Introduction
Material and Methods
Plant Material and Culture Conditions
Measurement of Physiological Characteristics
Statistical Analysis
Results and Discussion
Characteristics of Paprika Leaves
Selection of Regression Models
Introduction
Phalaenopsis bears flowers that are butterfly-like, elegant, attractive, and long-living compared to other flowering plants. Commercially, it is a very promising flower with a high value both domestically and abroad. It is native to the Himalayas, Myanmar, Philippines, Thailand, and Indonesia, with 40 to 50 species distributed from tropical Asia to Australia (Yoon and Jung, 2011). Since the orchid is genetically heterozygous, it is difficult to reproduce seeds with uniform characteristics for commercial cultivation.
Most orchids are therefore propagated asexually through tissue culture (Roh et al., 2012). However, the plantlets from tissue culture are normally weak and a certain portion of plantlets die during the acclimatization process. To address this problem, various studies have been conducted to determine the best environmental conditions for acclimatization, such as temperature and humidity (Jeon et al., 2006; Lee et al., 2018), light intensity (Shin et al., 2009), and medium (Choi et al., 2012).
Stomatal pores are generally highly distributed on the leaf abaxial surface and regulate the plant water status. More than 90% of a plant’s water loss occurs through the stomatal pores while they facilitate gas exchange (Haworth et al., 2015). Stomatal density, pore size, stomatal index, and stomatal aperture are commonly measured to understand stomatal activities (Kim et al., 2016). Among these criteria, stomatal density and pore size are known to be affected by relative humidity (Bakker, 1991).
A plant’s underground environment is also important for its growth. Underground environmental factors include media type, media temperature, moisture content, electric conductivity (EC) concentration, and porosity. Among these factors, the oxygen concentration in the rhizosphere is known to affect the rooting and root growth of the plant. Although the importance of promoting rooting and root growth during seedling acclimatization from tissue culture has been emphasized, studies on the effect of dissolved oxygen levels in the rhizosphere have rarely been conducted (Yang, 1996). In general, because orchid roots need a higher oxygen level than other plants, the soil porosity is crucial (Kim et al., 1997). Furthermore, Phalaenopsis seedlings grown in commercial factories are supplied to farms after a 10 to 15-d acclimatization period and a 3-6 month greenhouse nursing process (data not shown). Therefore, we investigated the effect of relative humidity and air supply on the physiological and stomatal responses in Phalaenopsis during seedling acclimatization.
Material and Methods
Plant Material and Culture Conditions
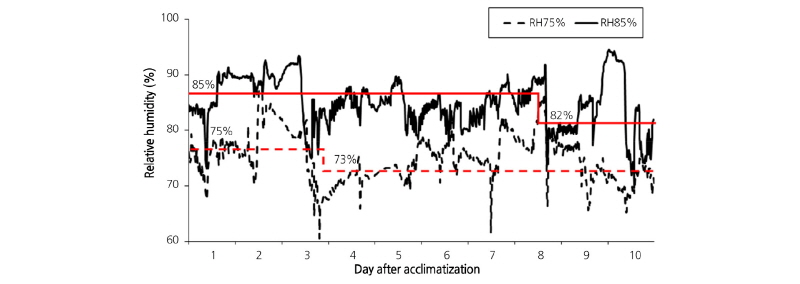
We used peloric Phalaenopsis ‘Breeding No.522’ from the agricultural corporation, UNI Plantech, Eumseong, Korea. For acclimatization, plantlets were extracted from the tissue culture bottle and the roots were carefully cleaned to thoroughly remove any remaining agar. The roots were wrapped with sphagnum moss medium and transferred to plastic pots (ø6 cm × 6 cm in height), which were then placed in an acclimatization container. Acclimatization conditions were set at a temperature of 22 ± 0.6°C, day light intensity of 47.9 ± 24 µmol·m-2·s-1 from a SST5-LED15 white-LED light source (Sigma LED Co., Korea), and 12 h /12 h day/night cycle. For humidity or air supply, the relative humidity (RH) was set to 75% or 85% with or without air supply, resulting in 4 different conditions: RH75% Non-air, RH75% Air, RH85% Air, and RH85% Non-air. For the RH85% Air and Non-air conditions, RH was maintained at 83-85% for the initial 7 days and 82-83% for the last 3 days. For the RH75% Air and Non-air conditions, RH was set to 75-77% for the first 7 days and 75% for the last 3 days (Fig. 1).
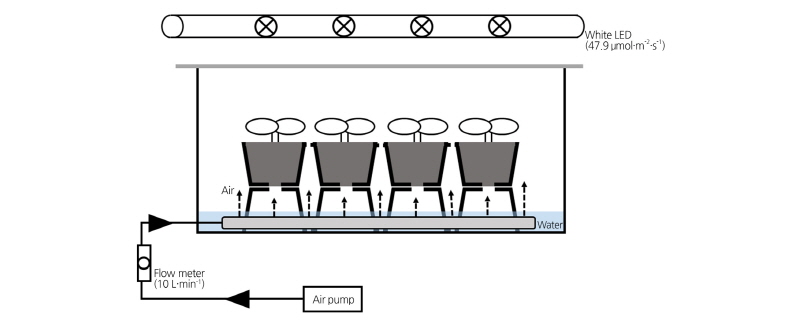
For air supply, an acclimatization container was custom-designed with dimensions of W60 × L40 × H20 cm (Fig. 2). Approximately 5 L of water was added to the bottom of the container and 10 L·min-1 air was continuously supplied by an air bubble bar using an electric air pump (RMA -22-SSV, Dwyer Instruments, Inc., USA). To prevent the supplied water directly contacting the plantlets, a 50-hole plug tray with plantlets was placed on top of an upside-down empty plug tray. Temperature and humidity were recorded using a Watchdog Data Logger 1000, Model 1650 (Spectrum Technologies, Inc., USA).
Measurement of Physiological Characteristics
To examine the physiological changes according to the modulation of RH and air supply during a 10-d acclimatization period, acclimatization rate, the damage rate, leaf pH, stomatal density, and stomatal pore size were examined on the 1st day immediately after the start of acclimatization, and again on the 5th and 10th days.
The relative water content of the medium was determined by repeating 10 plantlets per treatment based on the weight of the pot. The weight (An) of the pot was measured at the same time every day for the 10-d acclimatization period. The relative water content was calculated using the following formula:
Relative water content (%) = 100 - {(A₀ - An)÷ A₀ × 100}
A0 = pot weight at 0-day acclimatization
An = pot weight at n-day acclimatization
The acclimatization rate was determined by calculating the number of dead seedlings per treatment. The damage rate was classified as a percentage of the seedlings with 5% or more leaves with tipburn symptom. Photosynthetic rate, transpiration rate, and stomatal conductivity were measured on the 10th day of acclimatization using a portable photosynthetic analyzer, Li-6400 (Li-COR Inc., USA) at night between 22:00 and 02:00. The following variables were held constant during the gas-exchange analyses in the dark condition: CO2 concentration (400 µmol·mol-1), flow rate (300 µmol·s-1), photosynthetic photon flux density (PPFD, 80 µmol·m-2·s-1), and chamber block temperature (25°C).
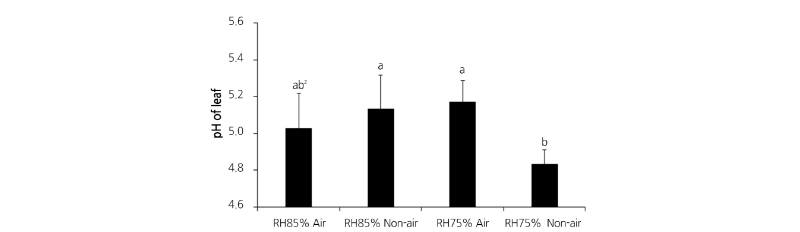
To examine the malic acid content in the leaf, pH was measured by the following method described by Kubota et al. (1997). Briefly, 1.0 cm2 of the leaf was excised after 10 d of acclimatization, placed in a 25 mL falcon tube with 2 mL of distilled water, and microwaved at 400 W for 2 min. Then, the total volume was adjusted to 15 mL with distilled water and the pH of the solution was measured by a S220 SevenCompact pH meter (Mettler Toledo, Switzerland).
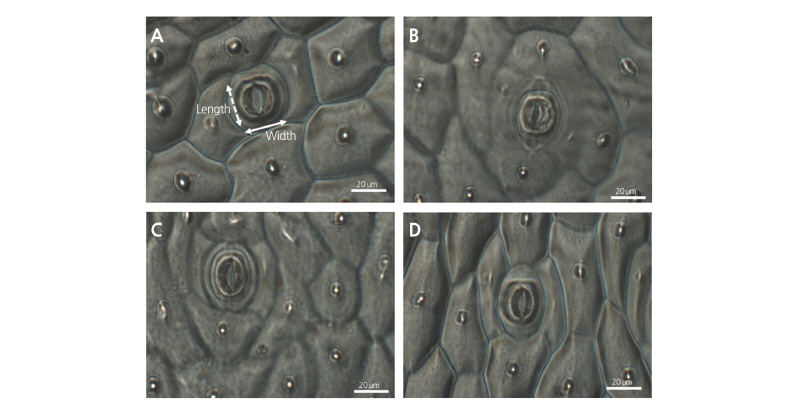
To measure the stomatal density and pore size, clear nail polish Shine top coat (Missha, Korea) was applied to the back of the leaf surface on the 5th and 10th days of acclimatization. A small piece of the dried nail polish was peeled off from the leaf surface using Scotch Tape, dried for 10-15 min, and used for imaging under a Zeiss Axio Imager A2 microscope (Carl Zeiss, Oberkochen, Germany). The number of stomata was counted from a leaf area of 1,500 × 1,080 µm, and the length and width of the guard cells were measured from 4 individual plantlets per treatment after 10 d of acclimatization. The stomatal pore area was calculated using the equation described by Doheny-Adams et al. (2012).
Chlorophyll content was measured using the method described by Mackinney et al. (1941). After the 10-d acclimatization period, 0.3 g of the leaf was weighed and incubated in 10 mL of 80% acetone in a dark room at 4°C for 48 h. The supernatant was measured at 645 nm and 663 nm in three replicates.
Statistical Analysis
Data was obtained from 40 replicates for acclimatization rate, 10 replicates for relative water content, and 4 replicates for physiological and stomatal responses of each treatment. The SAS package (statistical analysis system, version 9.3, SAS Institute Inc., NC, USA) was used to conduct ANOVA (analysis of variance) and Duncan’s multiple range test (DMRT) at 5% for data analysis.
Results and Discussion
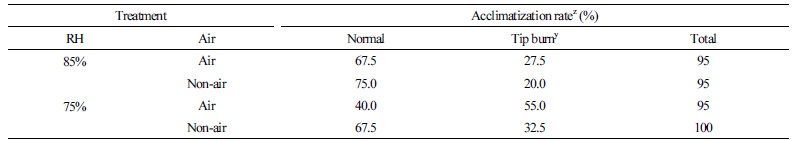
On the 10th day of acclimatization, more than 95% of the seedlings survived, but the leaf damage rate was 20-55% (Table 1). The incidence of leaf tip burning was 20-27.5% in the RH 85% with/without air supply and 32.5-55% in RH 75% with/without air supply plantlets. The highest tip burning rate was 55%, observed in the RH75% Air plantlets. During acclimatization, air supply appeared to increase the leaf damage rate, which could be worse with low humidity, as evident with the highest tip burning shown for the RH75% Air treatment plantlet. While Choi et al. (2005) reported that ventilating the culture container had positive effects on the leaf area, leaf number, and root growth of bellflower plantlets, our result showed a different effect for Phalaenopsis plantlets. However, it is possible that the effect of the direct air supply to the water could differ to that from ventilation. In addition, the bellflower and Phalaenopsis are very different plant species, and could be differently affected by aeration.
The higher leaf damage on plantlets in RH 75% with/without air supply compared to RH 85% could be due to the sudden changes of relative humidity and air flow rate from nearly 100% RH, the low air flow in tissue culture bottles to the RH75% plantlets, and the high air flow in the acclimatization container (Yang et al., 1996). In addition, it is known that stomata of in vitro photoheterotrophic plants grown on culture media do not respond well to sudden environmental changes. Therefore, plantlets from tissue culture can be detrimentally affected when they are placed in a low humidity environment, as seen in tip burning (Lee and Bang, 2004).
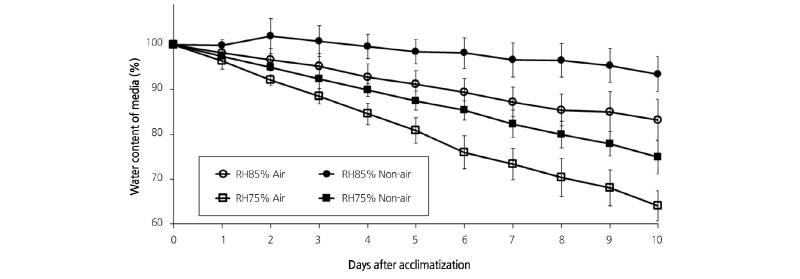
During the 10-d acclimatization period, the relative water content of the medium decreased gradually (Fig. 3). The highest decrease was observed in the RH75% Air plantlet with 35.98%, whereas the lowest decrease was found in the RH85% Non-air plantlet at 6.58%. The decreases of the relative water content in the media of the RH85% Air and RH75% Air plantlets were 2.56 times and 1.43 times higher than those in the RH85% Air and RH75% plantlets, respectively. The loss of water in the media was 11.7% in the RH85% and 30.5% in the RH75% plantlets, indicating that lower humidity conditions can cause significant water loss in the culture media.
From these results, it can be assumed that the reduction rate of the relative water content in the media tends to be accelerated by low humidity when additional air is supplied. While plant growth relies on appropriate air circulation in the acclimatization container (Heiskanen, 1993), it can be damaged by high CO2 concentrations (Hwang and Jeong, 2007). The air supply can increase the amount of air space in the media, which is beneficial for soil gas exchange. Under poor soil air circulation, Phalaenopsis roots do not branch well and grow as a fleshy form. Therefore, increasing the soil air by aeration can have a positive effect on root growth, and ultimately benefit the whole plant. Leaf tipburn is not only caused by Calcium (Ca) deficiency, but can also be caused by the chemicophysical properties of soil (soilless media) and by inadequate water conditions such as water deficit, strong light, and high humidity. In addition, the severity of leaf tipburn symptoms can differ among cultivars (Choi, 1999). Generally, the degree of leaf tipburn in Phalaenopsis varies depending on cultivars. ‘Breeding No. 522’ showed the occurrence of late tip burn, suggesting that this cultivar may be somewhat tolerant to tipburn. In addition, we found that long-term air supply under low humidity conditions can damage leaves, as shown in the RH75% Air plantlets.
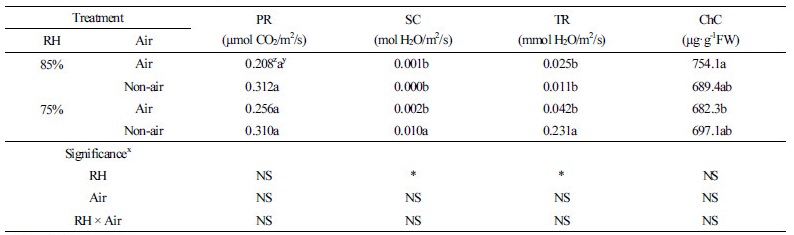
Stomatal conductance and transpiration rate were higher in the RH75% Non-air with 0.010 mol H2O/m2 per second and 0.231 mol H2O/m2 per second than in the rest of the treatments, suggesting that the plants grown at high humidity have poor stomatal activity (Kwon et al., 2004). Jeon et al. (2006) reported that the growth and survival rate of in-vitro Doritaenopsis was higher in a RH90% treatment. However, the leaf relative water content and physiological responses were different after the second day of acclimatization, but it recovered after 10 d and showed no symptoms after 30 d (Jeon et al., 2006). The photosynthetic rate, stomatal conductance, and transpiration rate were higher in the Non-air treatment plantlets than in the plantlets treated with air supply, but there was no statistical significance (Table 2). The highest chlorophyll content was observed in the RH85% Air plantlets with 754.1 µg·g-1 FW, whereas the lowest content was found in the RH75% Air plantlets with 682.3 µg·g-1 FW. No significant difference was observed in the chlorophyll content in response to changes in relative humidity or air supply. This result is inconsistent with a previous report that showed chlorophyll content was higher under low humidity conditions (RH65%) than plantlets growing under high humidity (RH80% or RH95%) (Cha-um et al., 2010).
Phalaenopsis, a typical CAM plant, opens stomatal pores at night to absorb CO2 and fixes it to malic acid, which is then stored in the vacuole. During the day, the plant breaks down the malic acid to CO2 for photosynthesis. To compare photosynthesis and leaf pH, leaf pH was measured at night on the 10th day of the acclimatization period. Leaf pH was 5.1 in both the RH75% Air and RH85% Non-air plantlets, pH 5.0 in the RH85% Air plantlet, and pH 4.9 in the RH75% Non-air plantlet. Leaf pH changed in all 4 treatments, indicating that plants were accumulating malic acid properly and acclimating to their environments. The highest stomatal conductance was observed in the RH75% Non-air environment (Fig. 4), indicating that low humidity increases gas exchange and has a positive effect on photosynthesis.
On the 5th day of acclimatization, stomatal density increased 2.1-2.8 times from 10.3 (data not shown) on the first day. On the 10th day, stomatal densities in the RH85% Air and RH75% Air plantlets were 28.0 and 28.3 (Table 3), respectively, significantly higher than those in the RH85% and RH75% plantlets without air supply, suggesting that stomatal density positively correlates with humidity increase and air supply. This result is consistent with previous findings that suggest stomatal density increases significantly under low vapor pressure deficit/difference (VPD) than high VPD (Bakker, 1991).
Table 3. Effect of relative humidity and air supply for Phalaenopsis stomatal number at 5 and 10 days after acclimatization
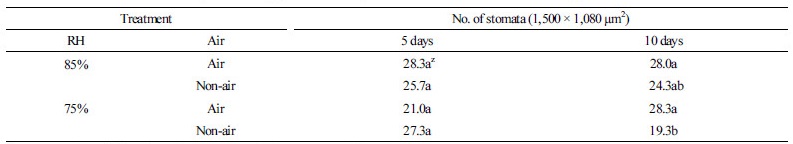 | |
zMean separation within columns by Duncan’s multiple range test, 5% level. | |
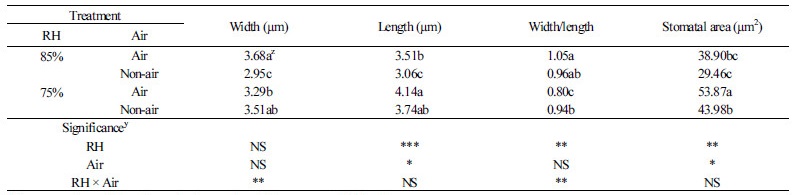
Table 4 shows the stomatal pore dimensions of width, length, and area on the 10th day of acclimatization. The length of the stomatal pore in the plantlets with RH75% treatment was longer than those with the RH85% treatments (Fig. 5). The stomatal pore size was the largest in the RH75% air treatment plantlets at 53.87 µm2, whereas the smallest pore size was 29.46 µm2 in the RH85% Non-air treatment plantlets. Overall, the stomatal pore size was larger in the treatments with air supply than those without air supply (Table 4). Interestingly, ‘Breeding No. 522’ showed both an increased stomatal density and stomatal pore size, which is inconsistent with a previous observation that pore size is negatively proportional to stomatal density (Bakker, 1991). This inconsistent result is likely due to the application of air supply in our test, which may have positive effects on the stomatal opening.
In summary, the stomatal density increased more than twice during acclimatization, and the air supply during acclimatization showed positive effects on both stomatal density and pore size of the seedlings. However, it showed a tendency to increase water loss from soilless medium and leaf tipburn damage during acclimatization. In the RH75% Non-air plantlet, stomatal conductance, transpiration rate, and leaf pH were higher than in the other conditions, but no significant difference was observed in the photosynthetic rate among the treatments. While the air supply plays a positive role for acclimatization under the high humidity condition, it can have a negative effect for acclimatization under low humidity, ultimately increasing leaf damage. In this study, slightly higher leaf damage was observed when air was supplied to the acclimatization container; this is likely because this experiment was performed under 10% lower relative humidity conditions than the general relative humidity condition with the RH90% ± 5% plantlet.
Since Phalaenopsis requires a relatively longer time to adapt to being transferred from tissue culture bottles, it is desirable to maintain the high humidity of the tissue culture bottle environment (80-90%), in the early stage of acclimatization. High humidity preserves stomata function during early acclimatization and reduces rapid water loss, minimizing tipburn and abiotic stress. The air supply during the acclimatization period can have positive effects on air circulation and medium aeration, as well as on stomatal opening, transpiration, and photosynthesis. On the other hand, low humidity can damage seedlings.