Introduction
Materials and Methods
Plant Material
Dry and Wet Conditions in Harvest and Shipping Stages
NaOCl and ClO2 in Wet Shipping Solution
Results
Dry and Wet Conditions in Harvest and Shipping Stages
NaOCl in Wet Shipping Solution
ClO2 in Wet Shipping Solution
Discussion
Dry and Wet Conditions in Harvest and Shipping Stages
NaOCl and ClO2 in Wet Shipping Solution
Introduction
South Korea’s total export volume of floricultural crops was 2,973 tons in 2016, about 8% of which was chrysanthemum. The export volume of floricultural crops to Japan alone was estimated at 1,989 tons, accounting for 67% of the total volume and 99.5% of the total chrysanthemum export (www.kati.net). However, export volumes of floricultural crops including chrysanthemum continue
to decline. Causes of this decline include economic slowdown in the import country (Japan), weakening of the currency of the import country, fierce competition with Southeast Asian countries, reduced profits due to increased management costs, and deteriorated quality of exported floricultural crops. If cut flowers are not managed adequately during the harvest, storage, and transport stages of export, their quality will be reduced, often manifested as non-flowering, early-flowering, poor expression of flower colors, and shortened vase life (Lee and Kim, 2016). In the case of cut roses, the current declining share in the export market has been reported to be due to flower quality deterioration and shortened vase life (In et al., 2016).
As most cut flowers are transported in dry conditions from harvest to export, freshness is often degraded at the customers’ stage, with a loss rate of 10-50% (Yang et al., 2014). For example, cut lilies are exported to Japan using a dry shipping method. Lee and Kim (2016) have reported that when cut lily cultivars ‘Medusa’ and ‘Siberia’ are treated with a wet shipping solution for 22 h immediately following harvest, these cut flowers have a lesser degree of stem bending and extended vase life compared to those transported dry shipping condition. Lim et al. (2016) have also reported that when the cultivar ‘Medusa’ is transported using wet shipping conditions in a simulated export environment after harvest, vase life is extended by 2 days. The transport of cut roses under wet shipping conditions for 48 h can maintain high fresh weight and significantly extend vase life compared to dry shipping conditions (Hu et al., 1998). In particular, wet shipping using a mixed solution of 8-hydroxyquinoline sulfate (8-HQS) and fructose has been shown to maintain freshness of cut flowers (Hu et al., 1998).
Many studies have reported post-harvest quality management and extension of vase life of cut chrysanthemums, specifically when cut flowers are treated with NaOCl (Lee and Lee, 2015; Yoo et al., 2016), ClO2 (Kim et al., 2014; Macnish et al., 2008), methanol (Petridou et al., 2001), or silver thiosulfate (STS) solution (Lee et al., 1996), benzyladenine (BA) (Lee and Lee, 2013; Suh and Kwack, 1994), gibberellic acid (GA3) (Suh et al., 2011), and sucrose (Yoo et al., 2016). These treatments can improve the quality and vase life of cut flowers. NaOCl (a fungicide), aluminium sulfate, or citric acid solutions mixed with sucrose and BA can reduce leaf chlorosis and extend vase life (Jain et al., 2009; Jain et al., 2014; Reyes-Arribas et al., 2000; Yoo and Roh, 2015b). Indeed, most studies on the quality and vase life of cut chrysanthemums have been conducted on pretreatment methods and holding solutions, but relatively few studies have been conducted using wet shipping conditions.
Most cut chrysanthemums for export to Japan are transported by ship under dry condition rather than by air. It takes an average of 3-5 days for cut flowers to arrive at a flower auction in Tokyo, Japan after being harvested at farms in South Korea (Lee and Lee, 2015). Since cut chrysanthemums are typically transported in conditions wherein they cannot absorb moisture for up to 5 days, withering of flowers and leaves will occur. Therefore, the objective of this study was to compare the quality of cut flowers from the standard chrysanthemum cultivar ‘Baekma’ transported under wet and dry conditions and to determine the efficacy of NaOCl and ClO2 disinfectants in wet shipping solutions to improve freshness and extend the vase life of cut flowers after export.
Materials and Methods
Plant Material
The plant material used in this study was the standard chrysanthemum ‘Baekma’ cultivated in a chrysanthemum farm in Jeonju city of Jeollabukdo Province, South Korea. Simulating export conditions to Japan, cut flowers were harvested at flowering stage 3 wherein the outermost ray flower covers three fourths of the flower buds (Yoo and Roh, 2015a).
Dry and Wet Conditions in Harvest and Shipping Stages
The standard chrysanthemum ‘Baekma’ was harvested on July 15, 2017. Stems were cut to 80 cm and leaves were removed up to 20 cm from the lower end of each stem. Both fresh weight (in g) and flower diameter (in cm) were measured. Immediately after the measurement, flowers were treated with dry and wet harvest conditions. Flowers were transported from the farm to Mokpo National University via refrigerator truck at 5°C for 2 h and then stored at 5°C in a cold storage room for 22 h. Under dry harvest conditions, cut flowers were placed in a corrugated paperboard box immediately upon harvest. Under wet harvest conditions, cut flowers were placed in a plastic container filled with tap water (pH 6.7, EC 0.1 dS‧m-1) immediately upon harvest.
To capture transport conditions and the actual time required for cut flowers to reach a flower auction in Tokyo, Japan, this study conducted a simulated transport at 5°C for 4 days under both dry and wet shipping conditions. Under dry shipping conditions, cut flowers were laid horizontally in a corrugated paperboard box (98 cm × 38 cm × 19 cm). Under wet shipping conditions, cut flowers were placed vertically in a wet container filled with tap water inside a corrugated paperboard box (23 cm × 21 cm × 78 cm). After 4 days of simulated transport, the fresh weight and flower diameter of cut flowers were measured.
Subsequently, fifteen cut flowers per treatment were put into a 2 L plastic jar filled with a holding solution of 50 mg‧L-1 NaOCl (3 replicates). These cut flowers were placed in a controlled environmental chamber at 20°C, with relative humidity between 50-60%, light intensity of 70 µmol‧m-2‧s-1, and photoperiod of 12 h. To evaluate the quality of cut flowers, the method of Yoo and Roh (2015a) was applied. Fresh weight and flower diameters were measured every 4 days while the vase life was examined daily. Chlorophyll fluorescence was also measured to determine differences in stress levels among cut chrysanthemum subjected to dry and wet conditions during harvest and shipping stages. A portable chlorophyll fluorescence analyzer (FluorPen FP100, Photon Systems Instruments, Czech Republic) was used to measure chlorophyll fluorescence of the fifth upper leaf of the cut chrysanthemum in holding solution.
NaOCl and ClO2 in Wet Shipping Solution
The standard chrysanthemum cultivar ‘Baekma’ was harvested on August 15, 2017. Immediately after measuring fresh weight and flower diameter, cut flowers were placed into a wet container filled with tap water (pH 6.5, EC 0.1 dS‧m-1) inside a corrugated paperboard box. Flowers were transported from the farm to Mokpo National University in a refrigerator truck at 5°C for 2 h and then stored at 5°C in a cold storage room for 22 h. Simulated transport was conducted using the same method as above inside a cold storage room at a temperature of 5°C for 4 days to mimic export conditions to Japan. Transport under wet shipping conditions was simulated by placing cut flowers in wet containers treated with NaOCl solution at different concentrations (0, 25, 50, 100, and 200 mg‧L-1) or with ClO2 solution (Vital OxideⓇ, Danbibio Ltd., Korea) at different concentrations (0, 0.5, 1, 2, and 4 mg‧L-1) inside corrugated paperboard boxes. After 4 days of simulated transport, fresh weight and flower diameters of the cut flowers were examined. Quality evaluation of cut flowers in holding solution was conducted using the method described above.
To examine the anti-bacterial effect of NaOCl and ClO2, solutions were collected after simulated transport for 4 days in cold storage, diluted 1:100 with tap water and plated on 3M™ Petrifilm™ (3M, USA) count plates. After incubation at 35°C for 2 days, bacterial colony counts were performed.
Results
Dry and Wet Conditions in Harvest and Shipping Stages
Cut flowers stored in dry conditions at 5°C for 24 h after harvest showed that fresh weight was reduced to 94.2% while flower diameter was increased slightly to 103% (Fig. 1B). Cut flowers treated with wet conditions after harvest showed that fresh weight was increased to 104.3% and flower diameter was increased to 109.9%, both of which were higher than under dry harvest conditions.
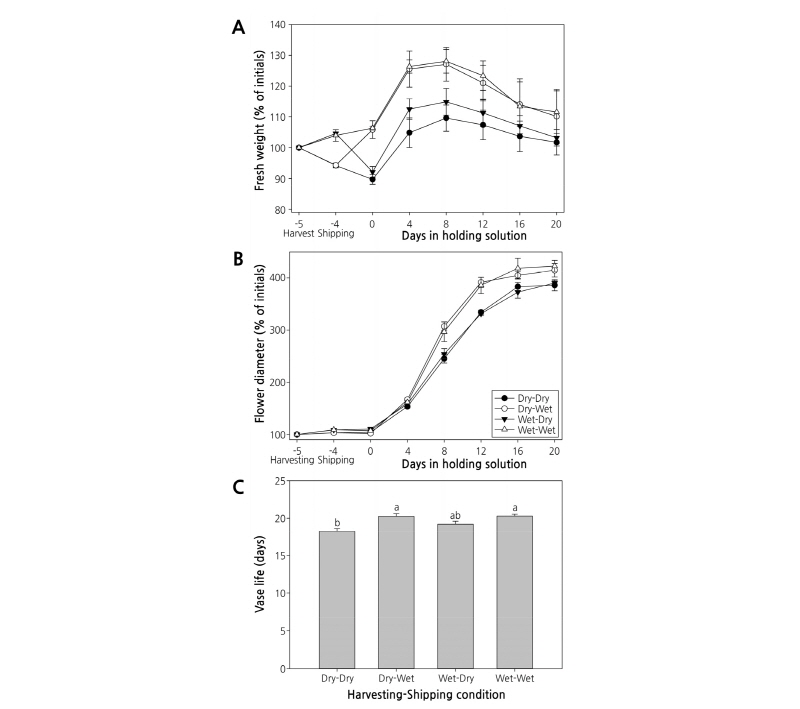
Fig. 1. Changes in fresh weight (A), flower diameter (B), and vase life (C) of cut flowers of the standard chrysanthemum cultivar ‘Baekma’ in dry and wet conditions during harvest and shipping stages. Vertical bars indicate standard error (n =3). Lowercase letters indicate mean separation at p < 0.05 by Duncan’s multiple range test.
After Dry-Dry and Wet-Dry treatments, fresh weights decreased to 89.7% and 92.1%, respectively (Fig. 1A). Some leaves withered (Fig. 2A-D). After Wet-Wet and Dry-Wet treatments, fresh weights increased to 106.4% and 105.9%, respectively, showing no significant difference between the two treatments. Flower diameters increased to 103-110%, also failing to show a significant difference between treatments (Figs. 1B, 2E-H).

Fig. 2. Quality of cut flowers of the standard chrysanthemum cultivar ‘Baekma’ after 4 days of simulated shipping in dry and wet conditions during harvest and shipping stages. A-D, Stem and leaf; E-H, Flower bud; A and E, Dry-Dry; B and F, Dry-Wet; C and G, Wet-Dry; D and H, Wet-Wet treatment; WL, withering leaf.
After cut flowers were treated with these four combinations of dry and wet harvest and shipping conditions, they were placed in holding solutions. Fresh weight showed an overall increase until the eighth day, when it began to decline gradually. However, Wet-Wet and Dry-Wet treatments showed higher fresh weights than other treatments. On the eighth day in the holding solution, fresh weights of cut flowers in Wet-Wet and Dry-Wet treatments increased to 128.3% and 127.1%, respectively, both of which were significantly higher than those in Dry-Dry and Wet-Dry treatments (Fig. 1A). All treatments showed increased flower diameter in the holding solution until the 20th day. Flower diameters of Wet-Wet and Dry-Wet treatments increased to 422.9% and 414.8%, respectively, significantly higher than those of Wet-Dry (390.3%) and Dry-Dry (386.1%) treatments (Fig. 1B). When the vase life of cut flowers in the holding solution was examined, leaves withered earlier in both Dry-Dry and Wet-Dry treatments (Fig. 3), while Wet-Wet and Dry-Wet treatments extended the vase life the longest (20.3 days), 2 days longer than the Dry-Dry treatment (Fig. 1C).
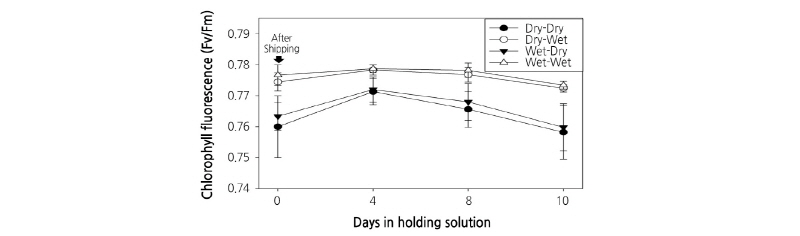
Chlorophyll fluorescence analysis can be used to determine the stress of a plant in a non-destructive manner. Stress levels in a normal environment typically range from 0.80 to 0.84 (Baker and Rosenqvist, 2004). In this study, Dry-Dry and Wet-Dry treatments showed stress levels between 0.760 and 0.763 after simulated transport, while Dry-Wet and Wet-Wet treatments showed a stress level between 0.774 and 0.777, indicating that cut flowers in the Dry-Dry treatment were exposed to high stress due to dry conditions (Fig. 4). In holding solution, the Dry-Dry and Wet-Dry treatments led to a lower level of chlorophyll fluorescence than both Dry-Wet and Wet-Wet treatments, indicating that stress as a result of dry conditions during transport was not reversed by the holding solution.
NaOCl in Wet Shipping Solution
After simulated transport for 4 days and holding in the wet solution with NaOCl, fresh weight of cut flowers was found to increase to 104.0-105.9%, but little difference was noted in fresh weight between treatments (Fig. 5A). The largest flower diameter (117.6%) received the 50 mg‧L-1 NaOCl treatment. However, flower diameters of cut chrysanthemums receiving 0-25 mg‧L-1 NaOCl treatments were as small as 107.1-109.3% (Fig. 5B).
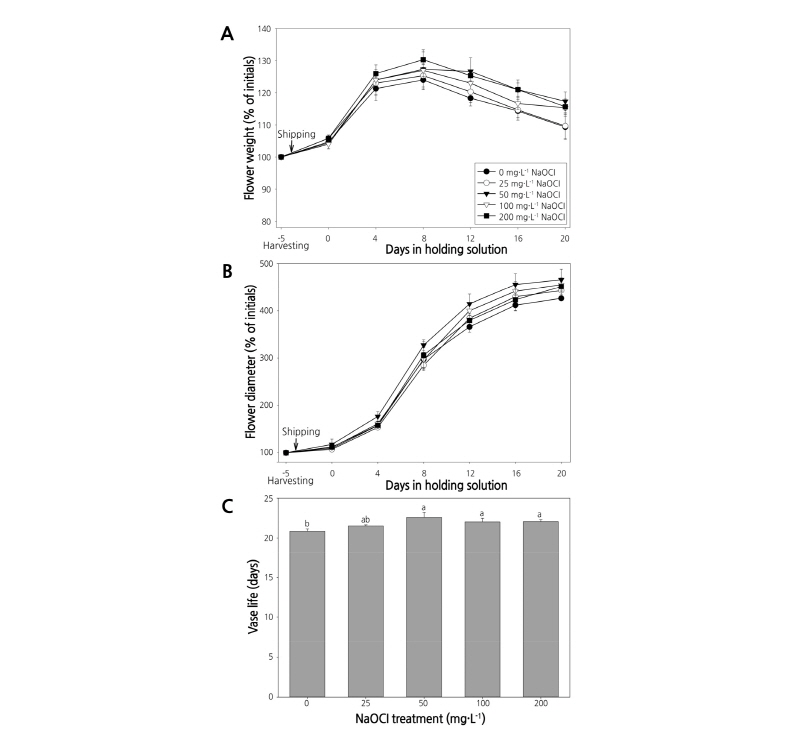
Fig. 5. Changes in fresh weight (A), flower diameter (B), and vase life (C) of cut flowers of the standard chrysanthemum cultivar ‘Baekma’ treated with NaOCl in the wet shipping solution. Vertical bars indicate standard error (n =3). Lowercase letters indicate mean separation at p < 0.05 by Duncan’s multiple range test.
When cut flowers were placed in holding solutions after transport for 4 days, the overall fresh weight increased until the eighth day and then declined. Treatment with 50-200 mg‧L-1 NaOCl increased fresh weight 127.0-130.3%, while the control showed the smallest fresh weight increase (124.0%) (Fig. 5A). On the 20th day, cut flowers in holding solution treatments with 50-200 mg‧L-1 NaOCl showed an increase in fresh weight up to 115.0-117.3%, 6-8% more than the control. Moreover, on the 20th day in holding solution, cut flowers receiving 50 mg‧L-1 NaOCl showed increased flower diameters (465.7%) compared to other treatments, while the control showed the smallest flower diameter increase (426.0%) (Fig. 5B). Flowers treated with 50-200 mg‧L-1 NaOCl had vase lives between 22.0-22.6 days, while the control had the shortest vase life of 20.9 days. However, there was no significant difference in the vase life of cut chrysanthemums among treatments (Fig. 5C).
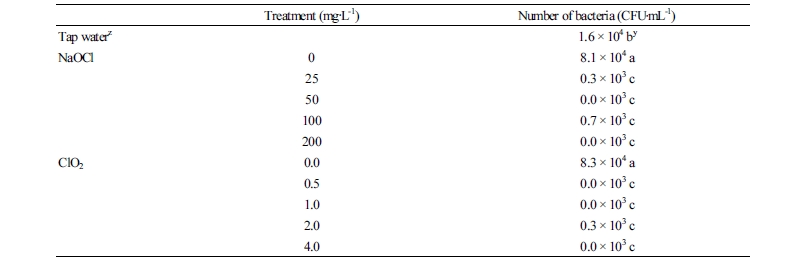
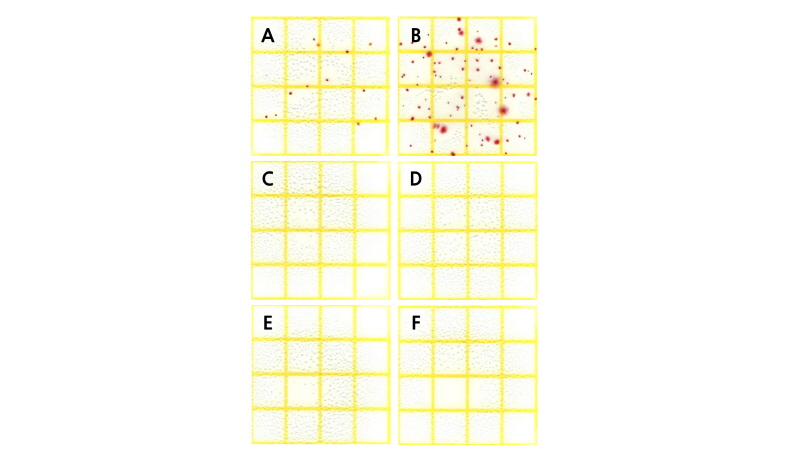
After transporting the standard chrysanthemum ‘Baekma’ at 5°C for 4 days, the number of bacterial colonies in the control increased significantly to 8.1 ×104 CFU․mL-1 compared to 1.6×104 CFU․mL-1 in tap water before transport (Table 1, Fig. 6A, B). In contrast, NaOCl treatments at 25 mg‧L-1 or higher reduced the bacterial colony count to 0.0-0.7×103 CFU․mL-1, indicating that bacterial growth was significantly inhibited by NaOCl (Table 1, Fig. 6C, D). Therefore, treatment with 50 mg‧L-1 NaOCl as a wet shipping solution can effectively inhibit bacterial growth, improving quality via fresh weight and flower diameter, and extending vase life of the standard chrysanthemum cultivar ‘Baekma’ for export to Japan.
ClO2 in Wet Shipping Solution
After simulated transport for 4 days in a wet solution with 0.5 and 1 mg‧L-1 ClO2, the fresh weight of cut flowers was increased to 107.2-107.6%, significantly heavier than the control (Fig. 7A). Flower diameters from all treatments were between 107.1-112.9%, showing no significant difference among treatments (Fig. 7B).
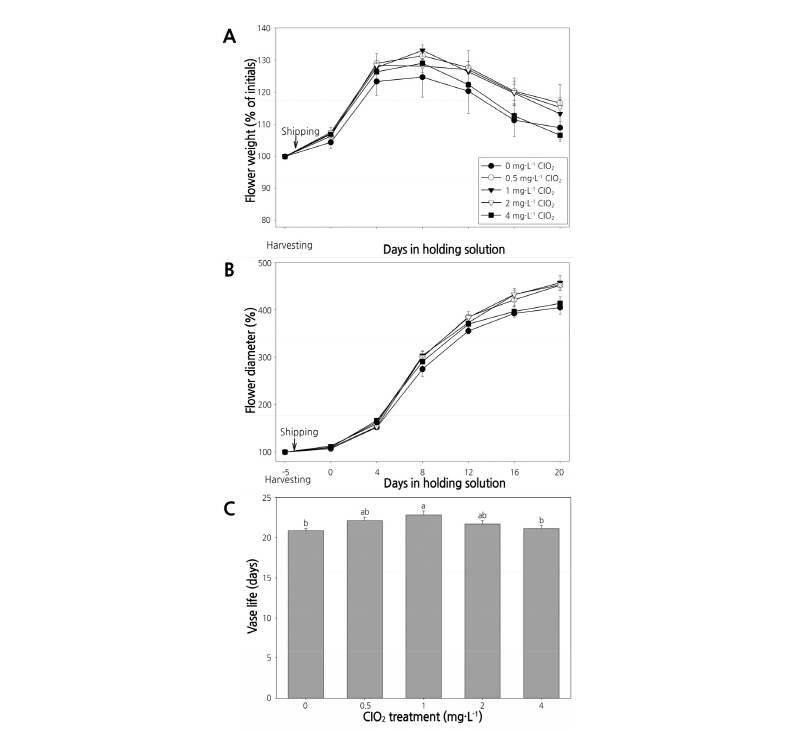
Fig. 7. Changes in fresh weight (A), flower diameter (B), and vase life (C) of cut flowers of the standard chrysanthemum cultivar ‘Baekma’ treated with ClO2 in the wet shipping solution. Vertical bars indicate standard error (n =3). Lowercase letters indicate mean separation at p < 0.05 by Duncan’s multiple range test.
When cut flowers were placed in the holding solution after transport for 4 days, fresh weight was increased until the eighth day and declined thereafter. On the eighth day in the holding solution, 0.5-1 mg‧L-1 ClO2 treatments resulted in the highest fresh weights (131.3-133.0%), while the control showed the lowest fresh weight (124.7%). On the 20th day in the holding solution, 0.5-2 mg‧L-1 ClO2 treatments resulted in flower diameters of 452.0-457.0%, much larger than flower diameters in other treatments, with the control flower diameter being smallest (405.0%) (Fig. 7B). Treatment with 1 mg‧L-1 ClO2 had the longest vase life of 22.9 days while the control had the shortest vase life of 20.8 days. However, there was no significant difference in vase life between treatments (Fig. 7C).
After transporting the standard chrysanthemum ‘Baekma’ for 4 days, bacterial colony counts in treatments of 0.5 mg‧L-1 ClO2 or higher were 0-0.3 ×103 CFU․mL-1, indicating that bacterial growth was significantly inhibited by ClO2 (Table 1, Fig. 6E, F). Therefore, treatment with 0.5-1 mg‧L-1 ClO2 as wet shipping solution is presumably effective in maintaining quality and extending the vase life of cut flowers of standard chrysanthemum ‘Baekma’ for export to Japan.
Discussion
Dry and Wet Conditions in Harvest and Shipping Stages
When cut flowers are exported overseas, they are often stored in dry conditions or at room temperature for extended periods from harvest to shipping overseas. This can degrade the quality of cut flowers and shorten their vase life, leading to deterioration in their market value. In the case of lilies and chrysanthemums exported from South Korea to Japan, cut flowers are usually shipped dried to flower auctions in Japan within 3-5 days after harvest. This can cause loss of freshness manifested as early flowering, non-flowering, and withered leaves (Lee and Lee, 2015; Lee and Kim, 2016). To solve these problems, wet shipping methods have been used for cut flowers such as roses and lilies. Compared to dry shipping, wet shipping offers the advantage that it can maintain freshness of cut flowers, as moisture is continuously supplied during transport. In contrast, when transported in dry conditions, cut flower stems can be penetrated by air, causing vessel blockage. When placed in the holding solution, dried cut flower stems can show poor moisture absorption (Ichimura et al., 2009).
Recently, the use of wet shipping conditions to maintain quality for export overseas have been studied in cut lilies (Lee and Kim, 2016; Lim et al., 2016). Lee and Kim (2016) have reported that when cut lilies ‘Siberia’ and ‘Medusa’ are transported wet by placing them in underground water for 22 h immediately after harvest, stem bending is reduced and their vase life is extended by 2 days compared to dry shipping. In this study, when the standard chrysanthemum cultivar ‘Baekma’ was treated with both dry and wet conditions at 5°C for 24 h immediately after harvest, the dry harvesting condition resulted in decreased fresh weight and flower diameter. However, when treated with wet shipping conditions for 4 days, there was no significant change in the quality of cut flowers in the holding solution. Therefore, for standard chrysanthemum ‘Baekma’, neither dry nor wet treatment immediately after harvest had a significant influence on the quality of cut flowers from the shipping to consumer stage.
Lim et al. (2016) have conducted a simulated export of the cut lily cultivar ‘Medusa’ using dry and wet methods with distilled water for 76 h. Their results showed that the wet shipping method resulted in improved petal and leaf elasticity and flower shape than the dry shipping method and also extended vase life. It has been reported that when cut roses are transported dry, they develop bent necks and their flower buds fail to blossom normally (Ruting, 1991). In the case of the cut rose cultivar ‘Red Sandra’ treated with dry and wet methods, when transport took longer, dry shipping was found to decrease fresh weight significantly compared to the wet treatment. In addition, when cut flowers were placed in holding solution after 48 h in transit, flowers treated with wet shipping solution had increased water uptake, larger flower diameter, and an extended vase life of 2.7 days compared to those shipped dry (Bang, 1999). In the case of cut carnation, dry shipping resulted in fresh weight decreases of 34% and a shorter vase life by 2.7 days compared to wet shipping (Uda et al., 2000). In this study, the standard chrysanthemum cultivar ‘Baekma’ was transported in dry and wet conditions for 4 days of simulated transit after harvest. Wet shipping conditions resulted in heavier fresh weights than dry conditions. When cut flowers were placed in holding solution, those treated with wet shipping conditions maintained higher fresh weights and larger flower diameters with extended vase lives of 2 days compared to the dry treatments.
Chlorophyll fluorescence (Fv/Fm) of leaves of cut flowers was examined to identify causes of quality degradation during dry shipping. When plants are exposed to salt or light environmental stress, the activity of photosystem II decreases, and the Fv/Fm value decreases due to photo-inhibition (Calatayud et al., 2006; Na et al., 2014; Zeng et al., 2017). Choi et al. (2014) have measured the response of chlorophyll fluorescence by pretreatment of cut lily cultivar ‘Siberia’ before export. They found that the non-treatment group showed a chlorophyll fluorescence value of 0.3, while fresh cut flowers pretreated with promalin showed a high chlorophyll fluorescence value of 0.77. Furthermore, when cut roses are exposed to water stress from dry shipping, they develop physiological disorders which in turn reduce their fresh weight and shorten their vase life (Hu et al., 1998; Mor et al., 1989). For standard chrysanthemum ‘Baekma’ flowers, the Dry-Dry condition resulted in significantly lower chlorophyll fluorescence values than the Wet-Wet condition after both simulated transport and storage in the holding solution. These results indicate that when cut chrysanthemum is shipped dry, they suffer severe water stress. When placed in holding solution, chrysanthemum petals and leaves withered quickly and their vase life was shortened. Therefore, when exporting the standard chrysanthemum cultivar ‘Baekma’ to Japan, wet harvest and wet shipping conditions are more effective in reducing water stress of cut flowers, maintaining flower quality through increased fresh weight and flower diameter, and extending vase life.
NaOCl and ClO2 in Wet Shipping Solution
NaOCl and ClO2 are used as pretreatment agents to maintain the freshness of cut flowers and extend vase life after harvest. It has been reported that when the standard chrysanthemum cultivar ‘Jinba’ is immersed in a mixed solution of 200 mg‧L-1 NaOCl and 0.1% sucrose for 6 h (Yoo et al., 2016) or immersed in 5 mL‧L-1 NaOCl solution for 4 h (Lee and Lee, 2015), the quality of cut flowers can be improved significantly. In addition, when the chrysanthemum cultivar ‘Albatron’ and gypsophila cultivar ‘Perfecta’ were placed in a holding solution of 10 µL‧L-1 ClO2, their vase lives were extended (Macnish et al., 2008). When rose cultivar ‘Antique Curl’ was pretreated with 2 mL‧L-1 ClO2 (Vital Oxide) for 4 h (Lee et al., 2016) or when cultivar ‘Beast’ was placed in holding solution of 4 µL‧L-1 ClO2, the vase life of cut flowers was extended (Lee and Kim, 2014). NaOCl and ClO2 inhibit the growth of aerobic bacteria either in the solution or on the cut flowers themselves. Blockage of vessels by propagation of bacteria during long-term overseas transport of cut flowers could explain the shortened vase life of many cut flowers (van Doorn et al., 1986). Therefore, when transporting cut flowers overseas, a wet solution that contains disinfectant should be used to inhibit bacterial growth (Ichimura and Shimizu-Yumoto, 2007). In this study, when the standard chrysanthemum cultivar ‘Baekma’ was exported to Japan for an extended period of 4 days, treatment with at least 25 mg‧L-1 NaOCl and treatment with at least 0.5 mg‧L-1 ClO2 could significantly inhibit the growth of bacteria during transport. When the standard chrysanthemum cultivar ‘Baekma’ was placed in the holding solution, the disinfection properties of these wet shipping solutions was found to improve the quality of cut flowers and extend their vase life.
For cut roses, wet shipping has been used to maintain freshness during long-term transport. Hu et al. (1998) have reported that when the cut rose cultivar ‘Bridal Pink’ is transported at 5°C for 48 h, a mixed solution of 0.3 mM 8-HQS and 0.1 M fructose was able to extend vase life by 3.6 days compared to the control (9.5 days). The rose cultivar ‘Lovely Lydia’ had an extended vase life of 2.7 days and a larger flower diameter compared to the control (distilled water) when transported in a mixed solution of 3% sucrose, 200 mg‧L-1 aluminum sulfate, 200 mg‧L-1 Mg(NO3)2, and 50 mg‧L-1 CaCl2 (Lee, 2011). Ichimura and Shimizu-Yumoto (2007) have also reported that when cut roses are transported with a wet shipping treatment in a mixed solution of 4% sucrose, 0.5 mL‧L-1 CMI/MI, and 50 mg‧L-1 aluminum sulfate, their vase life is extended 4.5 times and their flower diameters are increased compared to those transported with a wet shipping treatment in distilled water. Uda et al. (2000) have reported that when cut carnation is transported with a wet treatment, treatment with wet solution of 0.4 mM STS not only could maintain the freshness of cut flowers, but also could extend their vase life three times. These results indicate that although types of wet solutions vary based on the species or cultivar of flower, disinfectants such as 8-HQS and aluminum sulfate are always added by necessity. In this study, treatment with 50 mg‧L-1 NaOCl and 0.5-1 mg‧L-1 ClO2 as disinfectants in wet shipping solution was effective in maintaining the quality of cut flowers and extending their vase life. This might be due to smooth water uptake into the vessel caused by the bactericidal effect of NaOCl and ClO2 added to the solutions.