Introduction
Materials and Methods
Plant Material
Foliar Application
Morphological Attributes
Biochemical Attributes
Statistical Analysis
Results
Morphological Attributes
Biochemical Attributes
Discussion
Introduction
Gladiolus is a perennial flowering bulbous plant of the Iridaceae family, with 260 known species worldwide, in-cluding 250 gladiolus species originating in sub-Saharan Africa and the rest originating in Eurasia (Goldblatt and Manning, 1998; Manning and Goldblatt, 2008). Gladiolus has restricted uses in landscaping but is often used as a cut flower. The availability of gladiolus flowers in various colors makes them attractive and popular among consumers. Both the demand for gladiolus spikes and the production of this product have increased over the past few years. Approximately 60 million spikes worth 16 million US dollars were sold in the US in 2011 (USDA, 2012).
The production of good quality spikes has an important influence on obtaining a good price for gladiolus in the international market. Increased spike length and number of florets per spike are desirable characteristics in the marketplace. Various approaches are used to improve the commercially valuable characteristics of cut flowers. For example, exogenous application of growth bioregulators helps improve the quality of cut flowers (Sajid et al., 2009).
Gibberellins, cytokinins and auxins are well-known classes of plant growth regulators (PGRs) that are used to modify the morphological characteristics of ornamental plants. Recent studies have demonstrated the growth-promoting effects of chemicals (polyamines and vitamins) in rose (Farahi et al., 2013) and Syngonium (Nahed et al., 2007), but whether their effects are comparable to those of known PGRs remains to be investigated. Indeed, polyamines and vitamins can also regulate plant growth. Polyamines play an important role in plants and were categorized as a new class of plant growth bioregulators due to their plant growth-promoting effects (Mahgoub et al., 2006, 2011). Polyamines are low molecular weight nitrogenous com-pounds that are abundant in all living organisms. Higher plants possess high levels of the polyamines putrescine, spermidine and spermine (Ben Mohamed et al., 2010). Polyamines are biologically active compounds involved in various physiological processes in the cell, including the regulation of development, mor-pho-genesis and plant cell growth (Hanzawa et al., 2000), and they also reduce the negative impact of salinity on plant growth (Kamiab et al., 2014).
The most abundant vitamins found in plants are ascorbic acid, citric acid and thiamine, which are also known as antioxidants due to their strong antioxidant properties. Vitamins are also considered to be growth bioregulators, as low concentrations of vitamins may strongly affect metabolic pathways, plant growth regulation and physiological pro-cesses, including the biosynthesis of enzymes and co-enzymes (Hathout, 1995; Robinson, 1973). Foliar application of anti-oxidants may improve important plant metabolic and physio-logical processes, which in turn may increase plant growth (Abdel-Aziz et al., 2007; Maksoud et al., 2009). Foliar application of vitamins, even in small amounts, can influence plant growth and biological processes (Nahed et al., 2009). Ascorbic acid is a powerful antioxidant that is involved in regulating photosynthesis (Blokhina et al., 2003), and thiamine is important in the metabolism of carbohydrates and fats (Bidwell, 1979).
The beneficial effects of these vitamins vary, as different plants have different responses to the same concentrations of growth bioregulators. Therefore, it is important to optimize the dose for each type of plant. In view of the importance of bioregulators to the cut flower industry, this study was performed to determine the concentrations of selected polyamines (putrescine and spermine) and vitamins (ascorbic acid and thiamine) that can be effectively used to improve flower spikes, increase propagule production and enhance physiological attributes in Gladiolus grandiflorus.
Materials and Methods
Plant Material
Healthy gladiolus corms of the cultivar ‘White prosperity’ were purchased from a local market in Lahore, Pakistan. The corms were planted at a spacing of 15 cm (plant to plant) and 60 cm (row to row) in the floriculture research area at the University of Agriculture, Faisalabad, Pakistan. Cultural practices including weeding, hoeing and earthing up were performed manually. NPK fertilizer was applied at 200-180-200 kg・ha-1, and the irrigation schedule was designed according to plant requirements.
Foliar Application
The gladiolus plants were grown until the third leaf stage. The plants were then sprayed with different concentrations (0, 0.1. 0.4, 0.7 or 1 mM) of freshly prepared solutions of putrescine, spermine, ascorbic acid or thiamine containing 0.01% Tween-20 as an emulsifier. Spraying was performed in the afternoon before sunset to reduce the chance for evaporation and to increase the absorption of the solution into the leaves. Foliar application was repeated at the five-leaf and slipping stages (at the emergence of flower spikes). Each treatment consisted of 45 plants.
Morphological Attributes
Morphological parameters including plant height (cm), spike length (cm), spike diameter (mm), diameter of firstfloret (cm) and weight of first floret (g) were recorded after opening of the first floret of a spike in a gladiolus plant. The plants were then grown in the field. After corm development, the corms were lifted from the field and parameters including corm diameter (cm), corm weight (g), number of cormels per plant and total cormel weight per plant (g) were recorded.
Biochemical Attributes
Plant leaf samples (freeze dried) were homogenized in 96% ethanol and incubated overnight at room temperature. The chlorophyll contents in mature leaves of gladiolus plants were measured as described by Lichtenthaler (1987).
To measure macronutrient levels, 0.5 g of oven dried leaf samples were digested in 10 mL HCl at 250°C in digestion tubes, and nitrogen (N), phosphorus (P) and potassium (K) levels were measured according to the method of Chapman and Parker (1961). To measure total soluble sugars, oven- dried leaf tissue (1 g) was homogenized in 10 mL of 80% ethanol and centrifuged at 4000 rpm for 3 minutes. The supernatant was used to estimate total soluble sugar levels by spectrophotometry according to Riazi et al. (1985).
To measure phenolic contents, leaf tissue was homogenized in 95% methanol and incubated at room temperature for 36 h. Total phenolic contents (mg・g-1) were estimated according to Ainsworth and Gillespie (2007).
Statistical Analysis
The experiment was conducted in a randomized complete block design with three replications per treatment. The treatment means were compared via a Least Significant Difference (LSD) test at the 5% significance level using DSAASTAT ver. 1.101 software.
Results
Morphological Attributes
Foliar application of polyamines and vitamins improved the vegetative and floral attributes of gladiolus plants, as shown in Table 1. The plant height increased after foliar application of polyamines (putrescine or spermine) and vitamins (ascorbic acid or thiamine). In the control plants, the plant height was 92.12 cm, whereas this value reached a maximum of 107.85 cm after treatment with 1 mM putrescine and 93.15 cm after treatment with 1 mM thiamine.
The most commercially important traits of gladiolus spikes, including spike length, spike diameter, number of florets per spike, first floret diameter and floret weight were significantly improved by foliar application of polyamines and vitamins, as shown in Table 1. Putrescine treatment produced the greatest improvement in the floral charac-teristics of gladiolus, followed by the spermine, ascorbic acid and thiamine. The best performance was obtained using 1 mM putrescine, which increased the spike length (to 55.53 cm), spike diameter (7.53 mm), number of florets per spike (13.99) first floret diameter (9.66 cm) and first floret weight (4.90 g) compared to control treatment, as shown in Table 1 and Fig. 3. Treatment with 1 mM spermine, ascorbic acid or thiamine increased the spike length to 51.21 cm, 46.06 cm and 43.11 cm, respectively.
The development of underground structures of gladiolus, including corms and cormels, was also positively affected by the application of polyamines (putrescine or spermine) and vitamins (ascorbic acid or thiamine), as shown in Table 2. In non-treated plants, the corm diameter (3.47 cm), corm weight (17.66 g), cormels per plant (47.19) and total cormel weight (12.64 g) increased to 4.57 cm, 26.16 g, 80.86 and 23.03 g, respectively, after 1 mM putrescine treatment. Application of 1 mM ascorbic acid and thiamine increased corm diameter to 4.06 cm and 3.91 cm, respectively.
Biochemical Attributes
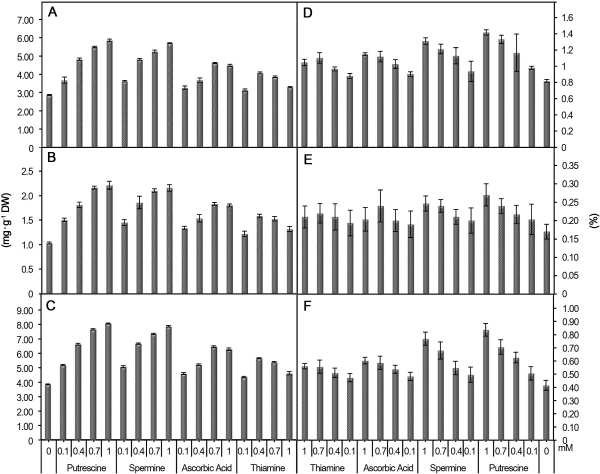
Chlorophyll a, b and total chlorophyll contents significantly increased in response to treatment with polyamines and vitamins (Fig. 1). The control plants contained 2.87 mg・g-1 chlorophyll a, 1.03 mg・g-1 chlorophyll b and 3.86 mg・g-1 total chlorophyll, which increased to the maximum levels of 5.85, 2.21 and 8.06 mg・g-1, respectively, after 1 mM putrescine treatment. Treatment with 0.7 mM ascorbic acid increased the total chlorophyll content to 6.46 mg・g-1, followed by 5.66 mg・g-1 in response to 0.4 mM thiamine treatment.
The macronutrient contents of leaves, including N, P, and K, increased in response to various treatments (Fig. 1). In the leaves of control plants, the macronutrient content was 0.82% N, 0.17% P, and 0.42% K, whereas after 1 mM putrescine treatment, the maximum concentrations of these nutrients were 1.42% N, 0.27% P, and 0.84% K. The next most effective treatment was spermine, which increased these levels to 1.31% N, 0.25% P, and 0.77% K, followed by 1 mM ascorbic acid treatment.
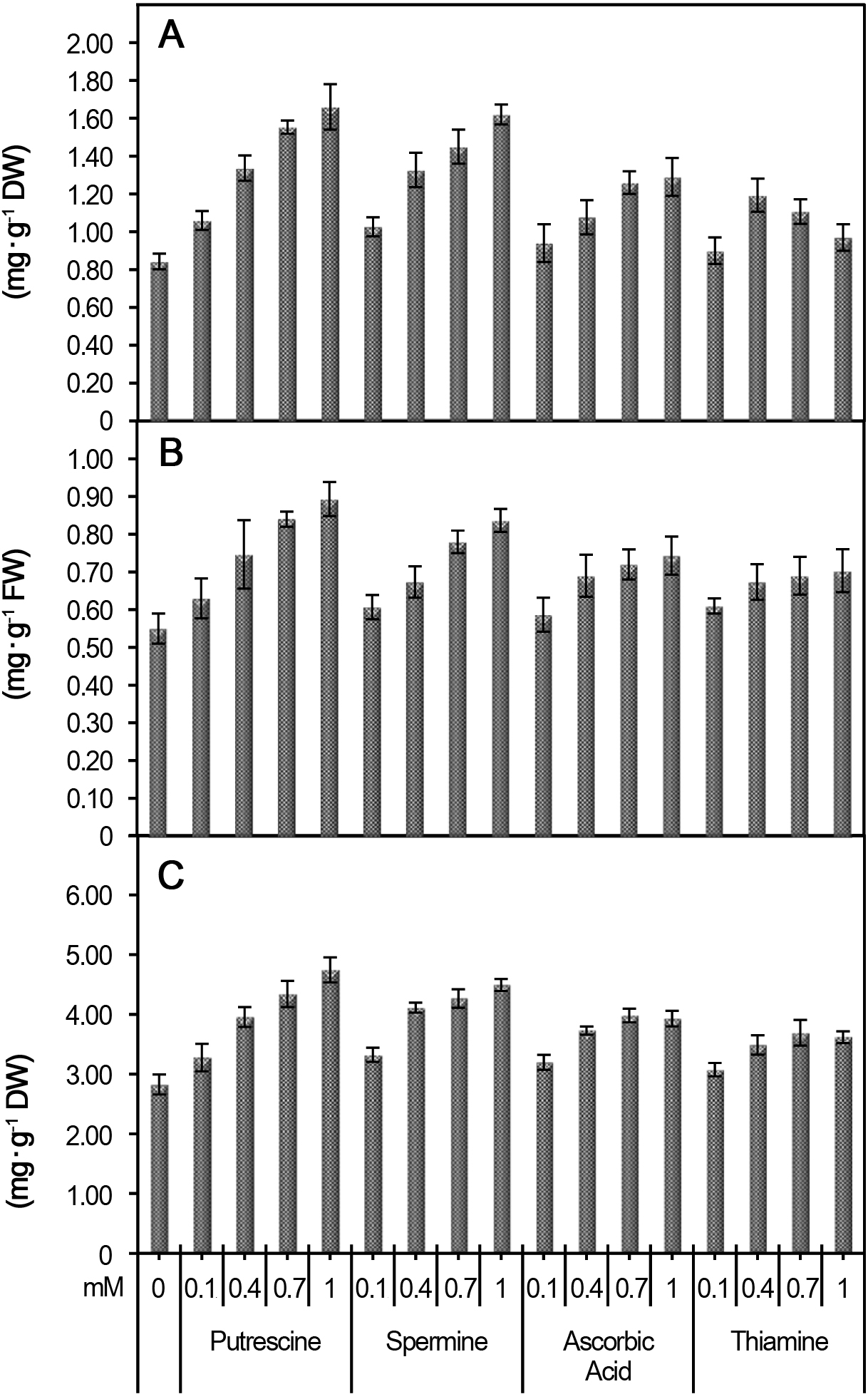
Total carotenoid contents increased in the leaves of plants treated with 1 mM putrescine (1.66 mg・g-1) compared to the control (0.84 mg・g-1), as shown in Fig. 2. Treatment with 1 mM spermine increased carotenoid levels to 1.62 mg・g-1, whereas carotenoid levels increased to 0.94 mg・g-1 and 1.29 mg・g-1 after treatment with 0.1 and 1 mM ascorbic acid, respectively.
|
Fig. 1. Effect of polyamines and vitamins on chlorophyll-a (A) chlorophyll-b (B), total chlorophyll (C), nitrogen (D), phosphorus (E) and potassium (F) contents in leaves. Vertical bars show SE. |
Total phenolic and total soluble sugar levels increased after putrescine and spermine treatment, but the greatest increases in phenolic (0.89 mg・g-1) and soluble sugar (4.75 mg・g-1) levels were observed after 1 mM putrescine treatment (Fig. 2). Ascorbic acid and thiamine treatment also increased in phenolic levels (to 0.74 mg・g-1 and 0.70 mg・g-1, respec-tively). Finally, treatment with 0.7 mM ascorbic acid and thiamine increased soluble sugar levels to 3.98 mg・g-1 and 3.69 mg・g-1, respectively.
Discussion
Enhanced plant growth in response to foliar application of polyamines and vitamins has been reported for various plant species. For example, Mahros et al. (2011) found that flowering characteristics were enhanced in Chrysanthemum indicum L. in response to foliar application of putrescine. In the current study, we found that polyamines enhance plant characteristics in gladiolus. These compounds are also effective growth bio-regulators in other plants, including Datura (Youssef et al., 2004), periwinkle (Talaat et al., 2005) and Dianthus caryophyllus (Mahgoub et al., 2006). We also found that vitamins (ascorbic acid and thiamine) regulate the growth of gladiolus plants, although less effectively than treatment with polyamines (putrescine or spermine). Our results demonstrate that ascorbic acid can regulate various growth parameters in gladiolus, which supports the previous finding that exogenous application of ascorbic acid has positive effects on the growth parameters of many plants, including marigold (Talaat, 2003), rosemary (Youssef and Talaat, 2003) and Syngonium (Abdel Aziz et al., 2007). The effects of these compounds observed in the present study are comparable to the effects of application of commonly used PGRs including gibberellic acid, ethephon, naphthaleneacetic acid (NAA) and Cycocel in various cultivars of gladiolus. Specifically, in the present study, application of putrescine (1 mM) increased plant height (to 107.85 cm), spike length (55.53 cm), florets per spike (13.99), floret diameter (9.66 cm), corm diameter (4.57 cm), corm weight (26.16 g), cormels per plant (80.86) and total cormel weight (23.03 g) in ‘White prosperity’ gladiolus. These results are comparable to the findings of Lahiji (2013), who applied a combination of the commonly used PGRs gibberellic acid and ethephon to gladiolus and achieved a spike length of 42.53 cm, floret diameter of 10.68 cm and corm weight of 7.46 g in ‘White prosperity’. Maurya and Nagda (2004) reported a plant height of 128.53 cm, florets per spike of 17.60, corm diameter of 7.52 cm and cormels per plant of 11.53 in ‘Oscar’ gladiolus through application of 100 ppm gibberellic acid. Finally, a plant height of 85.44 cm and florets per spike of 10.70 were achieved after application of 100 ppm naphthaleneacetic acid (NAA), and application of Cycocel (500 ppm) yielded a corm weight of 39.21 g, cormels per plant of 26 and total cormel weight of 5.72 g in ‘White friendship’ (Sudhakar and Kumar, 2012). These varied results may be due to the different concentrations of PGRs and different cultivars used in these studies.
|
Fig. 2. Effect of polyamines and vitamins on total carotenoids (A), total phenolics (B) and total soluble sugars (C) in gladiolus leaves. Vertical bars show SE. |
In the current study, the total chlorophyll contents in leaves significantly increased in response to foliar application of polyamines and vitamins compared to the control. These results are in line with the findings of Lee et al. (1997), who reported that exogenous application of putrescine inhibits the loss of chlorophyll content in carnation plants. Indeed, we found that foliar application of polyamines (putrescine or spermine) significantly increased the chlorophyll a, b and total chlorophyll contents compared to the non- treated control. Ascorbic acid is the most abundant antioxidant in plants and is also involved in various physiological processes, including photosynthesis (Bolkhina et al., 2003). The exogenous application of ascorbic acid in the present study resulted in an increase in photosynthetic pigment levels. Similarly, an increase in chlorophyll contents due to foliar application of ascorbic acids was reported in rosemary (Youseff and Talaat, 2003).
We found that foliar application of polyamines (putrescine or spermine) and vitamins (ascorbic acid or thiamine) significantly increased the levels of macronutrients, including N, P and K, in leaves. An increase in macronutrient (N, P, and K) levels, especially K, in response to foliar polyamine treatment was also reported by Salama (1999). Polyamines, especially putrescine, also enhance N uptake and accumulation in Myrtus communis (Hanafy-Ahmed et al., 2002). Shawky (2003) found that N, P and K concentrations in plants increase after polyamine treatment, thus demonstrating the role of polyamines in improving both the growth and chemical composition of plants. We also found that treating leaves with ascorbic acid or thiamine also had a positive effect on macronutrient (N, P, K) contents. Similarly, the levels of these micronutrients increase in response to foliar application of ascorbic acid in sweet pepper leaves (Talaat, 2003) and ascorbic acid and thiamine in rosemary (Youssef and Talaat, 2003).
We observed an increase in soluble sugar levels in the leaves of treated plants, which is a positive indicator of plant growth in gladiolus. These results are in agreement with the results of El- Bassiuony and Bekheta (2004), who found that carbohydrate contents in wheat plants increased in response to exogenous application of putrescine. This increase in sugar contents may be attributed to an increase in photosynthetic activity. Finally, Abdel Aziz et al. (2007) found that ascorbic acid or thiamine treatment increased the carbohydrate content in leaves of Syngonium podophyllum L., and Tarraf et al. (1999) observed increases in carbohydrate and total phenol contents in response to foliar application of ascorbic acid in lemon grass.