Introduction
Materials and Methods
Seed and Chemical Materials
Experimental Design
Hydration and Incubation
Evaluation of Germination
Statistical Analysis
Results
Effect of Priming on Seedling Vigor
Days to Percentage Germination vs Germination Rate
Effect of Priming on Shoot Height and Fresh Weight
Discussion
Introduction
Seed germination is a crucial factor for commercial agri-culture and substantial work has examined the conditions required for optimal germination. In the early 1950s, optimal soil temperatures for diploid seed germination were established in the range of 21-35°C (Harrington and Minges, 1954). Since then, new hybrid cultivars have replaced open-pollinated cultivars and new cultivars were found to respond to different temperature ranges. For proper seed germination, growth, and development, diploid watermelon requires warmer than 26°C (Whitaker and Davis, 1962), but triploid seeds originating from tetra-ploid ovaries require 24-38°C for 24-72 h. Triploid seedless watermelon was first developed in Japan in the early 1950s (Kihara, 1951). Triploid seedless watermelon has been becoming popular over the last two decades due to its crisp and sweet taste without the hard seeds of standard watermelon (Egel, 1999; Marr and Gast, 1991). Triploids result from breeding between a tetraploid watermelon, as the female parent, and a diploid watermelon, as the male parent. Triploid watermelon does not produce sufficient pollen for fertilization; thus the production of triploid seedless fruits requires a diploid plant for pollen (Maynard and Elmstrom, 1992; Zhang, 2004).
Watermelon growers face various problems in obtaining good seed germination, as triploid seeds have low germination due to the thicker seed coat, underdeveloped embryos, and larger air space cavities compared with diploids (Duval and NeSmith, 2000; Grange et al., 2000, 2003). Triploid seed germination is generally low, at 60-80% compared to up to 95% in diploid (Liu et al., 2010; Nerson et al., 1985). Heavy seeds germinate better because of higher seed coat tissue compared to light-weight seeds in triploids (Yang and Sung, 1994). Methods such as nicking, scarification and lateral splitting of seed coat enhance the germination rate in triploid, but also can injure the embryo (Duval and Nesmith, 1998; Grange et al., 2000). Hence, there is a need to explore more efficient and reliable methods to enhance seed germination rate and seedling establishment, as normal germination methods no longer produce good results, especially in tetraploids and triploids.
Seed priming has been reported to enhance seed germi-nation performance of various field crops (Eskandari, 2013; Shehzad et al., 2012). Various seed priming methods can improve germination; these methods include hydropriming (Huang et al., 2002; Moghanibashi et al., 2012), which improves germination by partially hydrating seeds, although radicle emergence does not occur. Other priming methods include halopriming (Siadat et al., 2011), osmopriming (Chen et al., 2010; Esmaelli and Heidarzade, 2012; Farooq et al., 2005, 2007; Rezaei and Ramezani, 2012; Rouhi et al., 2011), thermopriming (Yari et al., 2012), solid matrix priming (Mereddy et al., 2000), and biopriming (Moenzadeh et al., 2010). The growth regulator gibberellic acid (GA) can improve seed germination rates and reduce germination time in endive and chicory (Ghodrat and Rousta, 2012; Tzortzakis, 2009). The application of hydrogen peroxide (H2O2) enhanced germination efficiency with no effect on seed coat (Duval and NeSmith, 2000). Hydrogen peroxide has been suggested to mediate imbibition and hydrolytic activities required for germination and alleviate salt and temperature stresses (Çavusoglu andKabar, 2010). Fusicoccin (FC), from Fusicoccum amygdali is a novel 5-8-5 membrane tricyclic diterpene glucoside. It is a potent promoter of germination and dormancy-breaking agent. The mode of action of FC on plant 14-3-3 proteins and H+-ATPase are currently attracting substantial attention (Tajima et al., 2004). FC induced cell wall loosening and thus loss of turgor and cell enlargement by stimulating H+ extrusion and K+ uptake similarly to, but much more strongly than auxin (Marre, 1979).
The information on seed germination, emergence and uniform growth in polyploid watermelon remains inadequate, despite the different priming and seed treatment methods studied so far. Hence, this study was carried out to investi-gate the priming effects of different concentrations of chemicals, including H2O2, FC, and GA, on germination and seedling uniformity of triploid and diploid watermelon.
Materials and Methods
Seed and Chemical Materials
The experimental materials included seeds of three triploid cultivars, Seedless Plus (Nong Woo Bio Co., Suwon, Korea), Sinus (L & S Seed Company, Cheongju, Korea), and Sizero (Syngenta, Bangkok, Thailand). Sambokggul, diploid cultivar, was used as a control. These were subjected to hydropriming testing at the Watermelon Experiment Station, Daesan, Gochang during the year 2013. Hydrogen peroxide (H2O2), fusicoccin (FC), and giberrelic acid (GA) were tested as germination-enhancing agents. Different concentrations of H2O2 (2, 4%), FC (1, 5, 10 μM) and GA (1, 5, and 10 μM), water (H2O), and a control condition were used to analyze the effect on germination rate of seeds.
Experimental Design
The experiment was conducted as a three-factor compact family block design (CRBD) randomized with three repli-ca-tions and ten types of treatments: one water, two con-cent-rations of H2O2, three of FC, three of GA, and one control. However, the 4% H2O2 treatment was omitted for cultivar Seedless Plus due to insufficient seeds. Each replication contained twenty-five seeds of each cultivar.
Hydration and Incubation
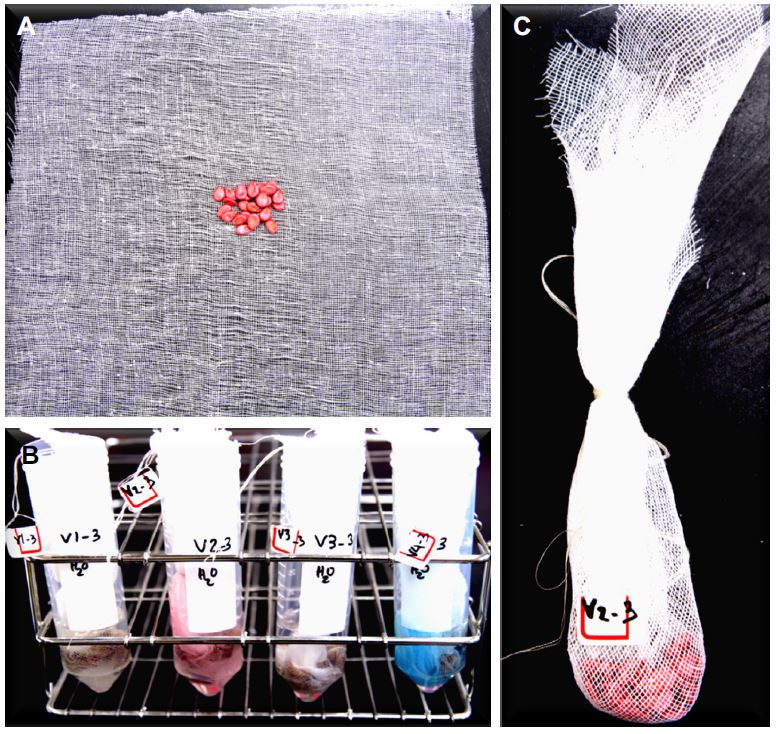
Twenty-five seeds per replication for each treatment were weighed. The seeds were gathered together on 22 × 22 cm squares of muslin gauze cloth, then wrapped and tied using thread attached to a unique number for each treatment (Seedless Plus V1, Sinus V2, Sizero V3, and Sambokggul V4). The gauze-wrapped seeds were soaked in centrifuge tubes containing 15 ml of each respective concentration of solution for 12 hours and kept at room temperature (Fig. 1). Imbibed seeds were rinsed with distilled water prior to incubation at 31 ± 2°C with 35% relative humidity until seeds were dried to their original seed weight (around 48 hours).
Evaluation of Germination
Primed seeds were sown in plastic trays containing commercial soil mixture and raised in a plastic house. The temperature and humidity were recorded every hour using a data log. The average temperature and humidity at day/night were 32 ± 8°C and 62 ± 21%/24 ± 2°C and 93 ± 8%, respectively. Seeds were considered germinated upon the protrusion of the cotyledon above the soil. After the third day, the number of germinated seeds was recorded daily for two weeks. The time to 50% germination (T50) was calculated according to the modified formula reported by Farooq et al. (2005) in Equation 1, and the final germi-nation percentage (FGP) was estimated using Equation 2. The germination index was calculated based on the formula described by the Association of Official Seed Analysts (AOSA, 1983) and used by Farooq et al. (2005) in Equation 3. Shoot length (cm) and seed weight (g) were recorded on the last day of observation and 20% of maximal and minimal height seedlings were dropped out.
Equation 1
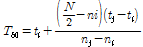
N = the final number of germinated seeds; nj , ni: cumulative number of seeds germinated by adjacent counts at times tj and ti when ni < N/2 < nj.
Equation 2
Equation 3
Statistical Analysis
The data were analyzed using the statistiXL program for general statistical analysis. For each of the measured variables, comparison of means or medians was conducted through a one-way analysis of variance (ANOVA) to determine whether differences are statistically significant (p ≤ 0.05). Means of treatments were separated by Fisher’s LSD test at p ≤ 0.05.
Results
Effect of Priming on Seedling Vigor
The different priming and non-priming treatments showed significant effects on the seedling vigor of triploids (Table 1). FGP was significantly different and higher in primed seeds compared to control in the triploids but not in the diploid. Seedless Plus had a higher FGP with 5 μM GA (68%), Sinus with hydroprimed (81.3%), Sizero with both hydroprimed (86.7%) and GA 5 μM (86.7%), and Sambokggul with hydro-primed (98.7%) all showed higher FGP compared with the control treatment. However, time to 50% germination did not show a statistically significant difference among triploid and diploid cultivars. A significant difference in GI was found only in Sizero with hydroprimed treatment (4.6) compared with the control treatment (3.5).
Days to Percentage Germination vs Germination Rate
Germination rate (Table 2) was highest in Sizero (control, 73%; hydroprimed, 87%) followed by Sinus (control, 59%; hydroprimed, 81%) and Seedless Plus (control, 53%, FC 5 μM, 68%). In addition, seeds of Seedless plus treated with 1 μM FC started germinating within three days (1.3%), but seeds treated with 5 μM GA started germinating by the fourth day (21.3%). The maximum germination percentage 22.7% was attained within 3 days and reached to 81% FGP within 8 days in hydroprimed seed of Sinus; in other treatments FGP ranged from 44% to 71%. Seeds of hydro-primed Sizero began to germinate within 3 days, then the germination rate continuously increased, reaching 87% (FGP) on the 7th day of the first week. However, seeds primed with other treatments started germinating within 4 days with maximal germination rate at the 7th day, from 2% to 77% FGP.
Effect of Priming on Shoot Height and Fresh Weight
The shoot height and fresh weight of triploids and diploid cultivars also showed the effect of different priming treat-ments (Table 3). Seed priming consistently had no positive effect on shoot height among triploid cultivars. Control treatment had higher shoot height with 8.1, 9.0, 9.0 cm in Seedless Plus, Sinus, and Sizero, respectively, compared to the primed treatments. Although FC likely had positive effect on the growth of shoot height in Sambokggul with
a height of 10.1 cm (1 μM FC), 10.1 cm (5 μM FC), and 10.3 cm (10 μM FC) over the control treatment (9.4 cm), a significant effect was not found. Fresh weight of shoots showed a marginal difference in primed treatment with 2% H2O2 over the control, FC, and GA treatments among triploids and diploid cultivars with the exception Seedless Plus. Fresh weight was 0.38 g in seedlings from seeds treated with 2% H2O2, compared to 0.51 g in control, 0.54 g in 1 μM FC, 0.55 g in 1 μM GA, 0.5 g in 5 μM GA, and 0.57 g in 10 μM GA in Seedless Plus. Inconsistent differences were observed in treatments with FC and GA over the control treatment among all triploids and diploid cultivars.
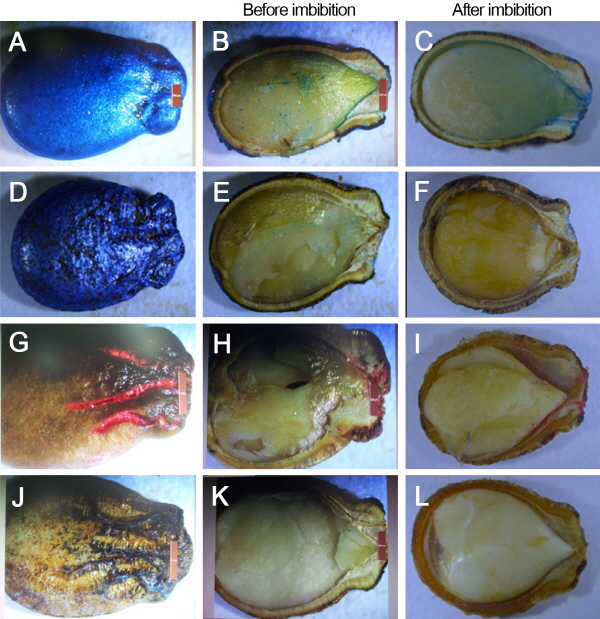
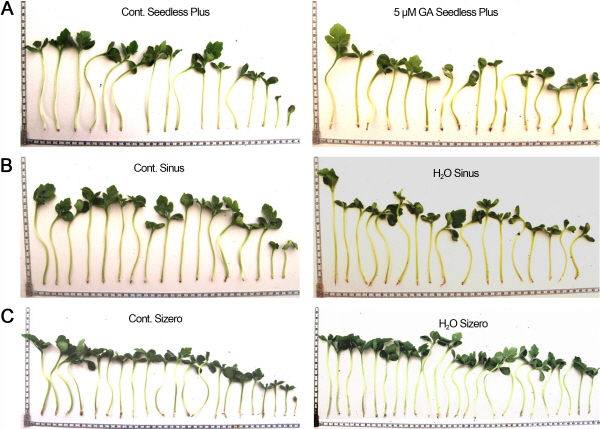
All these differences in seedling vigor were suggested to be due to differences in shape and sizes of seeds, seed coat thickness, and air space cavity. Larger air spaces in seeds were observed in Sinus, followed by Seedless Plus, Sizero, and Sambokggul (Fig. 2). Seedlings of control vs primed seeds with 5 μM GA, which was the most effective in Seedless Plus, hydroprimed Sinus, and hydroprimed Sizero are shown in the Fig. 3.
Discussion
The present experimental findings showed that seed priming has a positive effect on the germination rate in all triploid cultivars and germination index (GI) in Sizero, whereas time to 50% germination (T50) showed no effect. In our study, seeds of triploids and diploids had some differences in seed shape and sizes, thickness of seed coat, and air space cavity and these factors led to variation in response to priming treatments, resulting in differences in germination ability. FGP increased in all triploid cultivars and GI increased in Sizero, but T50 showed no differences. The results revealed that Seedless plus showed better performance with 5 μM GA (FGP, 68%; GI, 3.2), Sinus with hydropriming (FGP, 81.3%; GI, 5.0), and Sizero with hydro-priming (FGP, 86.7%; GI, 4.6) treatments. Sambokggul showed the highest FGP 98.7% with hydropriming treatment whereas GI and T50 showed no effect of any primed treat-ment over the control. Germination rate was 68% after 13 days in Seedless plus with 5 μM GA, 81% within 8 days in Sinus with hydropriming, and 87% with 8 days in Sizero with hydropriming. Seed priming consistently had no effect on shoot height among triploid cultivars and fresh weight of shoots had a marginal difference of regression in seeds treated with 2% of H2O2 over the control among triploids and diploid cultivars, with the exception of Seedless Plus. In particular, it was noted that as the concentration of different treatments increased, there was corresponding negative impact on the seedling vigor of the triploids and diploid cultivars. Hence, concentration played key factor in controlling the germination behavior in triploids. It has been reported that priming techniques were applied successfully to improve germination rate, uniformity of germination, final germination, or shorten T50 (Eskandari and Kazemi, 2011; Moghamibashi et al., 2012); however, germination efficiency was found to vary with species and priming method (Selvarani and Umarani, 2011). This result was in agreement with the results of Huang et al. (2002), which showed that hydropriming was effective in triploid water-melon cultivar Guangxi. FC was found to accelerate metabolic processes (Lutsenko et al., 2005), transmembrane electric potential, and germination (Ajmal Khan et al., 2009; Ballarin- Denti and Cucucci, 1979; Gul and Weber, 1998; Simonovic et al., 2000).
The amount of seed material and its composition within the seed is presumably utilized to support seed coat splitting and seedling emergence. In triploids particularly, due to the weak embryo, the energy required to overcome the resistance of the thick seed coat and the overlying growth media would be limited to some extent, resulting in low emergence rate. Hence prior seed priming treatment, i.e. before the seed began growing in the nursery, helped to provide this energy to enhance germination performance. The present findings helped to find the optimal priming conditions for achieving improved germination of triploid watermelon. GA and hydropriming helped to break down seed dormancy and thus led to the enhancement of FGP and GI. It was concluded that hydropriming and 5 μM GA seed treatments could efficiently enhance germination in triploids. These seed priming treatments could be used on a large scale for industrial applications. Hydropriming is a simple, effective, and costless method for improving seed germination and seedling vigor of Sinus and Sizero.