Introduction
Materials and Methods
Plant Material
Germination experiment
Seedling Experiment
Statistical Analysis
Results
Stress Tolerance at Germination
Stress Tolerance of Seedlings
Tolerance Consistency between Germination and Seedling Stages
Comparison and Analysis of Identified Stress Tolerance
Discussion
Introduction
Cucumber (Cucumis sativus L.) is an important vegetable, susceptible to chilling, salinity, and alkalinity stresses (Baysal and Tipirdamaz, 2007; Li et al., 2008; Huang et al., 2009; Li et al., 2013). In China, cucumbers are mainly cultivated in glasshouses from fall to spring; seedlings are sown in mid-to-late September, transplanted in mid-to-late October, and harvested by early-to-mid June the following year (Wang and Yu, 2013). The long growth cycle spans several seasons; therefore, cucumber plants can experience chilling injuries during winter and early spring, suppressing their physiological functions and reducing the cucumber yield and quality (Wang, 1990). A history of inappropriate and/or excessive fertilization has resulted in highly saline glasshouse soils in some sites, causing root damage and accelerated aging in plants, which has ultimately decreased cucumber production.
Many studies have concluded that grafting may help cucumber plants to withstand adverse conditions such as low temperatures and saline environments (Zhu et al., 2008; Colla et al., 2010; Huang et al., 2011; Rouphael et al., 2012; Falic and Ilic, 2014; Li et al., 2015; Xing et al., 2015). The tolerance level of grafted plants to abiotic stresses is usually an intermediate of that of the scion and the rootstock, weighted towards the rootstock phenotype (Schwarz et al., 2010; Huang et al., 2013; Li et al., 2015); therefore, choosing a rootstock that is highly resistant to abiotic stresses is critical for the generation of cucumber grafts more tolerant to chilling and salinity stresses.
The number of pumpkin species accepted by different specialists varies from 13 to 30 (Family: Cucurbitaceae; Genus: Cucurbita), representing a large germplasm resource for important traits such as adaptability, storage longevity, and tolerance to abiotic stresses. Pumpkin is also closely related to cucumber; therefore, pumpkin species that are highly tolerant to various stresses are usually selected as rootstocks for cucumber grafting (Li et al., 2008; Huang et al., 2013; Li et al., 2014a). Li et al. (2014b) analyzed the genetic diversity of 47 pumpkin cultivars commonly used for cucumber grafting by screening 63 morphological markers and 144 pairs of simple sequence repeat markers. Ferriol et al. (2004) investigated the diversity of 47 pumpkin cultivars collected from Central America, South America, and Spain, using sequence-related amplified polymorphism and amplified fragment length polymorphism markers. Li et al. (2013, 2014a) and Sun et al. (2013) evaluated the resistance of pumpkin rootstocks to chilling, heat, and salinity stresses at the germination and seedling stages.
Crops may be exposed to multiple concurrent stresses during cultivation. The combined effects of chilling and salinity stresses have considerable effects on seed germination and seedling growth under field conditions, with saline conditions exacerbating the effects of chilling stress (Mahajan and Tuteja, 2005). Thus, the use of genetic resources that address a single stress may not have any practical application in improving crop production. Determining the germination success of seeds and the success of seedlings in stress conditions is a convenient and efficient way to assess crop tolerance, potentially providing biologically, agriculturally, and ecologically useful information (Levitt, 1980; Scott and Jones, 1985; Schachtman, 2000; Homeijer et al., 2008). In the present study, we investigated the effects of chilling and salinity stresses alone or in combination at the germination and seedling stages of 15 pumpkin cultivars commonly used as cucumber rootstocks. We aimed to identify pumpkin rootstocks that are tolerant to a combination of chilling and salinity stresses, which may be useful for cucumber grafting.
Materials and Methods
Plant Material
A total of 15 pumpkin cultivars commonly used as rootstocks in cucumber grafting were selected (Suppl. Table 1s).
Germination experiment
Pumpkin seeds of uniform size (90 per cultivar) were soaked in warm water (55°C) for 20 min and scrubbed to remove potential germination inhibitors. The seeds were then soaked in 25°C water for 10 h and placed on filter paper in Petri dishes, with 30 seeds on each of the three Petri dishes per treatment. The samples were incubated in a growth chamber (RXZ-430B, 430L; Ningbo Jiangnan Instrument Factory Inc., Ningbo, China) for germination at either 26°C or 15°C (Li et al., 2013) on filter paper soaked with either distilled water or 125 mM NaCl (Sun et al., 2013). The seeds were germinated in the dark at a relative humidity of 80%.
The number of germinated seeds was counted daily until no additional seeds germinated. Germination was considered to have occurred when a 2-mm or longer radicle emerged from the seed, measured using a ruler from the top of the seed to the top of the bud. The germination rate, germination potential, germination index, and vigor index were calculated as described by Schwarz et al. (2010).
Seedling Experiment
Seeds germinated in control conditions (26°C in distilled water) were transplanted into 15-cell polystyrene trays (54 × 28 × 6 cm) filled with commercial organic substrates (vinegar waste compost:peat:vermiculite at a 2:2:1 ratio; Zhenjiang Beilei Technology Development Co., Ltd, Zhenjiang, China). Four trays were used for each cultivar, with 15 plants per tray. Plants were grown in a growth chamber (RXZ-1000B, 1000 L; Ningbo Jiangnan Instrument Factory Inc.) with a 25/15°C (14-h day/10-h night) cycle, under a light intensity of 400 μmol·m-2·s-1 and a relative humidity of 70%. At the two-true-leaf stage, the seedlings were divided into four groups and grown in separate growth chambers. Two chambers had a 25/15°C (day/night) cycle, while the other two were set at 15/5°C (day/night). One growth chamber for each of the temperature conditions was irrigated with distilled water, while the other was irrigated with 80 mM NaCl. The other environmental conditions were as described above.
After 15 d, the plant height, stem thickness, fresh weight, and dry weight of the surviving plants under each treatment were measured. The seedling index (Li et al., 2013; Shah et al., 2015) was calculated using the following formula:
After 15 d, the plant height, stem thickness, fresh weight, and dry weight of the surviving plants under each treatment were measured. The seedling index (Li et al., 2013; Shah et al., 2015) was calculated using the following formula:
seedling index = (stem thickness/plant height) × dry biomass.
Statistical Analysis
Cultivar variability was discounted by normalizing the values for treated samples with those of the controls; therefore, indices were calculated as a relative value:
Relative value (%) = (value under stress/control value) × 100.
Analysis of variance was performed at p ≤ 0.05 using SPSS (IBM Analytics, Armonk, NY, USA). A cluster analysis was performed using the unweighted pair group method with arithmetic mean (UPGMA) algorithm, from which dendrograms depicting similarities among cultivars were drawn and plotted using NTSYSpc (Pal et al., 2013). The degree of consistency was determined based on the matrix correlation (r), which was calculated between germination and seedling stages based on the average distances of morphological indices following stress treatments using the Mantel test (Mantel, 1967) in NTSYSpc. Excellent consistency was defined as r ≥ 0.9, moderate consistency occurred when 0.8 ≤ r <0.9, low consistency was indicated by 0.7 ≤ r < 0.8, and poor consistency was observed when r < 0.7 (Lapointe and Legendre, 1992). Cultivars were defined as stress tolerant only if tolerance was observed during both the germination and seedling stages.
Results
Stress Tolerance at Germination
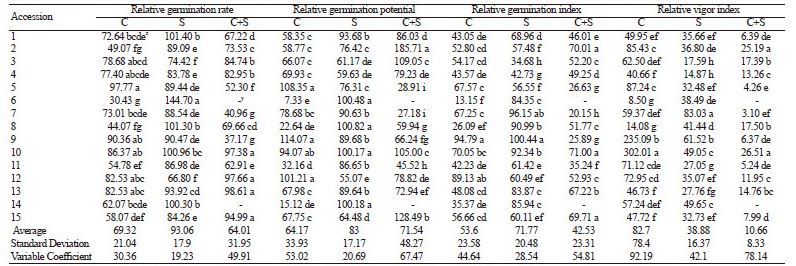
The germination characteristics of the 15 pumpkin cultivars treated with chilling stress, salinity stress, and a combination of chilling and salinity stresses are displayed in Table 1. There were significant variations (p ≤ 0.05) in the relative values for germination rate, germination potential, germination index, and vigor index in each treatment. The germination rates and germination indices of all cultivars during exposure to chilling stress relative to the control conditions were less than 100%. The only characteristics over 100% of the control values during chilling stress were relative germination potential for cultivars 5, 9, and 12, and the relative vigor index in cultivars 9 and 10. The dendrogram generated based on the germination indices revealed three major clusters for the 15 cultivars following chilling stress treatment (Fig. 1A): tolerant, moderately tolerant, and susceptible. Cluster I (tolerant cultivars) included cultivars 1-5, 7, 9, 10, and 12, cluster II (moderately tolerant cultivars) consisted of cultivars 11 and 13-15, and cluster III (susceptible cultivars) comprised cultivars 6 and 8.
zDifferent lowercase letters within a column indicate significant differences at p ≤ 0.05, as determined by a Duncan’s multiple range test.
y‘-’means that stresses treatment resulted in seed not germination, data not measured, which identified as stresses susceptible species.
zzDifferent lowercase letters within a column indicate significant differences at p ≤ 0.05, as determined by a Duncan’s multiple range test.
y‘-’ means that stresses treatment resulted in seedling plant death, data not measured, which identified as stresses susceptible species.
Fig. 1
Dendrogram of the pumpkin cultivar stress tolerances at the germination stage, generated using germination success characteristics. (A) Chilling stress (15°C). (B) Salinity stress (125 mM NaCl). (C) Combination of chilling and salinity stresses (15°C+125 mM NaCl). The 15 stress-treated cultivars were grouped into three clusters: tolerant (I), moderately tolerant (II), and susceptible (III).
Following treatment with saline conditions, the relative vigor indices of all cultivars were less than 100% of the control. The majority of the other measures of germination success decreased following exposure to salinity stress, except for the relative germination rate of cultivars 1, 6, 8, 10, and 14, the relative germination potential of cultivars 6, 8, 10, and 14, and the relative germination index of cultivar 9. Similar to that of the chilling stress experiment, the dendrogram based on the germination indices for the cultivars treated with salinity stress contained three major clusters (Fig. 1B); cluster I (tolerant cultivars) consisted of cultivar 6, cluster II (moderately tolerant cultivars) comprised cultivars 7-11, 13, and 14, while the susceptible cultivars (cluster III) were cultivars 1-5, 12, and 15.
The values for the four germination characteristics decreased following the combined stress treatment, with all relative values for germination below 100% of the control, except the relative germination potential of cultivars 2 3, 10, and 15. The dendrogram generated according to the germination indices for the cultivars treated with chilling and salinity stresses contained three clusters (Fig. 1C); cluster I (tolerant cultivars) consisted of cultivar 2, cluster II (moderately tolerant cultivars) comprised cultivars 1, 3-5, 7-13, and 15, and cluster III (susceptible cultivars) included cultivars 6 and 14.
Fig. 2
Dendrogram of the pumpkin cultivar stress tolerances at the seedling stage, generated using seedling growth characteristics. (A) Chilling stress (15/5°C). (B) Salinity stress (80 mM NaCl). (C) Combination of chilling and salinity stresses (15/5°C+80 mM NaCl). The 15 stress-treated cultivars were grouped into three clusters: tolerant (I), moderately tolerant (II), and susceptible (III).
Stress Tolerance of Seedlings
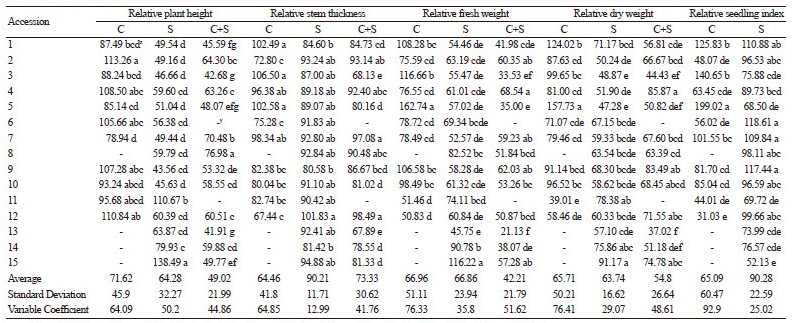
The seedling characteristics of the 15 pumpkin cultivars exposed to chilling stress, salinity stress, and a combination of chilling and salinity stresses are displayed in Table 2. There were significant variations in the values of plant height, stem thickness, fresh and dry weight, and seedling index in the plants that underwent the stress treatments, relative to the control. The values for the seedling characteristics for most cultivars decreased following chilling stress treatment, except for the relative plant height of cultivars 2, 4, 6, 9, and 12, the relative stem thickness of cultivars 1, 3, and 5, the relative fresh weight of cultivars 1, 3, 5, and 9, the relative dry weight of cultivars 1 and 5, and the relative seedling index of cultivars 1, 3, 5, and 7. A dendrogram based on the seedling indices for the cultivars treated with chilling stress defined three major clusters (Fig. 2A); cluster I (tolerant cultivars) contained only cultivar 5, cluster II (moderately tolerant cultivars) consisted of cultivars 1-4, 6, 7, and 9-12, and cluster III (susceptible cultivars) included cultivars 8 and 13-15.
The values of the seedling characteristics for the majority of cultivars decreased under salinity stress conditions compared to the control, except for the relative plant height of cultivars 11 and 15, the relative stem thickness of cultivar 12, the relative fresh weight of cultivar 15, and the relative seedling index of cultivars 1, 6, 7, and 9. A dendrogram based on the seedling characteristics of the 15 cultivars treated with salinity stress contained three main clusters (Fig. 2B); cluster I (tolerant cultivars) contained only cultivar 15, cluster II (moderately tolerant cultivars) included cultivars 11 and 14, and cluster III (susceptible cultivars) contained cultivars 1-10, 12, and 13.
Fig. 3
Matrix comparison based on the average distances of germination and seedling morphological indices according to the Mantel test. (A) Chilling stress. (B) Salinity stress. (C) Combination of chilling and salinity stresses. The correlation coefficients (r ) for chilling stress, salinity stress, and combined chilling and salinity stresses between the germination and seedling stages were 0.26 (p > 0.05), 0.16 (p > 0.05), and 0.17 (p > 0.05), respectively.
Table 3. Appearance of rootstock cultivars at the germination and seedling stages. Cultivars were considered stress tolerant only if tolerance was observed at the germination and seedling stages. 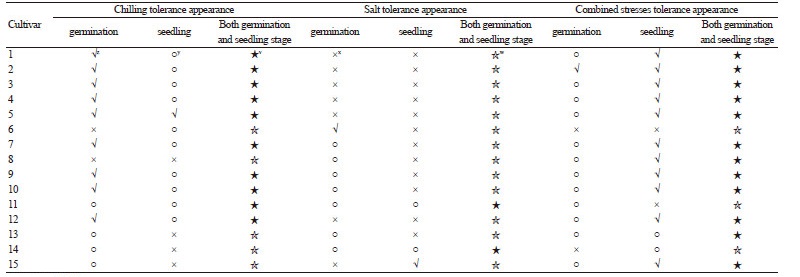 |
z√: high tolerance;
y○: moderate tolerance;
x×: low tolerance;
w☆: low tolerance at the germination and seedling stages;
v★: high tolerance at the germination and seedling stages.
The values for seedling stage characteristics decreased following exposure to chilling and salinity stresses except for the relative seedling index of cultivars 1, 4, 9, 12, and 15. The generated dendrogram for the seedling characteristics of the 15 cultivars exposed to the combined stress treatment contained three major clusters (Fig. 2C); cluster I (tolerant cultivars) comprised cultivars 1-5, 7-10, 12, and 15, cluster II (moderately tolerant cultivars) included cultivars 13 and 14, and cluster III (susceptible cultivars) contained cultivars 6 and 11.
Tolerance Consistency between Germination and Seedling Stages
The correlation coefficients (r) for chilling, salinity, and combined stresses between the germination and seedling stages were 0.26 (p > 0.05), 0.16 (p > 0.05), and 0.17 (p >0.05), respectively (Fig. 3). These results indicate there was no statistically significant correlation between the two stages for the rootstock pumpkin cultivars.
Comparison and Analysis of Identified Stress Tolerance
The tolerance of the various cultivars is presented in Table 3. Cultivars 1-5, 7, and 9-12 were tolerant to chilling stress at the germination and seedling stages, while cultivars 11 and 14 were tolerant to salinity stress, and cultivars 1-5, 7-10, 12, 13, and 15 were tolerant to the combined stress treatment. None of the cultivars were tolerant to all three stress conditions for both growth stages. Cultivar 11 did not exhibit tolerance to the combined stress treatment even though it was tolerant to the individual stress treatments (i.e., chilling and salinity stresses) at the germination and seedling stages.
Discussion
Screening plants based on morphological characteristics is a viable method for cultivar selection, although the optimal procedure is still under debate (Li et al., 2013). Shah et al. (2015) and Kuk and Shin (2007) identified stress-tolerant cucumber cultivars according to their germinability and morphological traits in particular conditions, such as plant growth and leaf area. In the present study, we evaluated the relative plant stress tolerance of 15 pumpkin cultivars used as rootstocks in cucumber grafts, calculating various growth and development indices. Most relative values were highly variable, and overall cultivar performance varied depending on the trait that was investigated; for example, cultivar 11 was tolerant to salinity stress at the seedling stage and had high values for relative plant height and fresh weight, but a low relative seedling index value. In contrast, cultivar 12, which was susceptible to chilling stress at the seedling stage, had high relative plant height values. Our results confirm the findings of Kuk and Shin (2007), who reported that the evaluation of a cultivar using a single characteristic is unreliable.
The UPGMA (Relethford, 1985) is a common method of cluster analysis that enables the consideration of multiple indices. It may be useful for identifying plant resistance while avoiding the problems associated with using a single index (Peeters and Martinelli, 1989). Borowski and Michalek (2014) evaluated six Polish soybean (Glycine max) cultivars and two Canadian soybean cultivars using the UPGMA. They focused on physiological and morphological characteristics at the germination and seedling stages to identify two resistant cultivars, ‘Jutro’ and ‘Nawiko’, as well as one susceptible cultivar, ‘Aldana’. Shahzad et al. (2012) evaluated the salinity tolerance of 129 Pakistani and 58 foreign wheat (Triticum aestivum) cultivars using agronomic traits at the germination and seedling stages, dividing the cultivars into three clusters. These previously reported experiments effectively evaluated the tolerance of crops to chilling and salinity stresses, and identified cultivars with practical applications. We also used UPGMA cluster analyses to evaluate tolerance to chilling, salinity, and combined stresses, dividing the cultivars by their tolerance levels for the three stresses at the two growth stages into three groups; tolerant, moderately tolerant, and susceptible. Our findings may be useful as reference material for choosing environment-appropriate pumpkin rootstocks for cucumber grafting.
Germinating and young seedlings are extremely susceptible to diverse environmental stresses (Çavusoglu and Kabar, 2010). Their physiological changes resulting from exposure to stress can be analyzed accurately and quickly (Sivritepe et al., 2003); thus, the germination and seedling stages can be useful indicators of stress tolerant cultivars. These studies of early plant development do not always identify the most stress-tolerant cultivars, however; salinity tolerance during the germination stage was not significantly related to the decreasing seedling growth rate observed in Australian and Chinese barley (Hordeum vulgare) cultivars (Tajbakhsh et al., 2006), and a study of salinity tolerance in Phaseolus (bean) species during germination and early seedling growth showed that tolerant seeds may not produce tolerant adult plants (Bayuelo-Jimenez et al., 2002). Similarly, there was no relationship between salt tolerance at the germination and seedling stages in a comprehensive study involving over 6,000 barley cultivars from diverse regions (Mano et al., 1996). There were similar problems with chilling stress studies in early plant development; no correlation was observed between the germination and seedling stage in rice (Oryza sativa) and tomato (Solanum lycopersicum) under cold stress (Wang et al., 2011; Ranawake et al., 2014; Liu et al., 2016). In contrast, Liu (1994) concluded that the degree of chilling tolerance during germination was consistent with that at the seedling stage.
Mantel tests are used routinely to assess the significance of the association between two matrices of phenotypic or genetic distances. They are also extensively used to assess how a matrix of genetic or phenotypic distances relates to a matrix of geographical distance. In the present study, a Mantel test revealed the lack of consistency between the germination and seedling stages for all stress treatments, indicating that cultivars that germinate well in stress conditions may not be tolerant at the seedling stage. There are a number of developmental stages plants must complete throughout their lifecycle (i.e., germination, seedling, adult-plant, flowering, and fruiting stages); therefore, stress tolerance at each stage is crucial. For example, cultivar 15 was tolerant to salinity stress at the seedling stage, but was susceptible during germination; consequently, the use of cultivar 15 must be considered carefully in crop production because while the seedlings may be able to grow in saline environments, its poor germination rates will decrease crop yields considerably. Although cultivar 6 may germinate well in saline soils, the poor tolerance of the seedlings to salinity stress means that crop yields will also likely be low if this cultivar is used for commercial crop production.
Stress tolerance is developmentally regulated and stage-specific in many plant genera; therefore, tolerance at one developmental stage may not be correlated with tolerance at other stages (Yildirim and Güvenç, 2006). To identify cultivars suitable for commercial crop production, it is important that stress tolerance at different stages is evaluated. Our results revealed that it is insufficient to use a single stage. Cultivars 1-5, 7-10, 12, 13, and 15 were tolerant to a combination of chilling and salinity stresses at both stages, suggesting that these cultivars may be viable options for crop production. Because of a lack of stress tolerance consistency between the germination and seedling stages, the use of the other cultivars for crop production should be carefully considered.
The physiological basis of chilling stress tolerance involves stabilizing the cell membrane and maintaining membrane lipids in their unsaturated state, as well as preserving the viscosity and fluidity of the cytoplasm (Parkin et al., 1989). The physiological basis of salinity stress tolerance is mainly associated with salt exclusion mechanisms, osmotic adjustment, and ion compartmentalization (Glenn and Brown, 1998; Jumberi et al, 2002). Because of these mechanistic differences, cultivars may exhibit different levels of tolerance to various stresses. Our results indicate that most pumpkin cultivars were not consistently tolerant to individual treatments of chilling or salinity stresses; therefore, the choice of pumpkin rootstock should depend on where the grafted plant will be grown. Rootstocks tolerant to chilling stress should be used in areas that experience low temperatures, while rootstocks tolerant to salinity stress may be suitable options in salinized regions.
Crops are usually exposed to multiple stresses either simultaneously or successively. Alexieva et al. (2003) reported that there are two types of responses to multiple stresses: cross-synergism and cross-adaptation. Cross-synergism refers to the fact that plants are more adversely affected when encountering different stresses simultaneously or successively than when they encounter only one; for example, the combined adverse effects of SO2 and NOx on plants are greater than the effects of only one compound because SO2 may inhibit the nitrate reductase activity that detoxifies NOx (Whitmore and Smith, 1982). In our study, cultivar 6 exhibited poor chilling and salinity tolerance, and was susceptible to the combined stresses. Additionally, cultivar 14, which was tolerant to salinity stress but susceptible to chilling stress, was also considered susceptible to the combined stresses. Cross-adaptation refers to the fact that when plants encounter a specific continuous or intermittent non-fatal stress, their tolerance to that stress will increase, but their tolerance for other stresses will also increase; for example, exposure to salinity stress may increase tolerance to chilling stress in potato (Solanum tuberosum) and spinach (Spinacia oleracea; Ryu et al., 1995), and ultraviolet-B radiation treatment may increase tolerance to chilling stress in cucumber (Caldwell, 1994). In our study, cultivars 13 and 15 were tolerant to the combined stresses, although they exhibited poor chilling stress tolerance and salinity stress tolerance, respectively. Cross-adaptation appears to be based on some physiological effects induced by particular stresses enhancing tolerance to other stresses locally or systematically; however, it is a very complex system with overlapping networks involving a series of environmental stimuli, extracellular signals, membrane receptors, intercellular signals, gene expression,and protein and molecular activities (Balagurova et al., 2000; Bowler and Fluhr, 2000; Mallick et al., 2000). The accurate determination of plant characteristics is therefore crucial when screening for cultivars with good tolerance to combined stresses.
In the present study, we evaluated pumpkin cultivars for tolerance to combined stresses and determined that cultivars 1-5, 7-10, 12, 13, and 15 were tolerant to chilling and salinity stresses. These pumpkin cultivars may be appropriate as rootstocks for cucumber grafting, with practical applications in large-scale crop production; however, cultivars tolerant to combined stresses are very limited, indicating the importance of collecting, generating, and using germplasms with tolerance to multiple stresses.