towards quenching energy, even in the presence of photo-inhibition. Enhanced FR absorption by photosystems is believed to be adequate for enhancing photosynthetic activity under FR light. ETR and NPQ can serve as indicators of photo-protection and photo-inhibition under a FR treatment at a specific LI for predicting photosynthetic responses to light induction under various FR treatments in bok choy. Furthermore, the SPAD, NDVI, PRI, Fv/Fm, CRI, ARI, ETR, and NPQ values showed rapid and efficient responses to the 2-FR treatment of bok choy (Figs. 4 and 6), which could be readily quantified under field conditions. These simple and non-destructive physiological measurements allow rapid assessments of plant responses under FR treatments. Changes in these physiological indicators can be used to identify unsuitable light conditions or, conversely, to determine the optimal light quality and minimal requirements for efficient plant growth. Therefore, these indicators serve as valuable tools for monitoring Brassica plants and for evaluating whether they are growing under optimal light intensity and quality in a controlled environment.
The alterations in the plant morphology and physiology induced by FR supplementation could impact plant-arthropod interactions by modifying plant defense traits (Cortés et al. 2016). Plants subjected to a low red to far-red (RL:FR) light ratio exhibit pronounced stem elongation, leaf expansion, and leaf hyponasty (Legendre and van Iersel 2021), which are characteristic features of the shade-avoidance syndrome, allowing the plant to capture more light. During shade-avoidance, plants often exhibit reduced photosynthetic efficiency, primarily due to decreased stomatal conductance and lower cytokinin accumulation (Boonman et al. 2007). These observations are consistent with our findings, in which plants exposed to high-FR light accumulated greater biomass despite reduced photosynthetic efficiency. Notably, the effects of FR light on root growth are species-specific in plants (Wang et al. 2025). In bok choy, supplemental FR increases root biomass, which may be associated with enhanced auxin biosynthesis or more efficient auxin transport (Christiaens et al. 2019). In addition, this adaptive response redirects resources toward vertical growth at the expense of defense-related pathways, thereby increasing the plant’s vulnerability to herbivore attacks (Lazzarin et al. 2021). In cucumbers, the enhanced resistance to Sphaerotheca cucurbitae when plants are treated with a high R-to-FR ratio is attributed to alterations in leaf thickness rather than changes in defense signaling (Shibuya et al. 2011). Changes in leaf morphology can also significantly influence plant-herbivore interactions. Shibuya et al. (2011) also reported that whiteflies exhibit a preference for plants cultivated under low R-to-FR conditions over those grown under high RL-to-FR conditions, a finding they attributed to the darker green color and thicker leaves of plants grown at a high RL-to-FR ratio. Consequently, plants grown under low R:FR conditions not only display accelerated morphological development but also demonstrate increased susceptibility to insect herbivory, often resulting in enhanced herbivore growth and performance (Cortés et al. 2016). Interestingly, S. litura caterpillars showed significantly lower performance when fed on 2-FR-treated bok choy plants, suggesting that not all FR supplementation has uniform effects on plant-insect dynamics.
Brassica plants are highly sensitive to the light quality and intensity characteristics, with these effects found in the amounts of secondary metabolisms (Zou et al. 2024). Generally, exposure to FR light decreases the production of several defensive secondary metabolites, consequently heightening plant vulnerability to biotic stress (Ballaré 2014) However, Carvalho and Folta (2014) suggested that FR treatments elevate aliphatic glucosinolates and Fla levels in Brassica napus, suggesting a potential enhancement of resistance to pest herbivory. In bok choy plants, PP and Fla contents were increased by 2-FR supplementation. Plant PP compounds not only contribute to color, odor, astringency, and oxidative stability but also play an important role in chemical defenses against biotic stress through insecticidal or insect deterrent activity (Singh et al. 2021; Ramaroson et al. 2022). Dicke and Lucas-Barbosa (2020) also reported that Fla protects plants against herbivory by influencing the behavior, growth, and development of arthropods. Similarly, high contents of certain types of PP, including quercetin, chlorogenic acid, and rutin, have been linked to the resistance to S. litura among wild-cultivated crosses of groundnut plants (Mallikarjuna et al. 2004). Therefore, different herbivore performance outcomes under certain FR conditions could be associated with the variance in the elevated contents of Fla and PP in bok choy plants. Additionally, studies in medical microbiology offer valuable insights, as several researchers have proposed detailed mechanisms of action for PP and Fla in searches for natural antibiotics beneficial to human health (Adamczak et al. 2019; Gutiérrez-Venegas et al. 2019). In mint plants, the expressions of the key enzyme genes involved in phenylpropanoid and flavonoid biosynthesis, including phenylalanine ammonia-lyase (PAL), cinnamyl-alcohol dehydrogenase (CAD), peroxidase (POD), phlorizin synthase (PGT1), flavonoid 3’,5’-hydroxylase (F3’5’H), and chalcone synthase (CHS), were up-regulated under FR treatments (Yu et al. 2024). These results suggested that FR treatments may enhance the accumulation of PP and Fla through the upregulation of enzyme genes involved in the biosynthetic pathways of Fla and phenylpropanoids in bok choy plants. Therefore, the integration of specific LED light spectra in bok choy cultivation represents a promising strategy not only for optimizing crop yields and insect resistance capabilities but also for enriching the nutritional and medicinal value of crops.
In conclusion, bok choy plants show several different morphological and physiological responses under FR treatments and display an intensity-dependent shade avoidance response that leads to increased growth. In addition, FR treatments have the potential to enhance metabolites and reduce arthropod herbivory. The SPAD, Fv/Fm, Y(II), qP, and ETR values provide comprehensive insights into plant physiology and serve as powerful indicators capable of informing plant selection in bok choy enhancement programs and sustainable management practices across different FR regimes. Consideration of the interactive effects of light with various wavelengths and the photosynthetic enhancement properties of FR light is crucial for optimizing photochemical efficiency and photosynthesis. Understanding the influence of supplemental FR light on not just plant growth performance but also defense ability to arthropod pests is essential for the effective utilization of LEDs. LEDs have the capability to irradiate plants with a range of light quality conditions and intensity levels at specific times and durations, enabling efficient control over plant growth, development, and metabolic regulation in plant factories. Despite the useful findings here, more comprehensive investigations of the molecular signaling and metabolic alterations of bok choy plants in response to FR light are necessary.


