Introduction
Materials and Methods
Plant Materials
Extraction of Volatile Flavor Compounds
Analysis of Volatile Flavor Compounds
Statistical Analysis
Results and Discussion
Volatile Flavor Compounds Found in Korean native Chrysanthemum Species
Alcohols
Ketones
Hydrocarbons
Esters
Acids
Aldehyde
Potential Usage of Korean Native Chrysanthemum Species
Introduction
Korean native Chrysanthemum species are perennial Asteraceae plants that have been used as natural medicine or food as well as an ornamental since the ancient times (Choi, 1992). Since Korean native Chrysanthemum species contain a lot of terpene compounds, they have been used as ingredients in cosmetics (Kim et al., 1995; Matsuda et al., 2002), foods (Ko and Jeon, 2003), and folk medicines (Kim et al., 1998; Nam and Yang, 1995) for a long time. Recently, the antioxidant activation and anti-inflammatory effects of the flavor com-pounds in Korean native Chrysanthemum species have been reported (Bae and Lee, 2008; Sung et al., 2007; Yoon and Cho, 2007), and food processing studies with its healthful benefits have been conducted, such as utilizing Chrysanthemum species for cookie powder (Bae et al., 2009) and rice cake powder (Park and Shin, 1998; Park et al., 2000). Chrysanthemum species also have been proposed as a potential ingredient for herbal cosmetics involving the effects of tyrosinase inhibitory activity associated with anti-oxidant activation, whitening, and moisturizing effects (Bae et al., 2009; Sung et al., 2007; Yoon and Cho, 2007).
There are three main native Chrysanthemum species in Korea: C. zawadskii, C. indicum, and C. boreale, with each species having several subspecies (Lee, 2006). In particular, C. zawadskii has been used for treating various women’s diseases, menstrual irregularity, cold stomach, indigestion, pneumonia, bronchitis, urinary disorders, and neurode-generative diseases (Lee et al., 2008). C. zawadskii also has a great potential as a pharmacological ingredient in shampoo products due to its high anti-bacterial activity against dandruff (Lee and Lee, 2007). Previous research indicated that C. indicum also contains several healthful volatile flavor compounds and various vitamins, which benefit blood flow in long-term use, refresh the body, help digestion, alleviate fever and headaches, lower blood pressure, and offer efficacy such as inhibitory effects against tuberculosis and various viruses (Shin et al., 2004; Sung et al., 2007). C. indicum had been used to make chrysanthemum wine for celebrations during the Chosun Dynasty (AD 1392 to 1897). C. indicum has also been used as a medicinal wine for patients with hypertension (Choi, 1992) and a natural flavoring ingredient in traditional foods (Bae et al., 2009) in Korea. Dried C. indicum was also used to make tea or added to foods (Yoon and Cho, 2007). The benefits of C. boreale were reported to be associated with sedation of the central nervous system, hypotensive effects (Nam et al., 1997), and anti-microbial effects (Cha et al., 2000; Jang et al., 1999; Nam and Yang, 1995). Additional benefits include reductase inhibitory effects of enzyme aldose (Shin et al., 1995), related to anti-cancer (Nam et al., 1997), and diabetic complications (Shin et al., 1995).
With the increasing interest and use of Korean native Chrysanthemum species as pharmacological ingredients, cosmetics ingredients, and in health food, several studies on volatile flavor compounds of Chrysanthemum species have been conducted. Byun et al. (2006) reported that anti-oxidant compounds such as flavonoids, terpenoids, and phenolic compounds were the main functional compounds of the Korean native Chrysanthemum species. Through previous research, it was reported that the main compounds of volatile flavors derived from C. indicum and C. boreale were 1,8-cineole, germacrene-D, camphor, 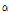 -pinene, and camphene, whereas those from C. zawadskii ssp. latilobum were mainly terpenoids such as camphene and ocimene (Hong, 2002; Jiang et al., 2005; Kim, 1997; Wang and Yang, 2006; Wang et al., 2008). However, despite these studies, species-specific volatile flavor compounds found in Korean native Chrysanthemum species are not clearly compared yet.
-pinene, and camphene, whereas those from C. zawadskii ssp. latilobum were mainly terpenoids such as camphene and ocimene (Hong, 2002; Jiang et al., 2005; Kim, 1997; Wang and Yang, 2006; Wang et al., 2008). However, despite these studies, species-specific volatile flavor compounds found in Korean native Chrysanthemum species are not clearly compared yet.
This study aims to provide a comparison of volatile flavor compounds among 15 taxa of Korean native Chrysanthemum species by analyzing and comparing the species-specific volatile flavor compounds. Results of the study provide clear species-specific volatile flavor compound resource, which could be used as basic data for the germplasms and potential ingredient for functional foods, medicines, and cosmetics.
Materials and Methods
Plant Materials
Fifteen taxa of Korean native Chrysanthemum species were collected from different regions, and grown in green-house at Highland Agriculture Research Center (37°40′N, 128°43′E, altitute 722 m), Rural Development Administration in Pyeongchang, Korea since 2010 (Table 1). From July 12-14, 2011, 20 g of leaves from each Chrysanthemum species were collected. The leaves were stored in -70°C deep freezer and freeze-dried using freezing dryer (FDT- 12012, Operon Co., Gimpo, Korea).
Extraction of Volatile Flavor Compounds
To extract the volatile flavor compounds, simultaneous steam distillation (SDE; Likens & Nickerson type) were done following the extraction method of Schultz et al. (1977). Two grams of freeze-dried leaf sample and 500 mL of distilled water were added to one of the flasks, and heated-reflux was performed for 2 h on a 100°C heating mantle. Meanwhile, the essential oils were extracted by adding 50 mL of n-pentane:diethyl ether mixture (1:1 = v:v) in the other flask and heated-refluxing to 40°C. The extracted volatile flavor compounds were dehydrated using anhydrous sodium sulfate (Na2SO4). Afterwards, they were filtered using filter paper (No. 1, Whatman Limited, Maidstone- kent, UK), and concentrated using 99.9% nitrogen gas. Finally, the concentrated compounds were dissolved in 0.2 mL of diethyl ether and analyzed using gas chromatography/ mass selective detector (GC/MSD) as below.
Analysis of Volatile Flavor Compounds
The extracted volatile flavor compounds were analyzed and identified using GC/MSD as described in Table 2. About 1.0 μL of the extracted volatile flavor compounds was injected into GC (Agilent 7890A, Agilent Co., Wilmington, DE, USA) equipped with HP-5MS capillary column (30 m × 0.25 mm i.d., 0.25 μm film thickness) and was analyzed by split mode (split ratio = 50:1). As for GC analysis conditions, the inlet and detector temperatures were maintained at 250°C, and the helium carrier gas flow rate at 1.0 mL per minute. The oven temperature was kept at 50°C for 5 min, raised by 7°C per min, and then maintained for 30 min at 250°C. For GC/MSD conditions, the electron ionization energy was 70 eV, the ion source temperature was 250°C, and the mass range was 20-400 a.m.u. Regarding verification of the compounds, the mass spectrum obtained using GC/MSD was compared with Wiley 275L database system (Wiley 70, Agilent Co., Wilmington, DE, USA) and mass spectral data from the previous literature (Kondjoyan and Berdague, 1996) for the identification. C5-C20 of alkane (Aldrich Chemical Co., Milwaukee, WI, USA) were used as reference flavor compound materials and expressed in terms of relative peak area (%).
Statistical Analysis
In order to compare the volatile flavor compounds among Chrysanthemum species, analysis of variance (ANOVA) was used to compare the means (n = 4) of the peak area from GC/MSD using SAS program (SAS institute, ver. 9.2, Cary, NC, USA). Levels of significance were calculated using ANOVA test at p ≤ 0.05, 0.01, and 0.001. Values of p ≤ 0.05 were considered to be statistically significant.
Results and Discussion
Volatile Flavor Compounds Found in Korean native Chrysanthemum Species
Identification of the volatile flavor compounds in 15 taxa of Korean native Chrysanthemum species was accomplished by comparing the mass spectra of their components with the Wiley database, and their GC retention time with GC/MSD spectral data (data not shown).
The flavor compounds of 15 taxa of Chrysanthemum species are constituted of six large functional groups, such as alcohols, ketones, hydrocarbons, esters, acids, and aldehyde. On the average, alcohols were the highest flavor compound group (28.7%) in 15 taxa of Chrysanthemum species, followed by ketones (21.2%), and hydrocarbons (13.2%) based on peak area (Table 3). Three functional groups consisted of 63.1% of the total flavor compounds in 15 taxa of Chrysanthemum species. These compositions were dependent on the taxa; alcohols were the most abundant flavor compounds in 8 taxa, and ketones were the most abundant in the other 7 taxa. All the taxa showed negligible amount (< 1%) or no aldehyde compound. Although most taxa contained little (< 2%) to none acid compound, C. indicum and C. lineare contained particularly high acid contents at 4.95% and 10.88% of peak area, respectively. Esters consisted of 0.0-11.3% of peak area depending on the species, and C. zawadskii ssp. acutilobum had the highest esters contents among the taxa.
Comparison of the flavor compounds in 15 taxa of Chrysanthemum species is shown in Table 4. GC chromatogram showed 45 volatile flavor compounds from 12 to 32 volatile flavors each depending on taxa. The six flavor compounds, including sabinene, cis-chrysanthenol, borneol, m-thymol, chrysanthenone, and camphor showed the most significant differences among the taxa (p ≤ 0.001). Sabinene was detected at a significant amount only in C. zawadskii subspecies and C. makinoi. cis-Chrysanthenol was detected only in four taxa of C. zawadskii subspecies such as ssp. naktongense, yezoense, latilobum, and latilobum var. leiophyllum. Significant differences (p ≤ 0.01) in γ-terpinene, 1-octen- 3-ol, linalool, 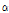 -terpineol, hinesol, and trans-chrysanthenyl acetate were found among the taxa. In particular, γ-terpinene, 1-octen-3-ol and
-terpineol, hinesol, and trans-chrysanthenyl acetate were found among the taxa. In particular, γ-terpinene, 1-octen-3-ol and 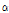 -terpineol were detected only in C. zawadskii subspecies and C. makinoi, while hinesol was detected only in C. indicum subspecies and C. boreale. To investigate the differences in flavor compounds among 15 taxa of Chrysanthemum species, we compared the flavor compounds within the six functional categories of the compounds below.
-terpineol were detected only in C. zawadskii subspecies and C. makinoi, while hinesol was detected only in C. indicum subspecies and C. boreale. To investigate the differences in flavor compounds among 15 taxa of Chrysanthemum species, we compared the flavor compounds within the six functional categories of the compounds below.
Alcohols
Alcohols were the most abundant flavor compounds (19.7-43.0% of peak area) in 8 taxa of Chrysanthemum species, which include three subspecies of C. zawadskii, and C. indicum subspecies, C. boreale, and C. lineare (Table 4). Although a total of 14 alcohols were detected from all taxa, their contents were different depending on the taxa. Chang and Kim (2009) reported that 12 alcohols were detected in flower of C. indicum, including 1,8-cineole, chrysanthenol, isopinocarveol, borneol, terpinen-4-ol, endo--borneol, carveol, eugenol, spathulenol, cedrol, vulgarol B, and 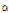 -bisabolol. Choi et al. (2006) reported 11 alcohols in flower of C. boreale, which was more than the number of alcohols detected from the same species in this study. These different results were mostly due to the plant parts where the flavor compounds were extracted.
-bisabolol. Choi et al. (2006) reported 11 alcohols in flower of C. boreale, which was more than the number of alcohols detected from the same species in this study. These different results were mostly due to the plant parts where the flavor compounds were extracted.
|
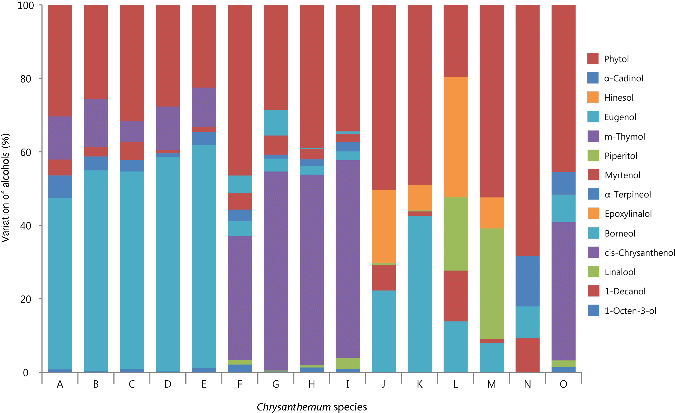
Fig. 1. Variation in the alcohols of volatile flavor compounds among 15 Korean native Chrysanthemum species in greenhouse of highland area. For species name, refer to Table 3. |
All 15 taxa of the common Chrysanthemum species contained two alcohols (phytol and borneol), which consisted of 32-92% alcohols, depending on the species (Fig. 1). Phytol was one of the most abundant alcohol compounds in 6 taxa, with an average of 50% of the total alcohols. In particular, the phytol content in C. lineare was 67.6% of total alcohols. Phytol was identified as an antioxidant compound in Melittis melissophyllum (Maggi et al., 2010) and Eriobotrya japonica (Ham et al., 2012), and it was reported having high anti- microbial activity against Staphylococcus aureus (Inoue et al., 2005). With high phytol contents, these taxa may potentially be a great source for medicinal terpenic compounds.
Borneol was the most abundant alcohol compound in five C. zawadskii subspecies (ssp. acutilobum, ssp. acutilobum var. tenuisectum, ssp. acutilobum var. alpinum, ssp. lucidum, and ssp. coreanum) and C. indicum var. albescens, which had more than 11.5% of peak area (Table 4). Other four C. zawadskii species had less than 1.1% of peak area. In previous studies, Shunying et al. (2005) detected borneol (8.3-18.3% of peak area) from C. indicum flower, however this study showed borneol was detected at 5.4% of peak area in the leaves of the same species, likely due to different plant parts where the flavor compounds were extracted. Borneol was reported to have an antimicrobial activity (Shunying et al., 2005).
All nine C. zawadskii species and C. makino contained 1-octen-3-ol. Interestingly, m-thymol was only detected in the five C. zawadskii subspecies with high borneol contents, whereas eugenol was only detected in the other four C. zawadskii subspecies.
In this study, large quantities of m-thymol were also seen in C. zawadskii ssp. lucidum (4.3% of peak area) and C. zawadskii ssp. coreanum (3.4% of peak area), indicating a potential as an ingredient for development of bactericidal drugs. Moreover, m-thymol has been reported to have a similar medicinal odor as phenol, with antiseptic and disinfectant qualities (Pirbalouti et al., 2013).
Linalool and cis-chrysanthenol were also detected from the four C. zawadskii subspecies and C. makinoi which contained low borneol. Linalool was often used as a raw incense in perfumes and cosmetics (Arctander, 1969), and had been reported to have strong inhibitory effect against 17 bacteria and 10 fungi (Pattnaik et al., 1997). cis-Chrysanthenol was not a common component in Chrysanthemum species as it had not been found in some C. zawadskii and C. indicum species (Hong, 2002; Shin et al., 2004; Woo et al., 2008). It has only been reported that C. indicum flowers (Jung, 2009) had a minor component of cis-chrysanthenol, and aerial part from C. boreale had cis-chrysanthenol as a major component (Hong, 2002). From the results of linalool and cis-chrysanthenol contents, there were two different subgroups within C. zawadskii subspecies, with different compositions of alcohols.
Piperitol and hinesol were only detected in three C. indicum subspecies and C. boreale. These two compounds might be good flavor compounds to identify C. indicum or C. boreale from other species. 1-decanol and 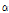 -cadinol were detected only in C. lineare, indicating very specific flavor compounds for C. lineare.
-cadinol were detected only in C. lineare, indicating very specific flavor compounds for C. lineare.
|
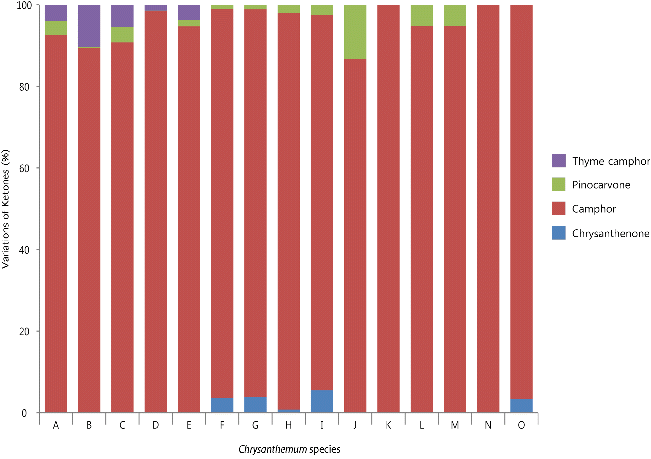
Fig. 2. Variation in the ketones of volatile flavor compounds among 15 taxa of Korean native Chrysanthemum species in greenhouse of highland area. For species name, refer to Table 3. |
Ketones
Ketones were the most abundant volatile flavor compounds in 7 taxa of Chrysanthemum species (13.1-36.1% of peak area), including six C. zawadskii subspecies, and C. makinoi (Table 4). Total of 4 ketones in Chrysanthemum species were camphor, pinocarvone, thyme camphor, and chrysanthenone. Camphor was the most common and the most abundant (86.7-100.0% of total ketones) in these species (Fig. 2).
Camphor was detected in all 15 taxa of Chrysanthemum species investigated. It was the most abundant compound in most species. In particular, C. zawadskii ssp. latilobum and ssp. latilobum var. leiophyllum had high camphor contents with over 30% of peak area (Table 4). Camphor has been reported as an important substance for medicinal purposes (Arctander, 1969); it can alleviate itchiness or suppress cough or food poisoning bacteria (Jang et al., 2010), mostly due to its antimicrobial properties (Tzakou et al., 2001; Viljoen et al., 2003). Previously, several researchers (Chang and Kim, 2008) reported that the most predominant compounds of Chrysanthemum species were camphor, although there were some variations of camphor within Chrysanthemum species (Chang and Kim, 2008; Huang et al., 2001). These results correspond with the findings of previous research in Chrysanthemum species as containing high amounts of camphor.
Pinocarvone was also commonly detected in most Chrysanthemum species except for three taxa: C. indicum var. albescens, C. lineare, and C. makinoi. Pinocarvone, a similar compound to iso-pinocamphone, was reported to be contained in a large amount in Artemisia iwayomogi (Choe et al., 2004).
Interestingly, thyme camphor was detected only in five C. zawadskii subspecies with m-thymol (also had high borneol contents), and chrysanthenone was detected only in four C. zawadskii subspecies and C. makinoi, which had linalool and cis-chrysanthenol. As previously discussed, nine C. zawadskii subspecies could be divided into two groups not only by its alcohols, but also with ketones. Formerly, Chang and Kim (2009) identified 6 ketones (15.3% of peak area) in C. indicum flower, which included camphor, filifolone, chrysanthenone, menthone, pinocarvone, and carvone.
Early researches revealed that chrysanthenone was not detected in Artemisia asictica or Matricaria camomilla in same Asteraceae, and was present only in C. boreale (Chang and Kim, 2009; Choi et al., 2006; Kim et al., 1994). However, this study revealed opposite results, suggesting that its content depedns on the plant organs such as leaf, flower or aerial part. Therefore, further research with plant part- specific comparative studies is needed.
Hydrocarbons
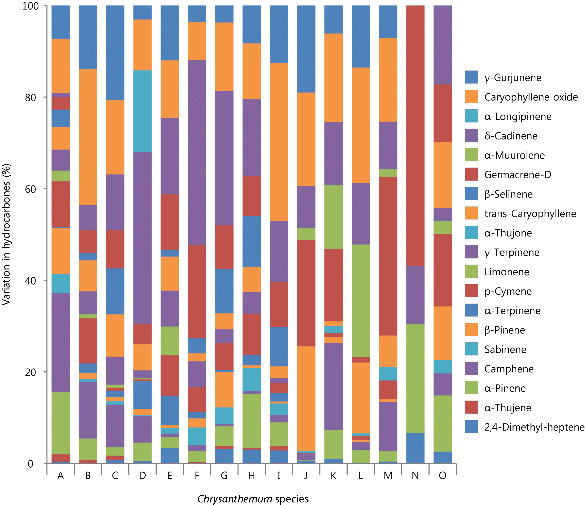
Hydrocarbons had the third most flavor compounds in Chrysanthemum species, which consisted of an average 13.2% of peak area (Table 2). Among 19 hydrocarbons, 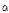 -pinene, camphene, and germacrene-D were the only three compounds that were detected from all species (Table 4 and Fig. 3).
-pinene, camphene, and germacrene-D were the only three compounds that were detected from all species (Table 4 and Fig. 3).
Interestingly, 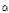 -thujene were detected only in four C. zawadskii subspecies such as ssp. naktongense, ssp. yezoense, ssp. latilobum, and ssp. latilobum var. leiophyllum, indicating specific flavor compounds in Chrysanthemum species. Jang et al. (2010) reported that
-thujene were detected only in four C. zawadskii subspecies such as ssp. naktongense, ssp. yezoense, ssp. latilobum, and ssp. latilobum var. leiophyllum, indicating specific flavor compounds in Chrysanthemum species. Jang et al. (2010) reported that 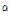 -thujone had outstanding anti-bacterial, anticancer, anti-inflammatory, anti-ulcer, and anti-diabetic efficacies, and C. zawadskii subspecies may have potential ingredient for medicinal products.
-thujone had outstanding anti-bacterial, anticancer, anti-inflammatory, anti-ulcer, and anti-diabetic efficacies, and C. zawadskii subspecies may have potential ingredient for medicinal products.
Amount of 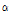 -pinene content varied from 0.01% in C. indicum to 1.27% in C. zawadskii ssp. acutilobum, with an average of 0.7% of peak area (Table 4). Choi et al. (2006) noted that small amounts of
-pinene content varied from 0.01% in C. indicum to 1.27% in C. zawadskii ssp. acutilobum, with an average of 0.7% of peak area (Table 4). Choi et al. (2006) noted that small amounts of 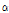 -pinene were detected in C. boreale, which was consistent with this study (0.2% of peak area). The
-pinene were detected in C. boreale, which was consistent with this study (0.2% of peak area). The 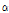 -pinene was often used as a perfume ingredient (Arctander, 1969), and it was known to have anti-inflammatory effects (Jang et al., 2010). Since C. zawadskii ssp. acutilobum, C. zawadskii ssp. latilobum and C. indicum var. albescens were rich in
-pinene was often used as a perfume ingredient (Arctander, 1969), and it was known to have anti-inflammatory effects (Jang et al., 2010). Since C. zawadskii ssp. acutilobum, C. zawadskii ssp. latilobum and C. indicum var. albescens were rich in 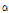 -pinene (peak area > 1%), they could be used as an ingredient for anti-inflammatory treatments. Although
-pinene (peak area > 1%), they could be used as an ingredient for anti-inflammatory treatments. Although 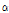 -pinene content of C. lineare was the highest (23.8%), it only had four hydrocarbons since the percentage of total hydro-carbons of the species was relatively low (0.79% of peak area).
-pinene content of C. lineare was the highest (23.8%), it only had four hydrocarbons since the percentage of total hydro-carbons of the species was relatively low (0.79% of peak area).
Camphene was also detected in all 15 taxa of Chrysanthemum species, but its content is also dependent on the species. In particular, C. zawadskii ssp. acutilobum and C. indicum var. albescens contained high camphene content (2.04 and 3.57% of peak area, respectively), and consisted more than 18% of hydrocarbons. Woo et al. (2008) reported that camphene content of C. zawadskii was 5 times higher than the quantity in C. indicum. However, this study showed that C. indicum var. albescens contained high camphene (3.57% of peak area), which was 21 folds of the amount detected in C. indicum (0.17% of peak area). C. zawadskii ssp. latilobum appeared to contain a rather small amount of camphene (0.06% of peak area), indicating that the camphene contents of C. zawadskii were also dependent on the subspecies. These results were different from previous report of Woo et al. (2008), and this is likely due to unclear distinction within C. zawadskii subspecies.
trans-Caryophyllene was detected in 14 taxa of Chrysanthemum species, except in C. lineare. Caryophyllene had been used in spices (Yeon et al., 2012), and it also was detected in Artemisia, Caryopteris, Cinnamonum, Citurs, Eucalyptus, Juniperus, Lavandula, Melissa, Menthe, Pinus, Salvia, Thymus, and Erigeron (Yu et al., 2008).
Germacrene-D was detected in all 15 taxa investigated. Its contents ranged from 0.18% in C. indicum var. acuta to 3.71% of peak area in C. zawadskii ssp. naktongense. Germacrene is a volatile hydrocarbon that exists in many plants, and it was known to exist as five isomers (A, B, C, D, and E). Germacrene-D was found to be an initial substance from the biosynthesis of sesquiterpene derivative (Ahn et al., 2002), and mainly known as a compound with high antibacterial and antioxidant effects (Rivero-Cruz et al., 2006). Woo et al. (2008) reported that C. zawadskii species contained higher germacrene-D than C. indicum species or C. morifolium. Hong (2002) also reported that the germacrene-D of C. boreale was 2.69% of peak area, and this study also showed the high germacrene-D content in C. zawadskii ssp. naktongense at 3.71% of peak area. Within nine C. zawadskii subspecies, germacrene-D contents also varied (0.25-3.71% of peak area) depending on the subspecies. Among three C. indicum subspecies, two species had high content of germacrene-D (2.67% and 2.96% of peak area for C. indicum and C. indicum var. albescens, respectively), and C. indicum var. acuta had the lowest germacrene-D content at 0.18% of peak area.
|
Fig. 3. Variation in the hydrocarbons of volatile flavor compounds among 15 taxa of Korean native Chrysanthemum species in greenhouse of highland area. For species name, refer to Table 3. |
In this study, cadinene was detected in 14 taxa of Chrysanthemum species, except in C. lineare. Among all the hydrocarbons, δ-cadinene content was the highest (16.6-40.3% of total hydrocarbons) in 6 taxa, including C. zawadskii ssp. lucidum, ssp. coreanum, ssp. naktongense, ssp. yezoense, ssp. latilobum, and C. makinoi. This compound was reported to display bioactivities such as insecticidal, antipyretic, anti-inflammatory, anti-bacterial, and anti-cancer (Lee et al., 2012).
Caryophyllene oxide content was the highest among hydrocarbons in four taxa, including C. zawadskii ssp. acutilobum var. tenuisectum (29.8%), C. zawadskii ssp. latilobum var. leiophyllum (34.5%), C. indicum var. albescens (19.3%), and C. indicum var. acuta (25.2%) of total hydro-carbons. C. indicum had particularly high trans-caryophyllene (22.8%) as well.
Esters
Although esters were relatively few (an average of 3.8% of peak area) compared with other functional groups such as alcohols, ketones, and hydrocarbons, Esters were detected in all 15 taxa of Chrysanthemum species (Table 4 and Fig. 4).
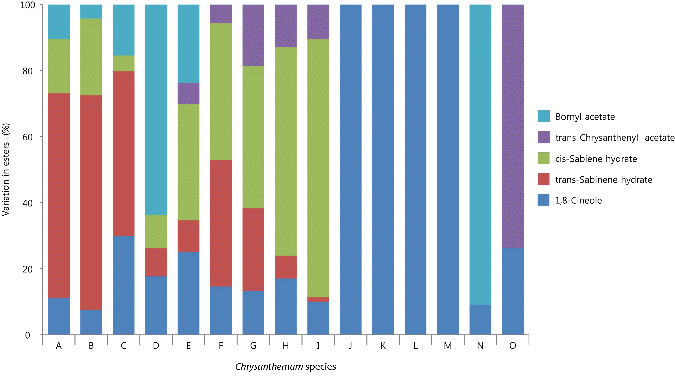
Although 1,8-cineole was commonly detected in all 15 taxa of Chrysanthemum species, it was a relatively small amount among esters found in this study. Previous research indicated that 1,8-cineole was found in C. boreale and C. indicum, but not in Matricaria recutita, which was another genus in Asteraceae, suggesting it as an index compound for the species classification of Chrysanthemum (Chang and Kim, 2008; Choi et al., 2006). In particular, C. indicum var. acuta had the highest 1,8-cineole content at 3.50% of peak area. 1,8-cineole has been reported to have a suppressive effect on occurrences of mutations (Kim et al., 1992) with the recognition of actual chemical treatment effect on breast cancer in mice (Kubo et al., 1992), suggesting a possible potential ingredient for medicinal use.
|
Fig. 4. Variation in the esters of volatile flavor compounds among 15 taxa of Korean native Chrysanthemum species in greenhouse of highland area. For species name, refer to Table 3. |
trans-Sabinene hydrate and cis-sabiene hydrate were detected only in only nine C. zawadskii subspecies. trans- Chrysanthenyl acetate and bornyl acetate were detected with a little amount depending on C. zawadskii subspecies. C. lineare contained bornyl acetate at 2.0% of peak area.
Esters synthesized by short-chain acids and alcohols are a large group of flavor and fragrance compounds, which are used extensively in food, cosmetic, beverage, and pharmaceutical industries (Shu et al., 2011). Although one ester from C. indicum was detected in this study, four esters in same the species were identified as trans-sabinene hydrate, cis-sabinene hydrate, trans-chrysanthenyl acetate, and bornyl acetate (Chang and Kim, 2009)
Acids
Acids were detected in all C. indicum subspecies, C. boreale, C. lineare, and C. makinoi, but not in all C. zawadskii subspecies. Although acid content was low in most of Chrysanthemum species (average 2.1% of peak area), C. indicum had hexadecanic acid at 5.0% of peak area and C. lineare had pentadecanoic acid at 10.9% of peak area (Table 4). In particular, only C. lineare contained pentadecanoic acid compound, whereas other species contained hexadecanoic acid. This specific compound was very unique to C. lineare, and this species had also unique morphological characteristics with different leaf and seed shapes from the other 14 taxa of Chrysanthemum species (Kim et al., 2011). Woo et al. (2008) reported trace amount of acids was detected in C. indicum, but it was not detected in C. zawadskii. However, our results showed that four C. zawadskii subspecies contained hexadecanoic acid, indicating subspecies specific volatile flavor compound contents.
Aldehyde
Aldehyde was detected in all Chrysanthemum species except C. lineare, and only one type (trans-2-hexenal) was identified (Table 4). All the Chrysanthemum species contained very little amount of aldehyde (less than 0.3% of peak area). Choi et al. (2006) also reported that aldehyde content in Chrysanthemum was negligible, which was consistent with this study. Chang and Kim (2009) identified five aldehydes from C. indicum flowers with major compounds being 2- hexanal, safranal, benzealdehyde, and phenylacetaldehyde. However, this study found only one aldehyde from C. indicum leaves, indicating difference in flavor compounds from different part of organ.
Potential Usage of Korean Native Chrysanthemum Species
This study was conducted to compare flavor compounds among 15 taxa of Korean native Chrysanthemum species by analyzing and comparing specific flavor compounds, which could be used as baseline data for the germplasms and potential ingredient for functional foods, medicines, and cosmetics. Korean native Chrysanthemum species showed a considerable variation in volatile flavor compounds in their leaves, and this study may provide a good indication of species specific potential usage for various applications.
Most C. zawadskii subspecies contained 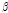 -selinene, which is effective in simulating appetite and a treating nausea and diarrhea (Ravindran et al., 2004). Two C. zawadskii subspecies (ssp. acutilobum var. alpinum, and ssp. lucidum) had the most abundant borneol content (18.5-20.9%), which was reported as a volatile compound with antimicrobial activity (Shunying et al., 2005). C. zawadskii ssp. latilobum and C. zawadskii ssp. latilobum var. leiophyllum contained over 30% peak area of camphor, which was reported as a volatile compound with antimicrobial properties (Tzakou et al., 2001; Viljoen et al., 2003). In particular, C. zawadskii ssp. lucidum and C. zawadskii ssp. coreanum contained higher contents of m-thymol, which had antiseptic and antibacterial qualities. So, both species can be potentially used as a disinfectant in medical fields. Four C. zawadskii subspecies (ssp. naktongense, ssp. yezoense, ssp. latilobum, and ssp. latilobum var. leiophyllum) contained chrysanthenone, which may have additional use as raw material for cosmetics (Arctander, 1969).
-selinene, which is effective in simulating appetite and a treating nausea and diarrhea (Ravindran et al., 2004). Two C. zawadskii subspecies (ssp. acutilobum var. alpinum, and ssp. lucidum) had the most abundant borneol content (18.5-20.9%), which was reported as a volatile compound with antimicrobial activity (Shunying et al., 2005). C. zawadskii ssp. latilobum and C. zawadskii ssp. latilobum var. leiophyllum contained over 30% peak area of camphor, which was reported as a volatile compound with antimicrobial properties (Tzakou et al., 2001; Viljoen et al., 2003). In particular, C. zawadskii ssp. lucidum and C. zawadskii ssp. coreanum contained higher contents of m-thymol, which had antiseptic and antibacterial qualities. So, both species can be potentially used as a disinfectant in medical fields. Four C. zawadskii subspecies (ssp. naktongense, ssp. yezoense, ssp. latilobum, and ssp. latilobum var. leiophyllum) contained chrysanthenone, which may have additional use as raw material for cosmetics (Arctander, 1969).
C. indicum subspecies and C. boreale contained 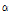 - thujone, which had outstanding anti-bacterial, anti-cancer, anti-inflammatory, anti-ulcer, and anti-diabetic efficacies (Jang et al., 2010). In particular, C. indicum var. albescens have a high utility in making perfumes, since it showed camphene content that was 21 times higher than C. indicum. Since C. indicum var. albescens also contains
- thujone, which had outstanding anti-bacterial, anti-cancer, anti-inflammatory, anti-ulcer, and anti-diabetic efficacies (Jang et al., 2010). In particular, C. indicum var. albescens have a high utility in making perfumes, since it showed camphene content that was 21 times higher than C. indicum. Since C. indicum var. albescens also contains 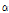 -pinene and offers anti-inflammatory effect, its value in cosmetics is expected to increase. C. indicum var. acuta contained a fairly high content of 1,8-cineole, which has an inhibitory effect on mutagenesis. Also, since 1,8-cineole has outstanding chemical effect (Kubo et al., 1992) in actual treatment of mice with breast cancer, this species may have a great potential as a medicinal plant material. C. lineare contained only penta-decanoic acid compound, whereas other species contained hexadecanoic acid. This species had also unique morphological characteristics from other 14 taxa of Chrysanthemum species. C. lineare was the most different species in hydrocarbon contents among the 15 taxa of Chrysanthemum species. It only contained three common compounds (
-pinene and offers anti-inflammatory effect, its value in cosmetics is expected to increase. C. indicum var. acuta contained a fairly high content of 1,8-cineole, which has an inhibitory effect on mutagenesis. Also, since 1,8-cineole has outstanding chemical effect (Kubo et al., 1992) in actual treatment of mice with breast cancer, this species may have a great potential as a medicinal plant material. C. lineare contained only penta-decanoic acid compound, whereas other species contained hexadecanoic acid. This species had also unique morphological characteristics from other 14 taxa of Chrysanthemum species. C. lineare was the most different species in hydrocarbon contents among the 15 taxa of Chrysanthemum species. It only contained three common compounds (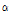 -pinene, camphene, and germacrene-D) and 2,4-dimethyl-heptene, with the least amount of hydrocarbons at 3.3% of the peak area.
-pinene, camphene, and germacrene-D) and 2,4-dimethyl-heptene, with the least amount of hydrocarbons at 3.3% of the peak area.
Overall, Chrysanthemum species were found to have great potential in multiple applications such as cosmetic raw material or food and medicinal ingredients; especially, Korean native Chrysanthemum species had species-specific volatile flavor compounds in the leaves. Therefore, selection of the right species is important to extracting specific volatile compounds. Nevertheless, comparing volatile flavor compounds from different plant organs and finding proper processing methods to extract the useful volatile compound would be done in the future.


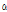 -Terpineol
-Terpineol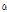 -Cadinol
-Cadinol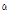 -Thujene
-Thujene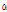 -Pinene
-Pinene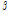 -Pinene
-Pinene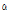 -Terpinene
-Terpinene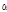 -Thujone
-Thujone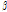 -Selinene
-Selinene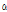 -Muurolene
-Muurolene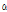 -Longipinene
-Longipinene