Introduction
Materials and Methods
Plant Materials
Green Tea Extract Preparation
PPO Inhibitory Activity of Green Tea Extract
DPPH Radical Scavenging Activity of Green Tea Extract
Copper Chelating Activity of Green Tea Extract
Treatments
Weight Loss Rate
Color
Respiration Rate Measurement
Microbiological Analysis
Sensory Evaluation
Statistical Analysis
Results and Discussion
Inhibition of Enzyme Activity and Antioxidant Activity of Green Tea Extract
Weight Loss Rate
Color
Respiration Rate
Microbiological Analysis
Sensory Evaluation
Conclusion
Introduction
The popularity of minimally-processed vegetables has been increased due to not only their health benefits as the fresh produce, but also the consumer demand as the convenience fresh-cut products. Fruits and vegetables are living organisms showing quick quality losses due to physiological aging, biochemical changes, increase in respiration rate, and the proliferation of microorganisms during processing and distribution (Martin-Belloso et al., 2007). In particular, the browning on the surface from cutting and peeling causes the quality loss for fruits and vegetables (Lunadel et al., 2011; Pristijono et al., 2006). It has been variously approached to prevent the browning of fresh-cut fruits and vegetables, such as citric acid, ascorbic acid (Sapers and Miller, 1998; Dong et al., 2000), and oxalic acid (Son et al., 2001) as the reducing agent, and calcium. In addition, sulfite-containing additives have been extensively used as anti-browning agents to keep vegetables and fruits looking fresh. However, consumer awareness of the risks associated with sulfites and increased regulatory scrutiny has created the urgent need for substituting those chemical treatments (Iyengar and McEvily, 1992).
Green tea (Camellia sinensis L.) is a popular beverage exerting the beneficial effects on several diseases due to their antioxidant, anticancer, and antimicrobial activities (Si et al., 2006; Rietveld and Wiseman, 2003; Cooper et al., 2005). Green tea is an excellent source of polyphenols, natural antioxidants used as alternatives to synthetic anti-oxidants (Martín-Diana et al., 2008). These antioxidants, which inhibit the oxidation of organic molecules, are very important for food preservation (Masuda et al., 2003). Green tea has been used to extend the shelf-life of candy jellies (Gramza-Michalowska and Regula, 2007) and cooked pork patties (Nissen et al., 2004). However, only a few studies can be found on the use of green tea in fresh-cut fruits or vegetables. NaCl is a food preservative and commonly used in most culinary habits. It binds water satisfactorily, thus rendering it unavailable for microbial growth on food products (Ehabe et al., 2006). In addition, it has been used to inhibit the browning of fruits and vegetables at home.
The heat treatment is able to inhibit the browning of plants after harvest without using chemicals (Zuo et al., 2004), and can be easily applied to fruits and vegetables by using either hot water dips, vapor heat, or hot dry air. Moreover, it costs less, and is easily able to be scaled-up for commercialization (Silveira et al., 2011; Hofman et al., 2002). The heat treatment can be positively applied for fruits to improve the storability and marketability as it can inhibit the ripening of fruits (Dea et al., 2010; Lurie 1998). They have been successfully used in the Amarillo melon (Aguayo et al., 2008), mangoes (Djioua et al., 2009), rocket leaves (Koukounaras et al., 2009) and shredded carrots (Alegria et al., 2010). This method was also reported to reduce growth phenomena, including sprout development in potatoes (Ranganna et al., 1998), and has been considered as an environmentally-friendly method for prolonging the shelf life of minimally processed products (Hong et al., 2000; Luna-Guzmán et al., 1999).
Burdock (Arctium lappa L.) is a root vegetable used as a folk medicine, diuretic, and antipyretic (Kan, 1981; Chen et al., 2004). Several studies have reported that the root of burdock possesses various pharmaceutical activities, including antibacterial activity (Chow et al., 1997), antioxidant activity (Duh, 1998), and anti-inflammatory activity (Lin et al., 1996). Hence, its demands have been increased, but more considered processed ones due to its inconvenience in handling. In addition, little is known about burdock physiology and its shelf life when processed into fresh-cut burdock slices in order to commercialize a high quality product with an acceptable shelf life as the marketable standard.
This study aimed to evaluate the effect of combined treatment applying heat, green tea extract, and salt for preventing browning and remaining the quality of fresh-cut burdocks while storing at low temperature.
Materials and Methods
Plant Materials
Burdocks (Arctium lappa L.) were harvested from Bongwha- gun, Gyeongsangbuk-do, Korea, transported to the laboratory, and stored at 4°C overnight before processing. Burdocks were selected based on their uniformity of size, color, and defects-free.
Green Tea Extract Preparation
Dried green tea (Camellia sinensis) was purchased from the medicine market in Seoul. Fifty-gram of the powdered sample was then extracted three times with 80% ethanol at 60°C for 6 h. After combining all extracts and evaporating (Rotary Vacuum Evaporator, EYELA, Japan), the freeze-dried powder was used in the experiment.
PPO Inhibitory Activity of Green Tea Extract
The assay was carried out by reacting 1.7 mL of phosphate buffer (pH 6.5, 50 mM), 0.1 mL of substrate (1%) and 0.2 mL of polyphenol oxidase (PPO) (500 units/mg). The increase in absorbance at 420 nm was monitored at 30 s intervals for 5 min, using a microplate reader (M2, Molecular Device, Canada) and the average change in absorbance per min was calculated. One unit of enzymatic activity was defined as the amount of enzyme causing a change of 0.1 in absorbance/min (Dennis and Miller, 1998).
PPO inhibition activity (%) = (1-A/B) × 100
(A: absorbance of sample, B: absorbance of blank)
DPPH Radical Scavenging Activity of Green Tea Extract
1,1-diphenyl-2-picryhydrazyl (DPPH) radical scavenging activity was analyzed by using a modification of the method by Loizzo et al. (2010). Using 96-well microtitre plate, reaction mixtures containing 50 μL of 0.1% sample and 150 μL of 0.3 mM DPPH were placed for 10 min at room temperature. The reacted mixtures were recorded at 517 nm.
DPPH radical scavenging activity (%) = (1-A/B) × 100
(A: absorbance of sample, B: absorbance of blank)
Copper Chelating Activity of Green Tea Extract
The copper binding capacity of extracts was determined according to the method of Wettasinghe and Shahidi (2002) with a slight modification. Tetramethylmurexide (1 mM・L-1) and CuSO4 solutions (5 mM・L-1) were prepared in 10 mM・L-1hexamine-HCl buffer (pH 5.0) containing KCl (5 mM・L-1). Absorbance of the reaction mixtures was record at 460 and 530 nm and the ratio of A460 to A530 nm was calculated. These absorbance ratios were then converted to corresponding free Cu2+ concentrations using a standard curve of free Cu2+ concentration vs absorbance ratio. The difference between the total Cu2+ and the free Cu2+ concentrations indicated the concentration of chelated Cu2+. Copper chelating activity was calculated using the following equation :
Chelating capacity activity (%) = (1-A/B) × 100
(A: absorbance of sample, B: absorbance of blank)
Treatments
Burdocks were washed with running tap water to remove the soil. Then they were peeled and cut into about 3 mm thick slices using a sharp ceramic knife. Each burdocks slice was dipped in the following solutions for 45 sec at 55°C (the selected heat treatment condition through the preliminary experiment) : 0.5% green tea extract, 1% NaCl (sodium chloride, > 99.5%, Sigma-Aldrich Corp., St. Louis, Mo., USA), and 0.5% green tea extract + 1% NaCl. Dipping in water (without browning inhibitors) at 55°C for 45 sec was used as a control treatment. After dipping, each burdock slice was kept over a plastic sieve and dried. 80 ± 5 g of untreated and treated burdock slices were packaged in polyethylene film bags (150 × 200 mm) of 30 μm and sealed. The samples were stored at 4°C for 8 days and weight loss, color, respiration rate, microorganisms, and sensory characteristics were measured.
Weight Loss Rate
Bags were weighed at 0 day and each sampling day during the storage period. Results were expressed as percentage of weight loss relative to the initial weight at 0 day, and the weight loss rate (%) was calculated using the following equation.
Weight loss rate (%) = [(W1 - W2) / W1] × 100 (W1: Initial weight, W2: Weight after storage)
Color
Color measurements were taken with a reflectance Chroma Meter (CR-400, Minolta Co., Japan) on the cut surface of burdock slices. There were ten measurements made per treatment and color determined by measuring Hunter L, a, and b values. The chromaticity difference of each treatment was analyzed using color difference (△E) with the calculation shown below. The Chroma Meter was calibrated on a standard white tile (L = 97.40, a = -0.49 and b = 1.96) before each series of measurements.
△E = (△L2 + △a2 + △b2)1/2
Respiration Rate Measurement
Respiration rates of the burdocks were measured using a closed system method (Hong et al., 2007). The 80 ± 5 g of burdock slices were taken at random from each treatment, placed into airtight glass jars (1 L) with silicon samplings ports and tightly sealed. Jars were stored at 4°C and a 1 mL gas sample from the headspace was taken periodically through an airtight septum and analyzed by an Oxygen Carbon Dioxide Headspace Analyzer (Model 6,600, Illinois Instrument Inc., USA). The mean value was calculated from the samples for each treatment.
Microbiological Analysis
To determine microbial load, a 10 g sample was mixed with 90 mL saline solution in a sterile stomacher bag and shaken for 1 min. Dilutions were made in 0.85% saline solution as needed for plating. Plate count agar (PCA, Difco Lab., USA) was used as the media for mesophilic aerobic counts and Chromocult agar (CM, Merck Co., Germany) was used as the media for coliform group counts. Prepared dishes were incubated at 35°C for 48 h. The microbial counts were expressed as log10 CFU/g. All measurements were performed in three replicates.
Sensory Evaluation
A panel of ten people was trained to recognize and score the quality attributes of the treated burdock slices. Appearance, odor and texture were scored on a nine-point scale, where 1 = complete lacking or soft and 9 = fully characteristic of fresh. On a similar scale for evaluating overall acceptability, 1 = inedible, 3 = poor, 5 = fair (limit of marketability), 7 = good and 9 = excellent.
Statistical Analysis
Statistical analysis of the data was performed using the SPSS Win program (Version 12.0, SPSS Inc., Chicago, USA). Analysis of variance (ANOVA) with Duncan’s multiple range test was used to test the significance of differences among means. Significance was accepted at p < 0.05. Three inde-pendent trials were carried out.
Results and Discussion
Inhibition of Enzyme Activity and Antioxidant Activity of Green Tea Extract
Green tea extract showed high PPO inhibitory activity (79.88%) (Table 1). The flavonoids present in green tea is mainly catechins (flavan-3-ols) (Cabrera et al., 2006). Catechin was probably acted as a competitive inhibitor for PPO because of its structural similarity, which was able to substrate for PPO (Nirmal and Benjakul, 2009). Green tea extract, containing phenolic compound could be used as the natural inhibitor against PPO.
DPPH is used as a free radical to evaluate antioxidant activity of some natural sources, and the degree of its discoloration is attributed to hydrogen donating ability of test compounds, indicating their scavenging potentials (Shimada et al., 1992). DPPH radical scavenging activity of green tea extract was 78.68% (Table 1). Green tea extract was the potential free radical scavengers, as donating their hydrogens and acted as primary antioxidants.
Green tea extract exhibited high copper chelating activity (45.93%) (Table 1). Commercial catechin had the lowest copper chelating activity. Due to copper chelating activity of extracts, they could inhibit PPO by chelating Cu (II) in the active site of PPO, thus leading to the lowered PPO activity (Nirmal and Benjakul, 2011). The capacity of phenolic compounds for chelating metals is strongly dependent on the number of hydroxyl groups in ortho position (Maqsood and Benjakul, 2010). PPO is one of the metalloproteins with two copper atoms in the active site (Jang et al., 2003). The copper chelating activity of extracts was in accordance with PPO inhibitory activity.
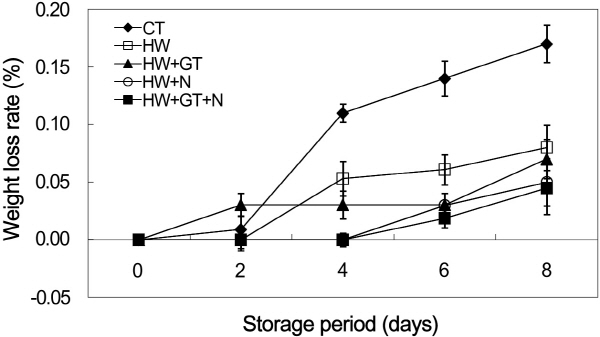
Weight Loss Rate
The weight loss is a crucial quality parameter in the economic stand point of view. In addition, the weight loss strongly impacts appearance as it causes the shrinkage. The weight loss of non-heat treated burdocks was compared to burdock dipped in green tea extract and NaCl with heat treatment during the storage period (Fig. 1). The use of heat treatment with green tea extract and NaCl as anti-browning agents induced to slow down the weight loss during the storage period. For non-heat-treated samples, the weight loss was 0.14% at 8 days of storage period in comparison with the initial weight. Throughout storage, the weight loss of non-heat-treated burdock slices was significantly (p < 0.05) higher than heat-treated burdock slices (2-3.5 times higher). It was reported that the increased weight loss is potentially associated with transpiration rate through the microscopic cracking occurring on the fruit and vegetable surface (Hong et al., 2007). If too much water is transported from the fruit and vegetable turgidity is able to be reduced, and even appeared slightly shriveled. The heat treatment had a significant effect on reducing weight loss during the storage period, compared to non-heat-treated samples. Similar results have been reported for hot water treatments applying to grapefruit (Porat et al., 2000) and minimally processed leeks (Tsouvaltzis et al., 2006). In hot water with the 0.5% green tea extract and 1% NaCl combination solution, samples showed the lowest weight loss (0.04%) at day 8 of storage period, whereas hot water with 0.5% green tea extract or 1% NaCl reached 0.05%. The presence of hydrophobic ingredients in the green tea extract and NaCl as edible coating could be the potential reasons decreasing the water vapor permeability of the surface, thus restricting water transfer.
Color
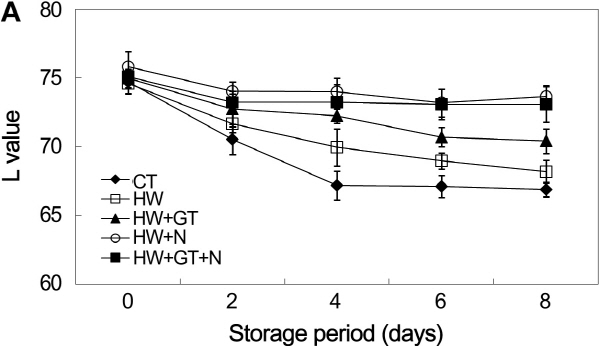
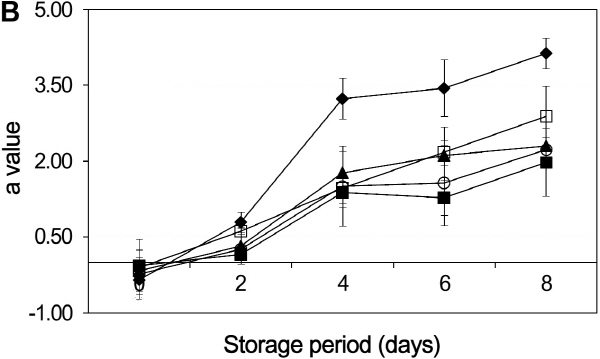
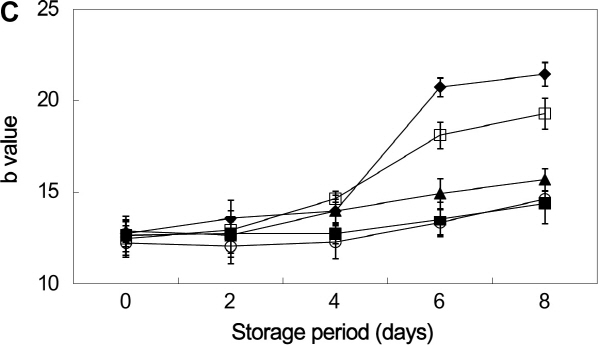
Color is a critical quality of fresh-cut fruits and vegetables, since cutting operations often lead to enzymatic browning (Oms-Oliu et al., 2010). Color changes are also caused from the wound-induced response due to the peeling and slicing (Siomos et al., 2010). The L value, generally depending on the surface reflectivity, is used to express the luminosity of the sample surface, and the a and b values indicate chromaticity on a green (-) to red (+) axis and blue (-) to yellow (+) axis, respectively (Du et al., 2009).
Changes in surface color of non-heat-treated were compared with heat-treated burdock slices, with and without treatments of anti-browning agents, during the storage period. The L and b values were decreased with extending the storage period (Fig. 2a and 2c), which can be related to the appearance of browning. The higher the L value of burdock slices, the brighter the surface. The L value of the hot water + 1% NaCl sample and non-hot water-treated sample were 73.67 and 67.13, respectively, at 8 days of storage period (Fig. 2a). It is believed that the NaCl is interacted with the copper at the active center of the enzyme (Iyengar and McEvily, 1992; Sapers, 1993). Several works have been reported the effect of NaCl on PPO activity. Luh and Phithakpol (1972) claimed that a 3% sodium chloride solution (0.51 M) is used as an inhibitor to hinder enzymatic browning in cling peaches. Therefore, the combined treatment of hot water with anti-browning agents was effective for inhibiting the browning on the surface of burdock slices. In addition, the darkening effect associated with decreased L values may have been linked to fruit and vegetable dehydration (Lydakis and Aked, 2003).
Generally the a value of burdock slices increased during the storage period, and the a value of the controls was highly increased in comparison with other treatments (Fig. 2b). The dipping hot water + adding 0.5% green tea extract + adding 1% NaCl treatment samples reported the slowest trend during storage. As a result, the combined treatment contributed to a slower accumulation of red color on the burdock slices and delayed the browning of burdocks, especially when slices were dipped in a hot water and 1% NaCl combination solution. Polyphenol oxidase (PPO), a key enzyme responsible for browning in fruits and vegetables by catalyzing the oxidation of phenolic compounds, results in this formation of colored melanins (Budu and Joyce, 2003). Green tea extract had a very high PPO inhibitory activity and it seemed to help for the reduction of color changes of burdocks.
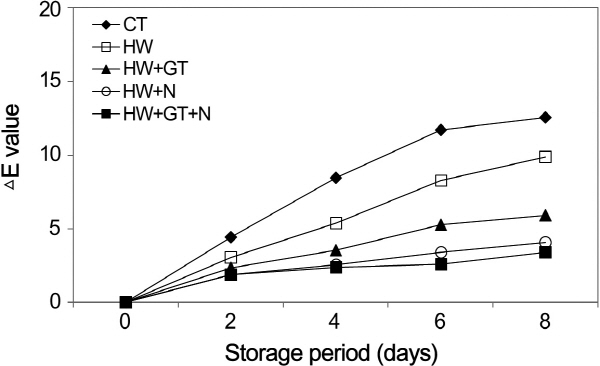
The color difference (△E) was calculated with the collected L, a, and b values. According to the measurement results, the △E value was generally higher in the non-heat-treated burdocks than the heat-treated burdocks (Fig. 3). In addition, the △E value was 3.28 for the burdock slices that were treated with combined heat, green tea extract, and NaCl, showing the smallest change in color. The discoloration was effectively prevented in the sample treated at 45 or 55°C optimally inhibiting PPO (Zuo et al., 2004). Moreover, the combined effect of heat treatment and anti-browning agents on the activities of PPO, POD, and pectin methylesterase (PME) was better than control and thermal treatments under the experiment conditions of this study. The burdock slices that were heat-treated with browning inhibitors showed the smallest change in color during the storage period.
Respiration Rate
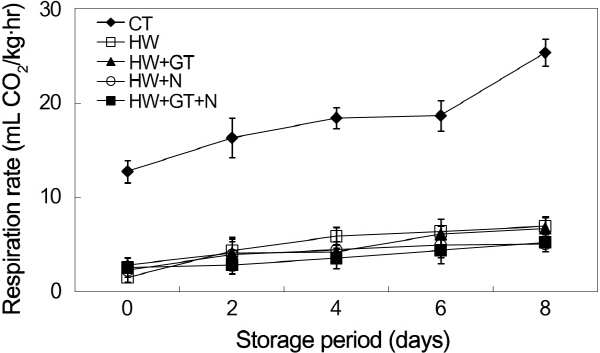
Fresh-cut fruits and vegetables are living tissues, even after treatment. Damaged plant tissues exhibit an increase in respiratory rate (Rico et al., 2007). The respiration rate of slices untreated and treated with hot water + anti-browning agents is shown in Fig. 4. It was shown that all treatments were increased in relation to the microbial growth and general tissue deterioration. Especially, the respiration rates in the non-heat-treated burdock slices were significantly higher than that in heat-treated burdock slices (5 times higher). Fallik et al. (1999) observed that the respiration rate on bell peppers dipped in hot water (55°C for 12 s) was significantly lower than that in untreated ones during 15 days of storage period at 7°C followed by 4 days at 16-18°C. This effect could be a physiological response to heat shock. The samples treated with the hot water and NaCl combination solution showed the lowest respiration rates (5.09 mL CO2/kg・hr) at day 8 days of storage period, while hot water-treated samples reached values of 6.97 mL C02/kg・hr. Similarly, Lamikanra and Watson (2004) and Luna-Guzmán and Barrett (2000) found a reduction in respiratory rates in fresh-cut cantaloupe melon treated with CaCl2 and lactate, in terms of ripening, which delayed the senescence as a result of the combined additive effects of hot water dipping and calcium salts. Hence, the combination of heat and anti-browning agents were very effective in markedly reducing the respiration rate of burdocks.
Microbiological Analysis
The evolution of total microbial load during the storage period of burdocks treated with green tea extract and NaCl with hot water treatment is shown in Table 2. Initial mesophilic aerobic count of 7.05 × 104 CFU/g was found in the untreated control. When compared to non-treated controls, the combination solution of hot water and anti- browning agents detected mesophilic aerobic counts of 4.54 × 102 - 2.38 × 103 CFU/g. The decrease in the microbial load produced by the heat treatment (by 3 logs) is in agreement with Alegria et al. (2009) and Aguayo et al. (2008) where total aerobic bacterial growth on fresh-cut Amarllo melon pieces was reduced by dipping in hot water (60°C for 1 min). Heat treatment was effective for reducing microbial count; furthermore, the heat treatment and anti-browning agent combinations inhibited the growth of microorganisms more effectively than heat treatment alone. In samples from hot water supplemented with anti-browning agents, there were low mesophilic aerobic counts (1.84-6.25 × 105 CFU/g) at 8 days of storage period, whereas samples with hot water treatment alone were detected with 7.18 × 107 CFU/g. According to our results, heat treatment, followed by adding green tea extract and NaCl, was successful for controlling bacterial growth.
Similar results were observed in the reduction of coliform group count on burdock slices (Table 3). Initial coliform group count of 1.88 × 103 CFU/g was found in the untreated control. When compared to non-treated controls, 4.79 × 102 CFU/g were found in the hot water-treated sample, and 1.13-2.57 × 101 CFU/g were found in samples that were treated with hot water and anti-browning agents. Heat treatment is an effective method for reducing microbial counts and effectively inhibits the growth of microorganisms. Heat treatment with green tea extract and NaCl seemed to have a synergistic effect for controlling the microbial growth of fresh-cut burdocks.
Sensory Evaluation
The overall quality and shelf life of fruits and vegetables are reduced by several factors, including water loss, enzymatic browning, texture deterioration, senescence processes, and microbial growth (Rojas-Graü et al., 2008). Sensorial results for appearance, odor, texture, and overall acceptability are shown in Table 4. Ranking scores of all sensory attributes were significantly different among samples (p < 0.05). Untreated samples suffered from poor overall visual quality and surface edge browning.
Appearance scores were decreased during the storage period. At 8 days, burdock slices treated with hot water + green tea extract + NaCl scored the highest (5.5 score). The odor of burdock slices was improved with heat treatment. Moreover, heat treatment with green tea extract and NaCl still was scored within the limit of marketability (5.0 score), and they were no significant differences. Texture decreased during the storage period, showing a similar trend with appearance values. Texture also did not differ between treatments at the beginning of the experiment, however, other treatments had unacceptable scores for consumption except of hot water + green tea extract + NaCl at 8 days of storage period. The lowest texture score was scored (2.0) for the non-heat-treated sample. Texture is a critical quality for consumer acceptability of fresh vegetables. Weigh loss has a key role in stem texture, since tissue dehydration is accompanied by increasing elasticity and fibrous structure (Serrano et al., 2006). At the end of storage, general accep-tability scored similar values for samples treated with hot water + 0.5% green tea extract and hot water + 1% NaCl (4.5 score). The hot water + 0.5% green tea extract + 1% NaCl treated samples generated high overall acceptability values (> 6.0). Browning is one of the main factors causing these changes during the storage period, and it strongly affects the extension of shelf life and consumer purchase decisions in fresh-cut fruit (Oms-Oliu et al., 2010). The organoleptic quality of burdocks treated hot water + green tea extract + NaCl were the best in sensory evaluations.
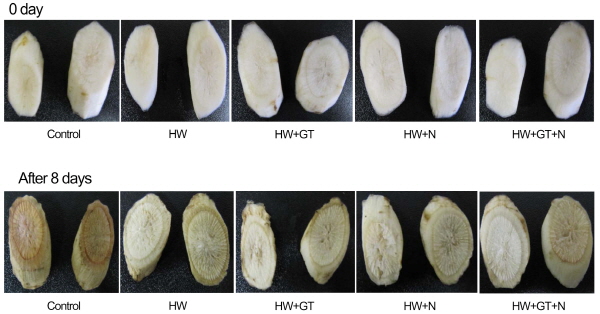
There was a significant difference in appearance score between non heat-treated and heat-treated burdocks (Fig. 3 and 5). Sample treated with hot water showed less change in appearance than the non-heat-treated burdocks. Changes in color and browning were especially found on the surface of the non-heat-treated burdocks. Browning was present mainly in the control sample (without the heat treatment), and reduced in samples with a previous heat treatment. Enzymatic browning was mainly present in control samples without a thermal treatment. Thermal treatment at 50°C probably inhibited the activity of phenylalanine ammonialyase (PAL), the enzyme involved in the first step of the phenyl-propanoid pathway leading to the production of the major phenolic compounds and causing the browning (Pereyra et al., 2005).
Conclusion
Hot water + 0.5% green extract + 1% NaCl treatment could be used as natural postharvest treatments in fresh-cut burdocks to delay postharvest browning and maintain quality. Hot water treatment at 55°C lowered the increase in weight loss rate of fresh-cut burdocks. The burdock slices with heat treatment in green tea extract and NaCl also showed a higher L value and lower △E value than the non-heat- treated burdocks in storage. In addition, the heat-treated burdocks had a lower respiration rate and showed a more gradual speed of microorganism proliferation. The hot water + 0.5% green extract + 1% NaCl solution was effective in reducing the browning of burdock slices. The use of heat treatment and anti-browning agent combinations for processing fresh-cut root vegetables may improve quality through the inhibition of browning, extending the shelf life for the products through an environmentally-friendly technique.