Introduction
Materials and Methods
Plant Materials
Experimental Design and LED Light Treatments
Plant Growth Parameters
Data Analysis
Results and Discussion
Survival Rate and Shoot-root Formation
Vegetative Growth and Development
Introduction
The succulent market has grown over the years, and the prices of succulents increased by more than 62% between 2012 and 2017 in the US, and more so in Asia (Smith-Wong, 2021). Pachyphytum, a popular succulent, isa small genus of the Crassulaceae family native to Mexico, with only over 20 commercially available species and various hybrids (Charles, 2014). These species are known to be slow-growing succulents compared to the Echeveria genus and are known for their thick and plump leaves arranged in rosettes or scattered on stems (Mason, 2015).
According to Cabahug et al. (2016) and Tuttle (2022), the propagation of succulents is unpredictable and oftentimes a game of chance. However, it may be aided by proper culturing practices and techniques. Currently, the rapid propagation of horticulture crops has been aided considerably using artificial lighting. Several studies have investigated the use of supplemental lights, including light-emitting diodes (LEDs), in relation to the growth, development, and increasing ornamental value of succulent plants (Nam et al., 2016; Cabahug et al., 2017; Lee et al., 2021a, 2022b; Kim et al., 2022; Lee and Nam, 2022, 2023). Nonetheless, most of these studies have predominantly focused on enhancing the ornamental value and plant growth outcomes, rather than placing emphasis on propagation.
According to various studies conducted in the past, it has been reported that in addition to light intensity, the spectral distribution or quality of light also plays a significant role in influencing plant growth, and ornamental values and enhancing physiological activities that contribute to increased photosynthesis, compound accumulation, and pigmentation (Virsile et al., 2017; Choi et al., 2020, 2022; Lee et al., 2020, 2021b, 2022a; Song et al., 2020a, 2020b; Roh and Yoo, 2023). Light-emitting diodes are considered the best choice among artificial light sources in controlled plant cultivation because they are generally more energy-efficient, have a long bulb lifetime, and produce low heat emission as compared to other types of lamps (Bourget, 2008; Fujiwara, 2016; Pattison et al., 2018). LEDs have a wide range of electric lamp systems capable of generating various qualities of white light, red and blue (monochromatic light), and purple (composite spectral distribution light) spectra. White light has been reported to induce stem elongation and leaf expansion, while red and blue light spectra have been used considerably as excellent energy sources for photosynthetic CO2 assimilation (Cope and Bugbee, 2013). However, the effects of these light spectra can vary depending on the plant species.
To date, no studies have sought to determine the effects of different qualities of light on the enhancement of the vegetative propagation of Pachyphytum leaf cuttings. Hence, this study was conducted to evaluate the effects of different LED lights: white, red, blue, and purple lights, to propagate and promote the growth and development of Pachyphytum species when grown via vegetative propagation most efficiently.
Materials and Methods
Plant Materials
The Pachyphytum species used in this study, specifically P. compactum, P. glutinicaule, P. machucae, P. oviferum, P. viride, and P. cv. Oviride (P. oviferum × P. viride) (Fig. 1), were obtained from the germplasm of the Cactus Industry-University Cooperation Foundation of Sahmyook University. Leaf cuttings of the lower whorl of the plants were planted in rectangular pots (48.5 × 33 × 8; W × L × H) filled with a fertilized horticultural substrate (Hanareumsangto, Shinsung Mineral, South Korea) and placed in a growing facility in the plant physiology laboratory of the Department of Environmental Horticulture of Sahmyook University. An environmental control device was used to maintain an average temperature of 24 ± 1°C, and the relative humidity was at kept at 46.3 ± 14.7% in this study.
Experimental Design and LED Light Treatments
The study was conducted using a completely randomized design with six light-emitting diode (LED) light-quality treatments. In the LED white light spectrum, 3000 K white LED (peaks at 455 and 600 nm), 4100 K white LED (peaks at 455 and 590 nm), and 6500 K white LED (peaks at 450 and 545 nm) were used. Additionally, while utilizing LED monochromatic light spectra, red LED (peak at 630 nm), blue LED (peak at 450 nm), and purple LED with a composite spectral distribution (peaks at 450 and 650 nm) were used. In this study, the purple LED used was not a combination of red and blue LED diodes, but rather emitted a composite spectral distribution from each individual diode. Each treatment consisted of three replications, with ten leaves per replication and thirty leaf cuttings per treatment.
Two 1.2 m LED lamps (T5 LED, Zhong Shan Jinsung Electronic, China) with an input power of 20 W were installed per treatment bench. The photosynthetic photon flux density (PPFD) of the LEDs used is presented in Table 1. The distance between the LEDs and leaf cuttings was adjusted to ensure that all treatments received the same PPFD. The spectral power distribution of the different LEDs is shown in Fig. 2. The light intensity levels for all treatments were 40 µmol·m-2·s-1 and a photoperiod of 16 hours light and 8 hours dark was used. During the experiment, the rectangular pots were sprayed with 200 mL of water every two weeks. To prevent the different lights from mixing, each section was blocked with black polyethylene shading films.
Table 1.
Photosynthetic photon flux density (PPFD) values of the light-emitting diodes (LEDs) used in this study
| LED light | PPFD (µmol·m-2·s-1) | |||||
|
350–400 nm (ultraviolet) |
400–500 nm (blue) |
500–600 nm (green) |
600–700 nm (red) |
700–800 nm (far-red etc.) |
Total PPFDz (350–800 nm) | |
| Red | 0.2 | 0.6 | 1.4 | 36.0 | 1.8 | 40.0 ± 0.2 |
| Blue | 0.1 | 37.9 | 0.8 | 0.6 | 0.6 | 40.0 ± 0.2 |
| Purple | 0.2 | 8.2 | 2.7 | 21.9 | 7.0 | 40.0 ± 0.2 |
| 3000 K white | 0.1 | 4.8 | 17.2 | 16.1 | 1.8 | 40.0 ± 0.2 |
| 4100 K white | 0.1 | 7.3 | 18.0 | 13.1 | 1.5 | 40.0 ± 0.2 |
| 6500 K white | 0.1 | 10.6 | 19.1 | 8.9 | 1.3 | 40.0 ± 0.2 |
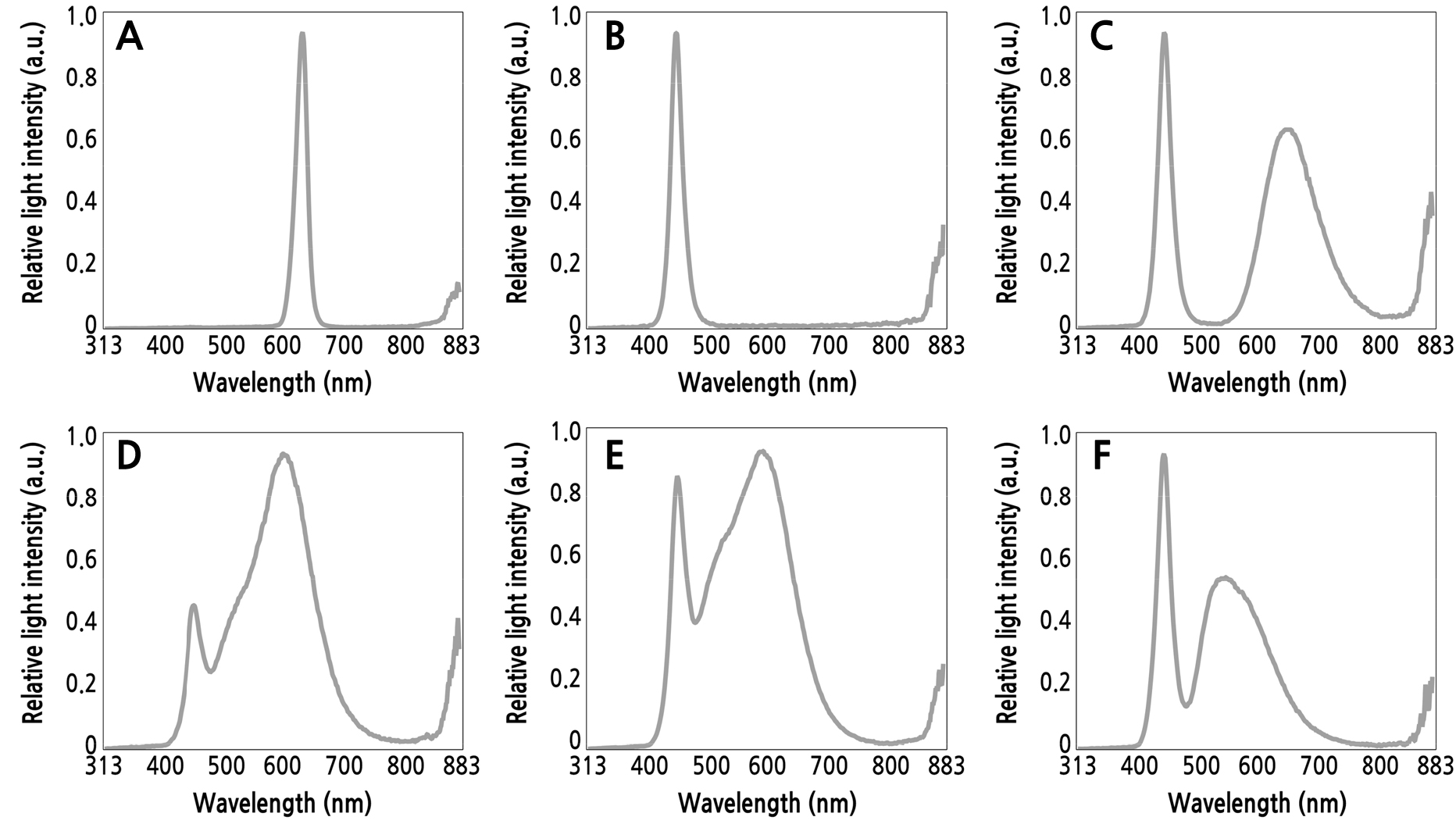
Fig. 2.
Different spectral power distributions of the light-emitting diodes (LEDs) used in this study: A) red LED (peak at 630 nm), B) blue LED (peak at 450 nm), C) purple LED (peaks at 450 and 650 nm), D) 3000 K white LED (peaks at 455 and 600 nm), E) 4100 K white LED (peaks at 455 and 590 nm), and F) 6500 K white LED (peaks at 450 and 545 nm).
Plant Growth Parameters
The survival rate was determined by counting the number of successfully propagated leaf cuttings over the total number of leaf cuttings planted times 100 to obtain the percentage. The percentage of root and shoot formation was determined every 15 days for two months. Other growth parameters, in this case, were the number of leaves, roots, and shoots; the shoot and root length; and the width were recorded after 60 days. Similarly, the moisture content was determined by subtracting the dry weight from the initial weight, which was then divided by the total or fresh weight and multiplied by 100 to obtain the percentage.
Data Analysis
All data collected in the experiment were organized and subjected to a standard analysis of variance (ANOVA) using SAS 9.4 (SAS Institute, USA). To compare the mean differences, Duncan’s multiple range test was applied with a 5% level of significance.
Results and Discussion
Survival Rate and Shoot-root Formation
The survival rate and shooting-rooting success of leaf cuttings from the Pachyphytum species, as affected by the different light qualities, are shown in Fig. 3. These results show that the P. glutinicaule and P. cv. Oviride (P. oviferum × P. viride) leaf cuttings had a 100% survival rate for all light sources. For P. compactum, the highest survival rate was found when the blue and 6500 K light-emitting diodes (LEDs) were used, while for P. viride the highest rate was found in the samples grown under the red and 6500 K LEDs. In P. oviferum, the use of red light resulted in the lowest survival rate (96.6%), while for the remaining light sources, the rate was 100%. Lee et al. (2007) suggested that photoreceptors involved in plant development and regulation are activated by specific light waves, which may vary from one species to another. However, among the different light qualities used here, the use of blue light and 6500 K light resulted in the highest average survival rate in the Pachyphytum species at 98.32% and 97.77%, respectively, with the only difference found in the survival of the P. machucae leaf cuttings.
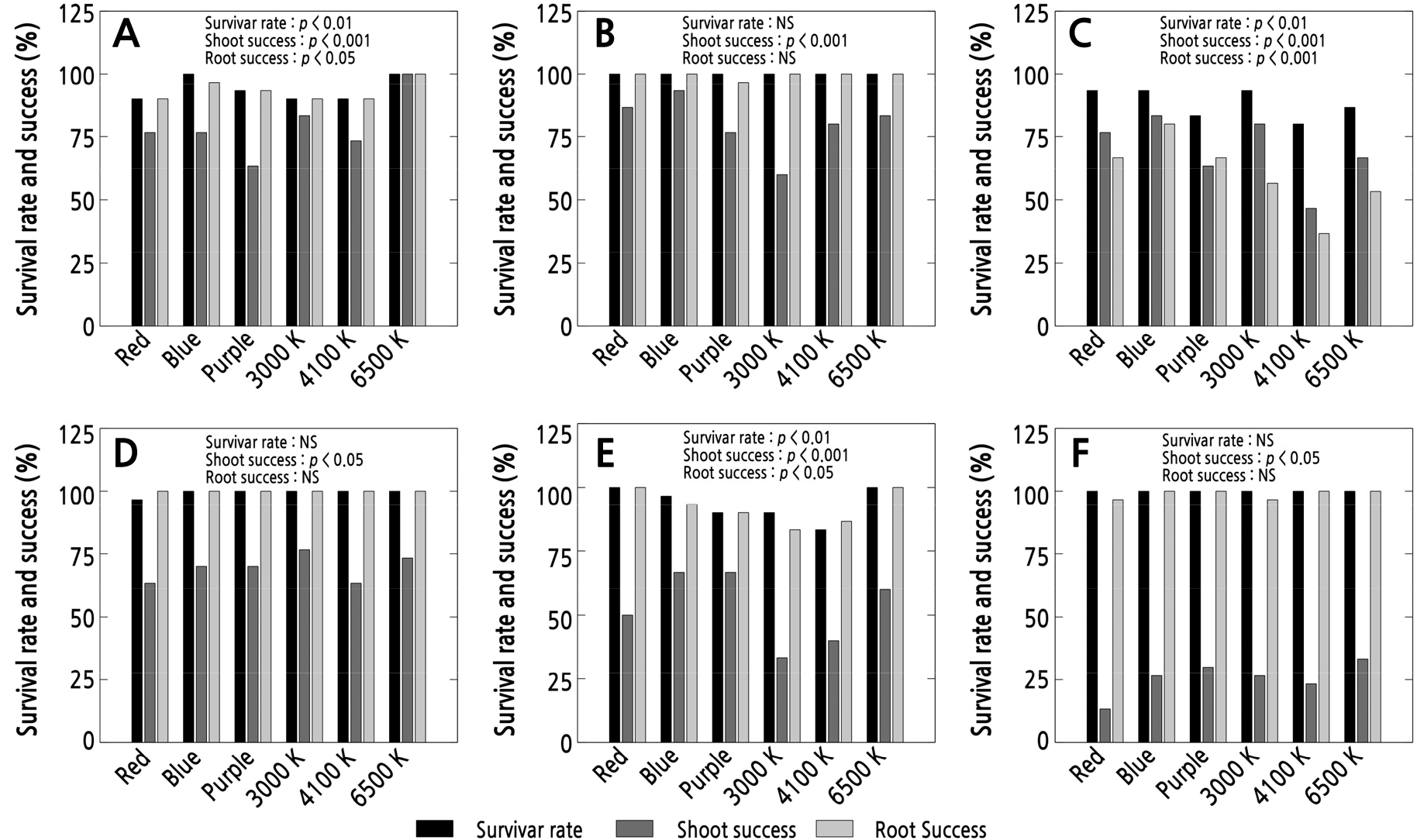
Fig. 3.
Survival rate and shoot and root success of leaf cuttings of Pachyphytum species propagated under different LED light qualities: A) P. compactum, B) P. glutinicaule, C) P. machucae, D) P. oviferum, E) P. viride, and F) P. cv. Oviride. NS indicates no significant difference (p > 0.05) (n = 3).
With regard to shoot and root formation, the Pachyphytum species regenerated in four types, developing into individual plants. Fig. 4 exhibits these developments, showing the leaf cuttings developing only roots, only shoots, producing both shoots and roots, and the development of multiple shoots. Similar results were observed by Cabahug et al. (2016). In all of these cases, these findings were considered as surviving leaf cuttings; however, both shoot and root development outcomes were desirable.
On the basis of the success in shooting, the use of 6500 K light resulted in the highest rate for P. compactum (100%), followed by P. cv. Oviride (33.3%), while the use of blue light resulted in the highest rate for P. glutinicaule (93.3%), followed by P. machucae (83.3%) and P. viride (66.6%). Shoot formation 15 days after planting was observed to be earliest in P. compactum (Fig. 5A), while the other species (Fig. 5C, 5D, and 5F) developed slowly 30 days after planting. The results suggest that the use of blue light led to the highest rate of shooting success for most of the species, i.e., in P. viride, P. glutinicaule, P. machucae, P. and P. oviferum. However, when considering the average shooting success of all species, the 6500 K and blue light treatments led to the highest rates of 69.40% and 69.42%, respectively.
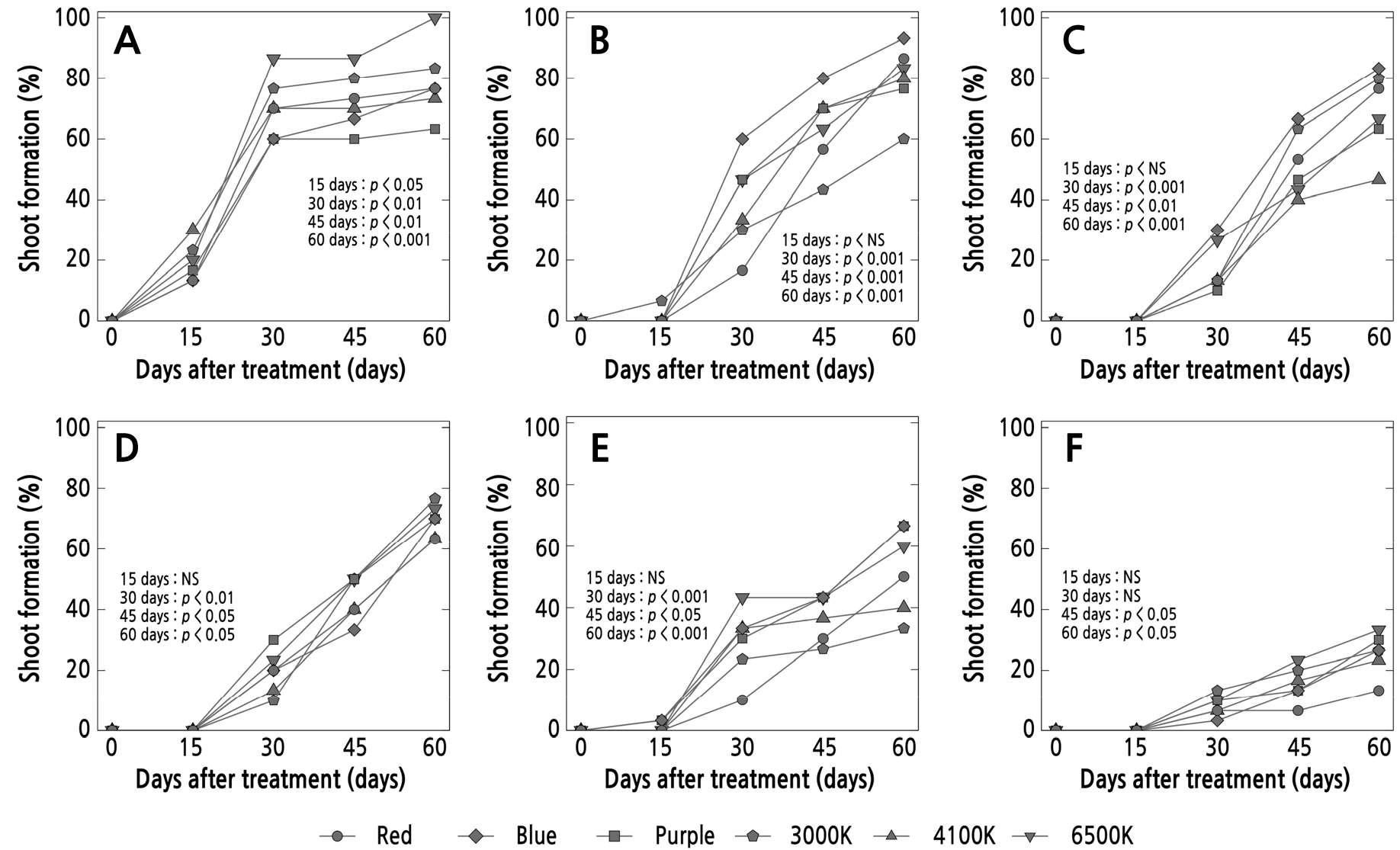
Fig. 5.
Percentage of shoot formation at 15, 30, 45, and 60 days after planting of leaf cuttings of Pachyphytum species under the influence of different LED light qualities: A) P. compactum, B) P. glutinicaule, C) P. machucae, D) P. oviferum, E) P. viride, and F) P. cv. Oviride, propagated under different LED light qualities. NS indicates no significant difference (p > 0.05) (n = 3).
Regarding the results of rooting, most of the Pachyphytum species were observed to develop roots within 15 days, except for P. machucae (Fig. 6). The use of the 6500 K spectrum led to a 100% success rate in rooting in all species, except for P. machucae, wherein the highest rates were observed from leaf cuttings grown under the blue light (80%). In addition, excluding P. machucae, the blue light spectrum was close to achieving a 100% rooting success rate for most species. In the case of P. compactum and P. viride, they exhibited rooting success rates of 96.6% and 93.3% respectively.
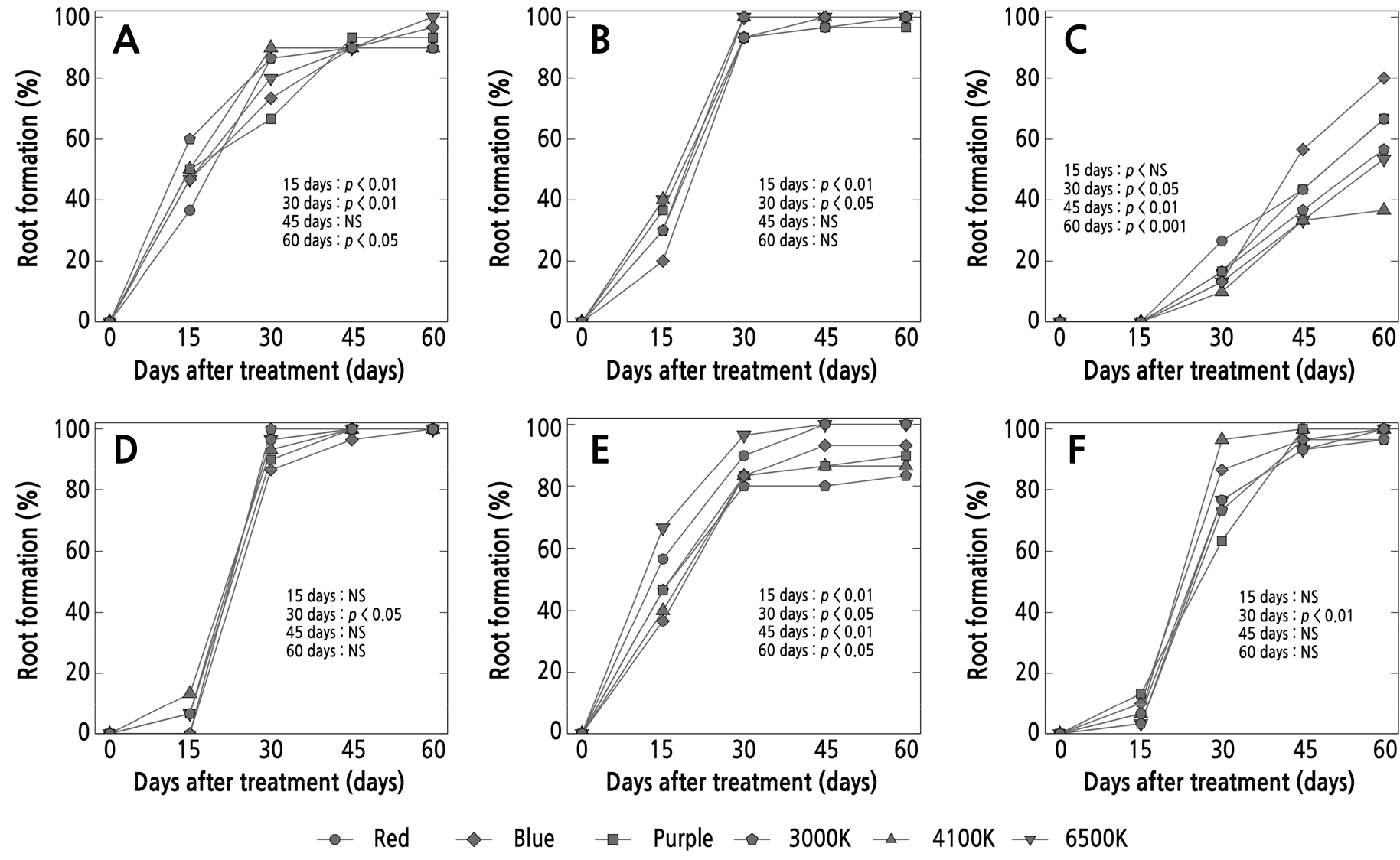
Fig. 6.
Percentage of root formation at 15, 30, 45, and 60 days after planting of leaf cuttings of Pachyphytum species under the influence of different LED light qualities: A) P. compactum, B) P. glutinicaule, C) P. machucae, D) P. oviferum, E) P. viride, and F) P. cv. Oviride, propagated under different LED light qualities. NS indicates no significant difference (p > 0.05) (n = 3).
On the basis of these results, it was noted that the use of 6500 K and blue light spectra provided higher survival and shoot-root formation success rates. The findings from Kettenring et al. (2006) indicated that the Carex species exhibited a higher probability of germination when exposed to 6500 K and blue lights, which was consistent with the results of this study; however, variations in shoot and root formation responses were observed among species, even within the same genus.
Day light color LED, such as 6500 K LED, together with cytokinins, stimulate the accumulation of flavonoids which in turn induces cell elongation and stimulates membrane abundance of PIN1, a secondary transporter facilitating the direction of the flow of auxins (Buer and Muday, 2004; Silva-Navas et al., 2016). Generally expressed in the vascular bundle of the roots, PIN1 is one of the most thoroughly characterized efflux auxin transporters, which help regulate and distribute auxin, a substance that enhances root growth and root elongation (Kleine-Vehn and Friml, 2008). Established roots improve the odds of the development and growth of shoots.
Blue light has also been found to have a stronger effect on plant development in terms of vegetative growth and chlorophyll content, as it is highly absorbed by the photoreceptors, which contributes greatly to shoot formation (Yeh and Chung, 2009). In a study of irregular rooting during the propagation of rosemary stem cuttings, the application of blue irradiance guaranteed the successful and uniform growth of cuttings, an outcome attributed to the upregulation of IAA synthetic genes in the apical and middle nodes (Gil et al., 2021).
The use of both 6500 K and blue light spectra was also found to initiate or stimulate root formation in petunias (Witomska and Ladyzynska, 2002), basil cuttings (Lim and Eom, 2013), and Michaelmas daisy cuttings (Schroeter- Zakrzewska and Kleiber, 2014).
Vegetative Growth and Development
The growth parameters of the Pachyphytum speciesvegetatively propagated from leaf cuttings were significantly affected by the different light qualities (Table 2).
Table 2.
Growth parameters of Pachyphytum leaf cuttings as influenced by different LED light qualities
| Species |
LED light treatment | Shoot parameters | Root parameters |
Fresh weight (mg) |
Dry weight (mg) | ||||||
|
No. of leaves |
No. of shoots |
Length (mm) |
Width (mm) |
No. of roots |
Length (mm) | ||||||
| P. compactum | Red | 7.04 az | 1.50 b | 10.18 a | 8.27 a | 18.26 ab | 21.80 a | 142.30 a | 12.26 ab | ||
| Blue | 7.18 a | 1.77 ab | 4.86 c | 6.27 b | 20.14 a | 20.21 a | 74.28 b | 9.17 b | |||
| Purple | 8.00 a | 1.33 b | 6.11 bc | 7.44 ab | 15.22 b | 22.25 a | 78.88 b | 9.11 b | |||
| 3000 K white | 9.54 a | 2.45 a | 10.12 a | 8.20 a | 20.42 a | 18.69 a | 159.23 a | 15.69 a | |||
| 4100 K white | 9.95 a | 2.09 ab | 9.71 a | 8.76 a | 19.96 a | 18.80 a | 136.53 a | 14.92 a | |||
| 6500 K white | 9.37 a | 2.00 ab | 8.79 ab | 7.96 a | 21.37 a | 20.37 a | 137.58 a | 14.72 a | |||
| P. glutinicaule | Red | 5.16 a | 1.28 a | 5.12 a | 6.04 c | 11.37 a | 28.31 b | 98.96 b | 11.96 b | ||
| Blue | 5.37 a | 1.74 a | 6.22 a | 7.48 bc | 9.44 b | 34.44 a | 120.68 a | 15.65 a | |||
| Purple | 5.68 a | 1.31 a | 5.45 a | 7.90 ab | 8.67 bc | 35.53 a | 116.42 a | 14.85 ab | |||
| 3000 K white | 5.47 a | 1.70 a | 5.82 a | 9.41 a | 9.31 b | 38.34 a | 112.06 ab | 12.68 ab | |||
| 4100 K white | 5.00 a | 1.39 a | 5.78 a | 7.65 bc | 7.51 c | 35.17 a | 108.62 ab | 16.03 a | |||
| 6500 K white | 5.04 a | 1.37 a | 5.50 a | 6.70 bc | 8.06 bc | 35.17 a | 118.96 a | 16.13 a | |||
| P. machucae | Red | 4.81 a | 1.13 b | 3.40 b | 5.86 a | 7.26 a | 10.63 a | 60.00 a | 4.50 a | ||
| Blue | 5.95 a | 1.20 b | 3.29 b | 5.83 a | 6.86 a | 10.73 a | 55.65 a | 3.86 a | |||
| Purple | 4.83 a | 1.22 b | 2.44 c | 4.22 b | 5.94 a | 10.68 a | 20.52 b | 3.10 a | |||
| 3000 K white | 5.31 a | 1.72 ab | 4.31 a | 6.63 a | 6.06 a | 11.20 a | 54.54 a | 2.63 a | |||
| 4100 K white | 5.84 a | 2.30 a | 4.61 a | 6.00 a | 6.90 a | 9.80 a | 43.84 a | 3.76 a | |||
| 6500 K white | 5.73 a | 1.42 b | 3.47 b | 5.31 ab | 5.86 a | 10.46 a | 43.15 a | 3.63 a | |||
| P. oviferum | Red | 9.05 a | 2.11 a | 3.77 a | 6.61 a | 15.60 a | 51.32 a | 173.92 a | 24.17 a | ||
| Blue | 7.05 a | 1.75 a | 4.65 a | 7.15 a | 13.55 a | 53.41 a | 151.72 a | 19.17 a | |||
| Purple | 11.10 a | 1.75 a | 4.90 a | 7.80 a | 14.41 a | 44.31 b | 148.27 a | 25.24 a | |||
| 3000 K white | 6.63 a | 1.72 a | 4.13 a | 5.40 a | 13.93 a | 50.86 a | 131.37 a | 23.51 a | |||
| 4100 K white | 8.22 a | 1.83 a | 4.66 a | 6.72 a | 13.65 a | 49.06 ab | 135.17 a | 27.00 a | |||
| 6500 K white | 9.47 a | 2.14 a | 4.04 a | 6.71 a | 15.48 a | 52.06 a | 148.27 a | 23.24 a | |||
| P. viride | Red | 5.50 a | 1.28 a | 2.86 a | 4.78 a | 10.13 a | 51.96 ab | 72.75 b | 11.55 b | ||
| Blue | 6.31 a | 1.57 a | 3.57 a | 6.31 a | 10.92 a | 44.03 c | 119.25 a | 16.14 ab | |||
| Purple | 5.68 a | 1.73 a | 3.47 a | 5.42 a | 11.07 a | 53.76 a | 111.53 a | 17.03 a | |||
| 3000 K white | 6.44 a | 2.11 a | 4.66 a | 5.22 a | 9.91 a | 48.50 abc | 70.83 b | 13.91 ab | |||
| 4100 K white | 5.45 a | 1.54 a | 4.18 a | 5.00 a | 10.37 a | 47.83 bc | 71.25 b | 11.91 ab | |||
| 6500 K white | 7.47 a | 1.88 a | 4.23 a | 6.29 a | 10.00 a | 50.06 ab | 111.72 a | 16.65 ab | |||
| P. cv. Oviride | Red | 2.01 a | 1.08 a | 2.66 a | 4.66 ab | 9.75 a | 38.89 c | 118.21 ab | 17.92 b | ||
| Blue | 2.71 a | 1.14 a | 3.28 a | 4.71 ab | 8.13 a | 46.44 ab | 103.10 abc | 20.13 ab | |||
| Purple | 2.87 a | 1.16 a | 4.25 a | 6.12 a | 8.32 a | 44.14 bc | 98.57 bc | 20.07 ab | |||
| 3000 K white | 2.71 a | 1.14 a | 3.00 a | 3.71 ab | 9.19 a | 50.92 a | 120.08 a | 23.38 a | |||
| 4100 K white | 2.16 a | 1.08 a | 3.16 a | 4.83 ab | 9.55 a | 40.58 bc | 97.58 bc | 20.10 ab | |||
| 6500 K white | 2.22 a | 1.22 a | 2.03 a | 2.77 b | 8.55 a | 45.31 ab | 90.68 c | 17.72 b | |||
| Significancey | Species (A) | *** | * | *** | *** | ** | *** | *** | *** | ||
| Treatment (B) | NS | NS | * | ** | * | ** | ** | * | |||
| (A) × (B) | NS | NS | ** | *** | ** | ** | *** | ** | |||
These results suggest that the use of different light qualities did not significantly affect the number of leaves. The number of shoots significantly differed with the light spectrum for only two species, wherein the use of 3000 K and 4100 K white light resulted in the highest numbers, at 2.45 and 2.30, for P. compactum and P. machucae, respectively. Similar results were obtained for the shoot length in P. machucae, for which the use of the 3000 K and 4100 K white light spectra resulted in corresponding high values of 4.31 mm and 4.61 mm, but the results did not differ significantly from each other. Similarly, in P. compactum, the aforementioned white lights also yielded high corresponding shoot lengths of 10.12 mm and 9.71 mm. The means, however, did not differ significantly from those resulting from the use of red light, which garnered the highest shoot length value of 10.18 mm. The use of 3000 K white light resulted in the highest shoot width of 9.41 mm for P. glutinicaule. The highest shoot widths for 3000 K and 4100 K for P. compactum (8.20 mm and 8.76 mm) and P. machucae (6.63 mm and 6.00 mm) did not differ significantly from each other. P. cv. Oviride, irradiated with purple light, showed the highest shoot width of 6.12 mm.
Considering how light spectra strongly influence plant growth and physiology outcomes, it is commonly reported that plant leaves absorb 90% of red and blue light illumination (Terashima et al., 2009). Johkan et al. (2010) reported that red light affects hypocotyl expansion, which later develops into the plant’s stem and also promotes leaf expansion. This explains why the P. compactum and P. machucae here showed an improvement in the wider shoot width as compared to those obtained from the other light spectra. These results were also observed in the propagation of in vitro grape plantlets (Heo et al., 2006) and chestnuts (Park and Kim, 2010). Among ornamental plants, plantlets of Protea cynaroides under red light illumination were observed to show stimulated root formation (Wu and Lin, 2012). Similarly, red light treatment significantly increased the number of roots of P. glutinicaule. The authors of the aforementioned study attributes the observed growth to red light due to the low concentrations of endogenous 3,4-dihydroxybenzoic acid and ferulic acid.
On the basis of the results of the root parameters, the use of white light (3000–6500 K) yielded the highest number of roots for P. compactum (19.96–21.37 mm) and the longest roots for P. glutinicaule (35.17–38.34 mm) and P. cv. Oviride (50.92 mm). In contrast, the use of red LED light yielded the highest number of roots for P. glutinicaule. Leaf cuttings grown under blue light yielded the longest root length of 53.41 mm for P. oviferum, while the use of purple LED light resulted in the longest root length (53.76 mm) for P. viride.
White light promotes root growth and physiological activities. In studies focusing on Cunninghamia lanceolata (Xu et al., 2019) and Calibrachoa cuttings (Olschowski et al., 2016), white light was reported to yield the highest number of roots per plant, but this type of light also resulted in the shortest roots. These results were observed to be similar to those of the Pachyphytum species, apart from P. glutinicaule, suggesting that white LEDs lead to the more rapid development of roots. The abovementioned study was based on the hypothesis that the development of numerous roots would support higher values of the shoot parameters in plants, as observed in the majority of the Pachyphytum species. Vigorous rooting supported shoot growth, acquiring the necessary water and mineral nutrition required by the plant. Having an established number of roots ensures that these requirements are properly met and increases the shoot-to-root ratio to a value more suitable for growth (Johkan et al., 2010). Although blue light is generally known to increase root growth due to the generation of relatively more auxins, a study by Hogewoning et al. (2010) suggests that the absence of either light waveband would create photosynthetic or physiological inefficiencies in the plants, which may explain the long roots in P. oviferum.
The plant fresh weight (FW) and dry weight (DW) values suggested that leaf cuttings subjected to white lights (3000–6500 K) weighed more than those subjected to the monochromatic red, blue, or composite spectral distribution of purple light for P. compactum (DW 14.72–15.69 mg, FW 136.53–159.23 mg), P. glutinicaule specifically at 4100–6500 K (DW 16.03–16.13 mg), and for P. cv. Oviride, specifically at 3000 K (DW 23.8 mg, FW 120.08 mg). In contrast, the use of purple LED resulted in the highest dry weight (17.03 mg) and fresh weight (111.53 mg), comparable to those from blue light (119.25 mg) for the fresh weight of P. viride leaf cuttings. However, when computing the moisture content percentage, the use of red light resulted in significantly higher values for P. compactum, P. glutinicaule, and P. cv. Oviride, whereas P. oviferum and P. viride had the highest moisture contents under blue light (Fig. 7). However, it should be noted that the use of purple LED light led to results that were statistically comparable to those of P. glutinicaule and P. viride. For P. machucae, 3000 K LED light led to a higher moisture content compared to the other light settings.
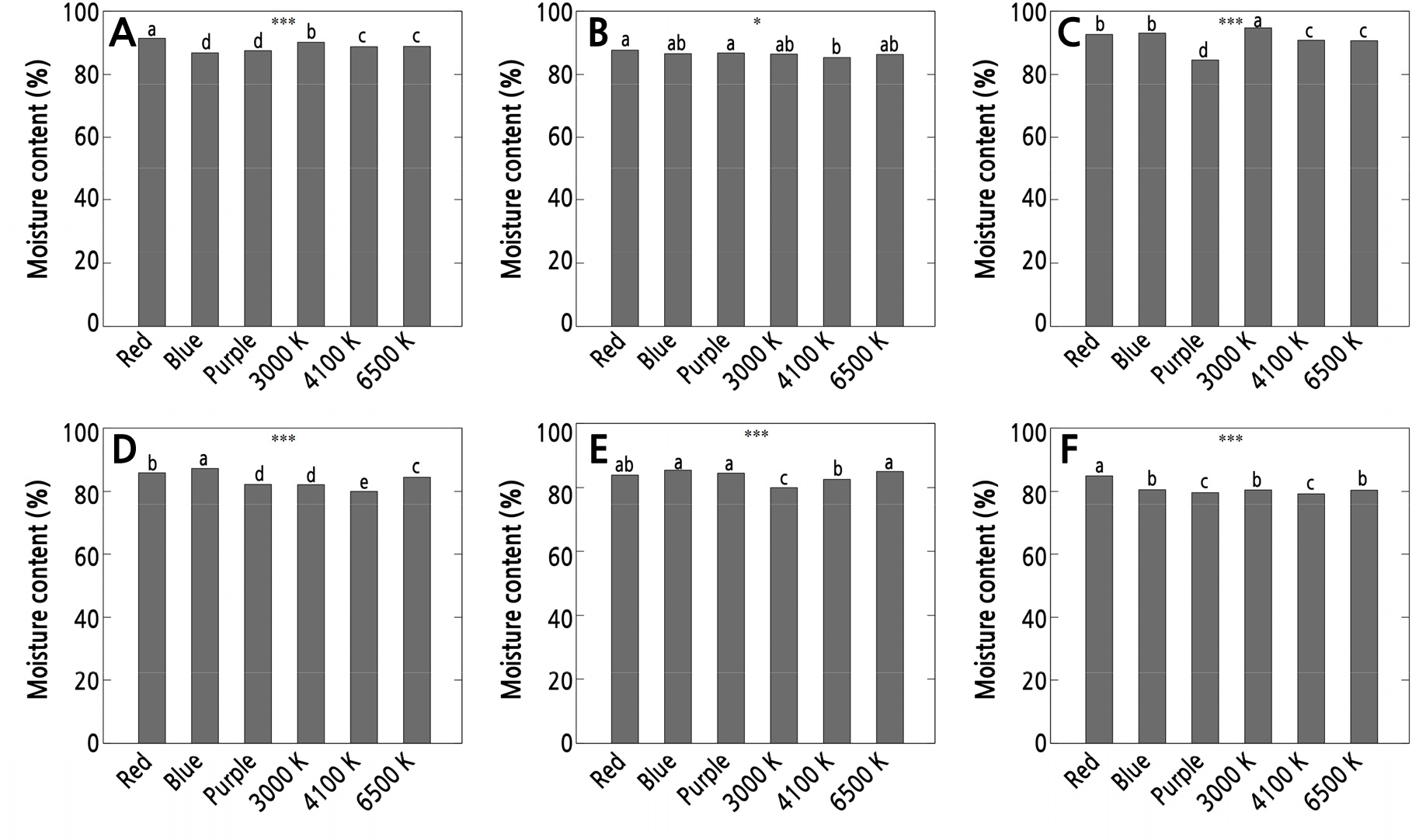
Fig. 7.
Moisture content of Pachyphytum species under the influence of different LED light qualities: A) P. compactum, B) P. glutinicaule, C) P. machucae, D) P. oviferum, E) P. viride, and F) P. cv. Oviride, in responses to different LED light qualities. Asterisks within each figure represent statistical significance of p < 0.05 (*) or 0.001 (***). Different letters indicate significant differences between treatments based on Duncan's multiple range test at p < 0.05 (n = 3).
Monochromatic red and blue lights and a combination of red with blue light reportedly affect plant growth and photosynthesis, thus affecting the accumulation of biomass (Darko et al., 2014; Yoo and Lee, 2017; Oh et al., 2019; Shang et al., 2023). However, these outcomes can vary according to the species (Kasajima et al., 2008). Red light is commonly used in plant factories as it controls stem elongation, and this type of light is involved in the photosynthetic apparatus, leaf expansion, and chlorophyll content of crops (Szopa et al., 2018). This suggests that the light spectrum used yielded good properties in Pachyphytum in terms of how it maintained and accumulated water and promoted cell turgidity. In contrast, it is commonly reported that the use of red light alone may cause photosynthetic abnormalities, deformation of leaves, or altered functionality (Trouwborst et al., 2016; Seif et al., 2021). It has been reported that red and blue lights when used in combination increase the carbohydrate content by a significant amount; this was attributed to the capability of the blue light along with the red light to induce a high photosynthetic rate in ornamental plants and other horticultural crops (Almansa et al., 2014; Garcia-Caparros et al., 2020). Similar studies of Lilium (Lian et al., 2002; Shang et al., 2023), tomatoes (Liu et al., 2011), and Chrysanthemum (Kim et al., 2004) also reported increased fresh and dry weights due to the aforementioned bioassimilation induced by the combination of red and blue light. In this study, the purple LED has been observed to have positive effects on the fresh and dry weight of Pachyphytum, suggesting that there is potential for the purple LED to replace the combination of red and blue LEDs.
To summarize, different light qualities significantly affected the growth and development of Pachyphytum species. The use of blue and 6500 K LED lights was observed to increase survival rates, rooting, and shooting success, while the use of white LED lights showed a positive response on the shoot and root parameters, as well as on FW and DW. However, the use of monochromatic red, blue, and composite spectral distribution of purple lights of these types resulted in the highest values for the moisture content percentage. These results suggest that further investigations of the use of different light qualities at different stages of growth and development of succulent leaf cuttings should be undertaken. The results of this study can provide a benchmark for better propagation methods of succulent cuttings of Pachyphytum as well as other related genera.




