Introduction
Materials and Methods
Plant materials and cultivation management
Light quality experiment
RH experiment
Temperature experiment
Plant growth assessment
Statistical analyses
Results
Growth under different light conditions
Growth under different RH conditions
Growth under different temperature conditions
Discussion
Blue light induces adventitious rooting
High RH promotes adventitious rooting
Specific temperatures enhance adventitious rooting
Conclusions
Introduction
Cannabis is an annual dioecious herbaceous plant (Small 2015). Originating in Central Asia, it has been cultivated in China for over 6,000 years (Li 1974; Russo et al. 2008). It is used as a multipurpose crop in the fiber, fuel, and food industries (Piluzza et al. 2013). Cannabinoids produced in the glandular trichomes of cannabis are primarily used recreationally and for medicinal purposes (Xie et al. 2023).
Cannabis is mainly propagated via seed and stem cuttings (Campbell et al. 2021). Seed propagation creates genetic diversity, enhancing the adaptability of the plant to various environments (Lei 2010). However, this strategy can also lead to inconsistent production outcomes (Potter 2014). In contrast, stem cuttings, which root easily (Campbell et al. 2019), produce genetically identical offspring, thereby ensuring uniformity (Campbell et al. 2021). Propagating stem cuttings from female parent plants only yields female plants, with higher cannabinoid production than that observed under seed propagation (Coffman and Gentner 1979). Therefore, this method is widely used on cannabis farms in the USA (Chandra et al. 2020).
Adventitious root (AR) formation is vital for the successful propagation and growth of hemp stem cuttings. AR formation involves the emergence of new roots from non-root tissues after the embryonic phase (Druege et al. 2019). These roots are crucial for restoring the plants’ ability to absorb water and nutrients after cutting, thereby improving their survival (Druege et al. 2019). Therefore, effective AR formation is essential for promoting plant survival and root growth, which are fundamental for the large-scale production of high-quality clones (Tian et al. 2024).
External conditions, such as light, temperature, and relative humidity (RH), are pivotal for successful AR formation in cannabis stem cuttings. RH affects the internal water balance of plants via transpiration (Chia and Lim 2022), and water stress impedes root development and adversely affects the cuttings (Puri and Thompson 2003). Moreover, high relative RH has been reported to enhance root formation by facilitating auxin transport through the phloem (Chhun et al. 2007). For instance, in Cirsium arvense, cuttings exposed to high RH levels exhibited a twofold increase in both the shoot length and dry weight compared to those grown under a 50% RH condition. Conversely, root growth was significantly suppressed under lower RH conditions (Hunter et al. 1985). These findings highlight the critical role of RH in root development, emphasizing the necessity of determining the optimal RH conditions for rootless cannabis cuttings to promote successful rooting.
Temperature influences plant growth outcomes overall, including cellular growth, respiration, and photosynthesis (Went 1953). In the early stages of plant development, root growth is particularly sensitive to the temperature, which affects auxin biosynthesis and helps maintain balanced growth between shoots and roots (Gaillochet et al. 2020). However, extreme temperatures—either too high or too low—can negatively impact root development. For instance, high temperatures have been shown to reduce cell division significantly in root meristems, thereby inhibiting root growth (González-García et al. 2023). Similarly, cold stress can reduce both the root length and biomass (Cutforth et al. 1986). Therefore, the temperature influences root development in plants, making it essential to determine the optimal rooting temperature for hemp. Given that the optimal temperature can vary depending on the hemp cultivar (Geneve et al. 2022), it is necessary to identify the ideal temperature conditions for each cultivar.
Considering the above, precise environmental control is necessary to optimize root induction in early stage cuttings. Additionally, photomorphogenic factors, including the light quality, intensity, and photoperiod, significantly influence growth and the transition to reproductive stages (Eaves et al. 2020; Moher et al. 2021). Light quality is crucial for root initiation and affects AR development during the early cutting stage (Hedger 1961). For instance, a study of hemp reported that red light significantly promotes root initiation (Pepe et al. 2021). However, another study found no significant effect of light quality on root formation in hemp (Moher et al. 2023). These conflicting findings are likely due to physiological variations among different hemp cultivars. Therefore, it is necessary to conduct light quality experiments on new cultivars to gain a better understanding of their specific responses and to optimize the rooting conditions accordingly.
As cannabis regulations continue to evolve globally, many studies are exploring various cannabis cultivars. Cannabis research in Korea is also progressing owing to the recent easing of regulation. In this study, cultivars established by the Rural Development Administration of Korea were used to conduct a comparative study of AR formation in stem cuttings under different light quality, temperature, and RH conditions. The experiment examined how these factors influenced underground growth in cuttings under controlled conditions. Light quality was categorized into that provided by red, green, and blue light, with an additional treatment using white light, which includes the full spectrum, resulting in a total of four different light conditions. For temperature, the optimal temperature for hemp is reportedly 24°C (Jin et al. 2019). Based on this, this study established four temperature conditions (15°C, 20°C, 25°C, and 30°C) to observe root development trends across different thermal environments. RH was also analyzed by establishing the four treatment levels of 30%, 50%, 70%, and 90%, allowing for a comprehensive assessment of RH effects on underground biomass accumulation. Cannabis propagation studies have focused on single-variable experiments, overlooking factor interactions. This study aimed to identify the optimal environmental conditions for adventitious root formation in hemp developed by the Korean Rural Development Administration. By determining the ideal light quality, temperature, and humidity conditions, the findings here can provide a foundation for future integrated research.
Materials and Methods
Plant materials and cultivation management
Hemp seedlings were grown under 200 ± 10 µmol·m-2·s-1 light intensity, 70–75% RH, and an 18/6 h light/dark photoperiod. A photoperiod of 15 hours or less can lead to the formation of incomplete inflorescences, whereas a photoperiod of 18 hours or more completely inhibits flowering (Park et al. 2023). Therefore, in this study, a photoperiod of at least 18 hours was applied to prevent flowering. Hemp (Cannabis sativa L.) cultivars ‘IT342810’ and ‘IT342811’ provided by the Rural Development Administration (Umsung, Korea) were used as the mother plants. These mother plants were irrigated using a drip system with the Hoagland nutrient solution, adjusted to an electrical conductivity (EC) level of 2.0 dS·m-1 and pH of 6.5. Once the mother plants reached a height of 1.7 ± 0.2 m, all leaves except for three to four true leaves were removed, and green cuttings were planted in rockwool cubes (Grodan, Roermond, Netherlands). A total of 144 rockwool cubes (25 × 25 × 75 mm) were used, with six plants per treatment in the cultivation trays. Green cuttings were grown in a chamber (DS-310S; Daewon Science Inc., Bucheon, Republic of Korea), which is a system that allows control of the temperature, humidity, light intensity, and photoperiod. The light intensity was maintained at 80 ± 5 µmol·m-2·s-1 with a photoperiod of 20/4 h (light/dark). The temperature, RH, and light quality varied according to the experimental design over three weeks.
Light quality experiment
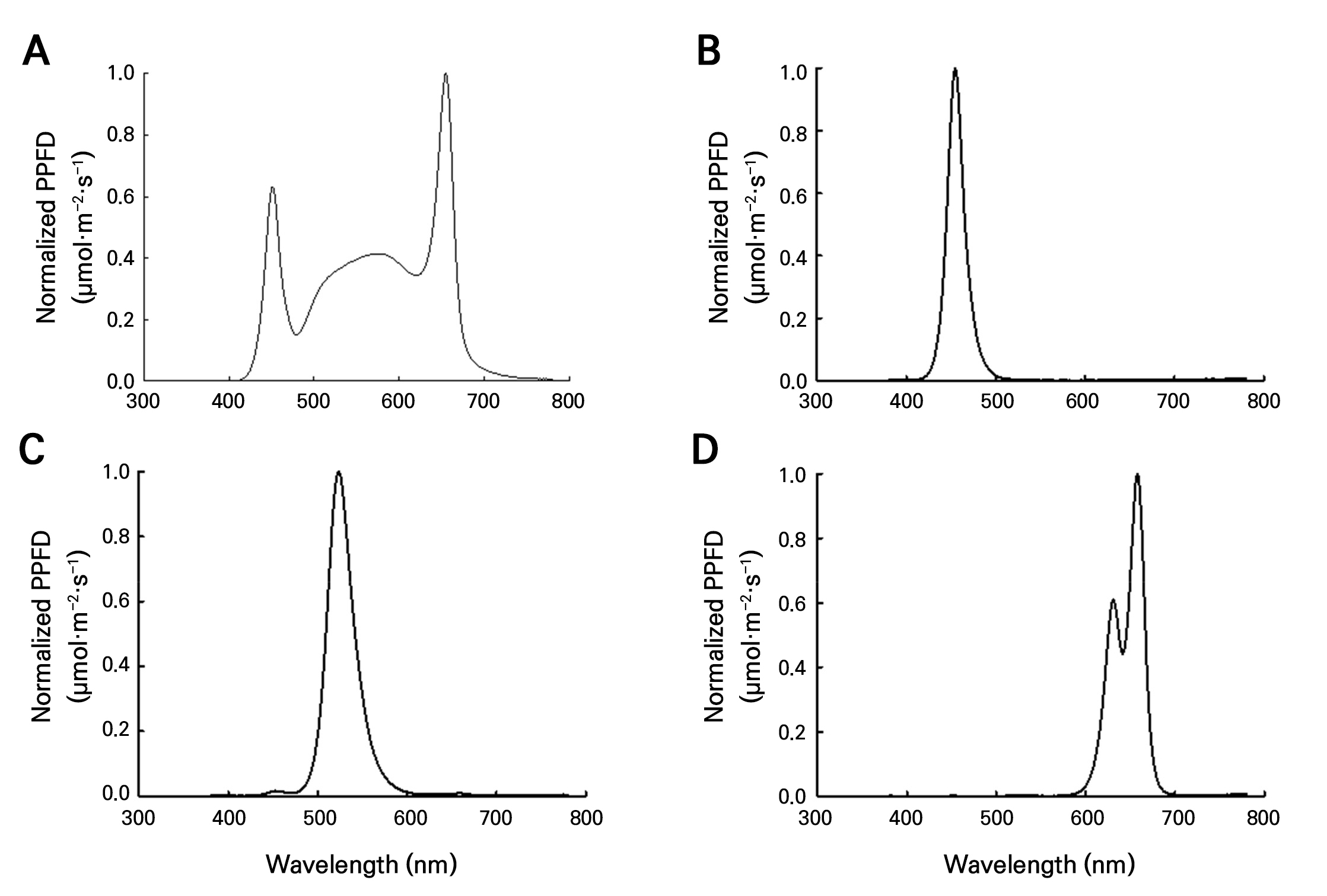
C. sativa L. ‘IT342810’ and ‘IT342811’ plants were grown under the blue, green, red, and white light conditions (Fig. 1). Light intensity at the canopy was maintained at 80 ± 5 µmol·m-2·s-1 with a 20/4 h light/dark photoperiod. The cultivation bed has a height of 2000 mm, a width of 1380 mm, and a depth of 700 mm. It consists of three internal layers, each with a height of 400 mm. An adjustable light source with controllable RGB values was used, and black cloth was installed to prevent light interference during the experiment. Rockwool cubes (25 × 25 × 75 mm) were used as the growth medium. The temperature was set to 24°C and the RH to 90 ± 3%. The Hoagland nutrient solution with 2.0 dS·m-1 EC and pH 6.5 was supplied (Table 1). The experiment lasted three weeks.
RH experiment
C. sativa L. ‘IT342810’ and ‘IT342811’ plants were at 30, 50, 70, and 90% RH in a growth chamber capable of controlling the RH (Fig. 2). The temperature was maintained at 24°C using white light (400–700 nm) with intensity of 80 ± 5 µmol·m-2·s-1 and a photoperiod of 20/4 h (light/dark). Rockwool cubes (25 × 25 × 75 mm) were used as the growth medium. The nutrient solution with 2.0 dS·m-1 EC and pH 6.5 was supplied (Table 1). The experiment in this case lasted three weeks.
Table 1.
Environmental conditions for hemp (Cannabis sativa L. ‘IT342810’ and ‘IT342811’) cutting adventitious root development
Temperature experiment
C. sativa L. ‘IT342810’ and ‘IT342811’ plants were grown at 15, 20, 25, and 30°C in a growth chamber capable of controlling the temperature (Fig. 3). RH was maintained at 90 ± 3% under white light (400–700 nm) with intensity of 80 ± 5 µmol·m-2·s-1 and a photoperiod of 20/4 h (light/dark). The substrate was a 50:50 blend of horticultural soil (cocopeat 51%, peat moss 10%, vermiculite 13%, perlite 15%, zeolite 10%, and humic acid 0.1%; Asia Seeds Co. Ltd., Korea) and vermiculite. Prior to the temperature experiments, a preliminary study was conducted to identify the optimal substrate. The 50:50 blend of horticultural soil and vermiculite yielded the best rooting results, leading to its use instead of rockwool for this experiment. A nutrient solution with 2.0 dS·m-1 EC at pH 6.5 was supplied (Table 1). The experiment was conducted for three weeks.
Plant growth assessment
Hemp growth was analyzed using six individual plants per treatment group. The plants were separated into shoots and roots. Shoot fresh weight (SFW) and root fresh weight (RFW) were measured using an electronic scale (MW-2N; CAS Co. Ltd., Yangju, Korea). The number and lengths of adventitious roots (AR) were analyzed using ImageJ software (version 1.54 k; National Institutes of Health, Bethesda, MD, USA) with a scale bar. The AR number (ARN) and AR lengths, including the average AR length (ARL), total AR length (TRL), and longest root length (LRL), were measured using ImageJ. For accuracy, only roots measuring at least 5 mm in length were counted and measured. An iPhone was used for photography, with the plant positioned at the center and a ruler placed beside it for the ImageJ analysis. To ensure accuracy, photos were taken vertically from a distance of one meter from the subject, using LED lighting mounted on the ceiling.
Statistical analyses
Statistical analyses of the growth experiment results were conducted using SPSS software (version 29.0.2.0 (20); SPSS Inc., Chicago, IL, USA). The mean and standard error (SE) were calculated, and analysis of variance (ANOVA) was used to analyze the data. Tukey’s multiple comparison test was used to identify significant differences among the means (p < 0.05).
Results
Growth under different light conditions
Both C. sativa L. ‘IT342810’ and ‘IT342811’ exhibited the greastest extent of root development under blue light. In contrast, root development was significantly slow under green light (Fig. 4). Additionally, SFW showed no significant difference under all light qualities (Figs. 5 and 6). For C. sativa L. ‘IT342810’, blue light resulted in the greatest growth, with RFW of 0.6 ± 0.09 g/plant, ARL of 25.2 ± 5.07 mm, ARN of 21 ± 4.21 roots, LRL of 60.6 ± 8.44 mm, and TRL of 586.9 ± 167.16 mm. Green light resulted in the lowest values for all growth parameters. Blue and white light showed no significant differences in growth parameters, except for RFW (Fig. 5). For C. sativa L. ‘IT342811’, blue light resulted in the highest growth values, with RFW of 0.41 ± 0.02 g/plant, ARN of 29.6 ± 6.1 roots, and TRL of 321 ± 85.6 mm. ARL and LRL were highest under red light, with values reaching 15.3 ± 2.07 mm and 41.1 ± 2.7 mm, respectively. ‘IT342810’ exhibited the highest values for all parameters except ARL under blue light, whereas IT342811 did not show a specific light source favoring root-related parameters (Figs. 5 and 6).
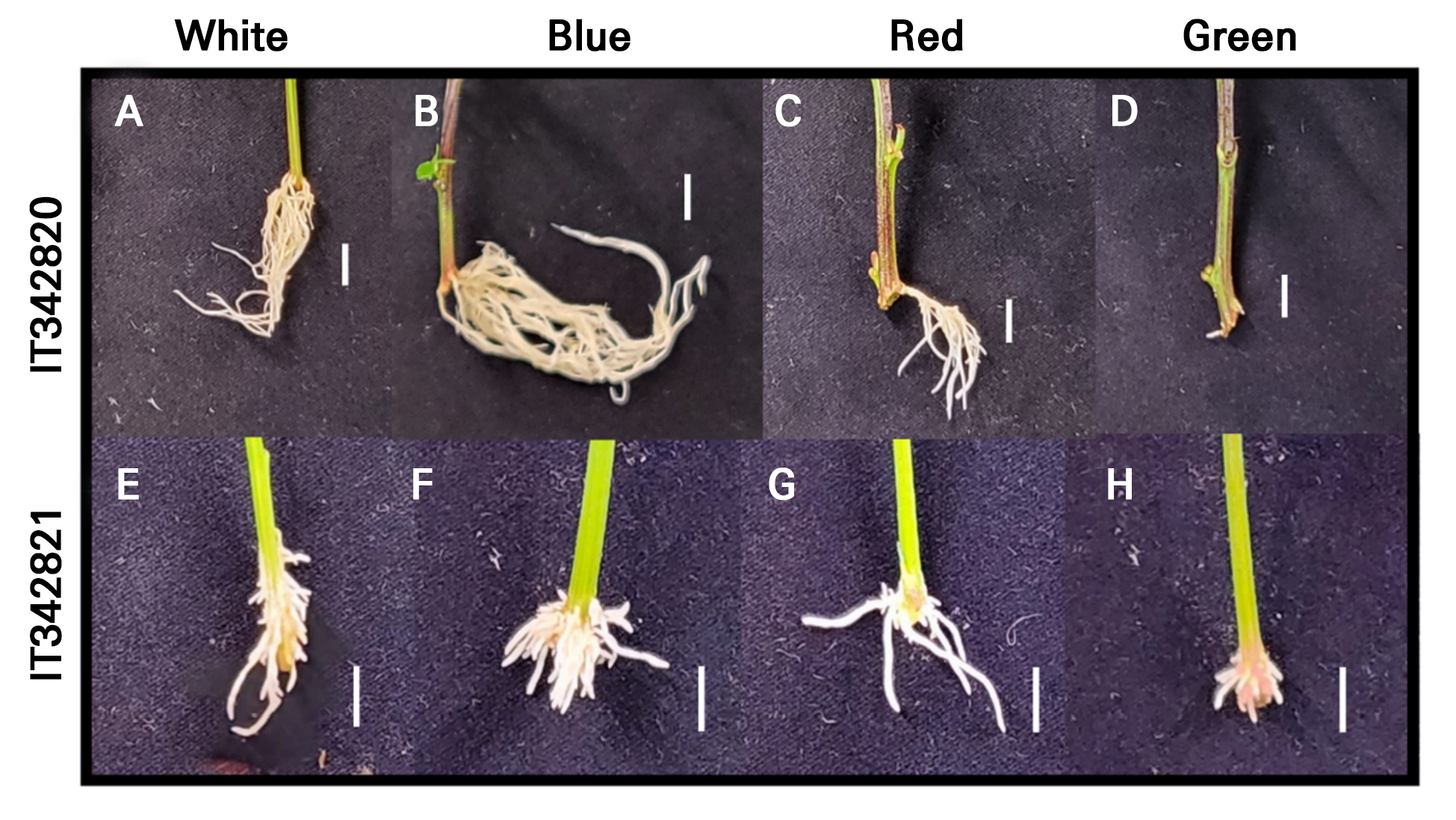
Fig. 4.
Morphology of hemp (C. sativa L. ‘IT342810’ and ‘IT342811’) adventitious roots grown under different light conditions for 21 d. (A and E) White light. (B and F) Blue light. (C and G) Red light. (D and H) Green light. (A–D) C. sativa L. ‘IT342810’. (E–H) C. sativa L. ‘IT342811’. Scale bar = 1 cm.
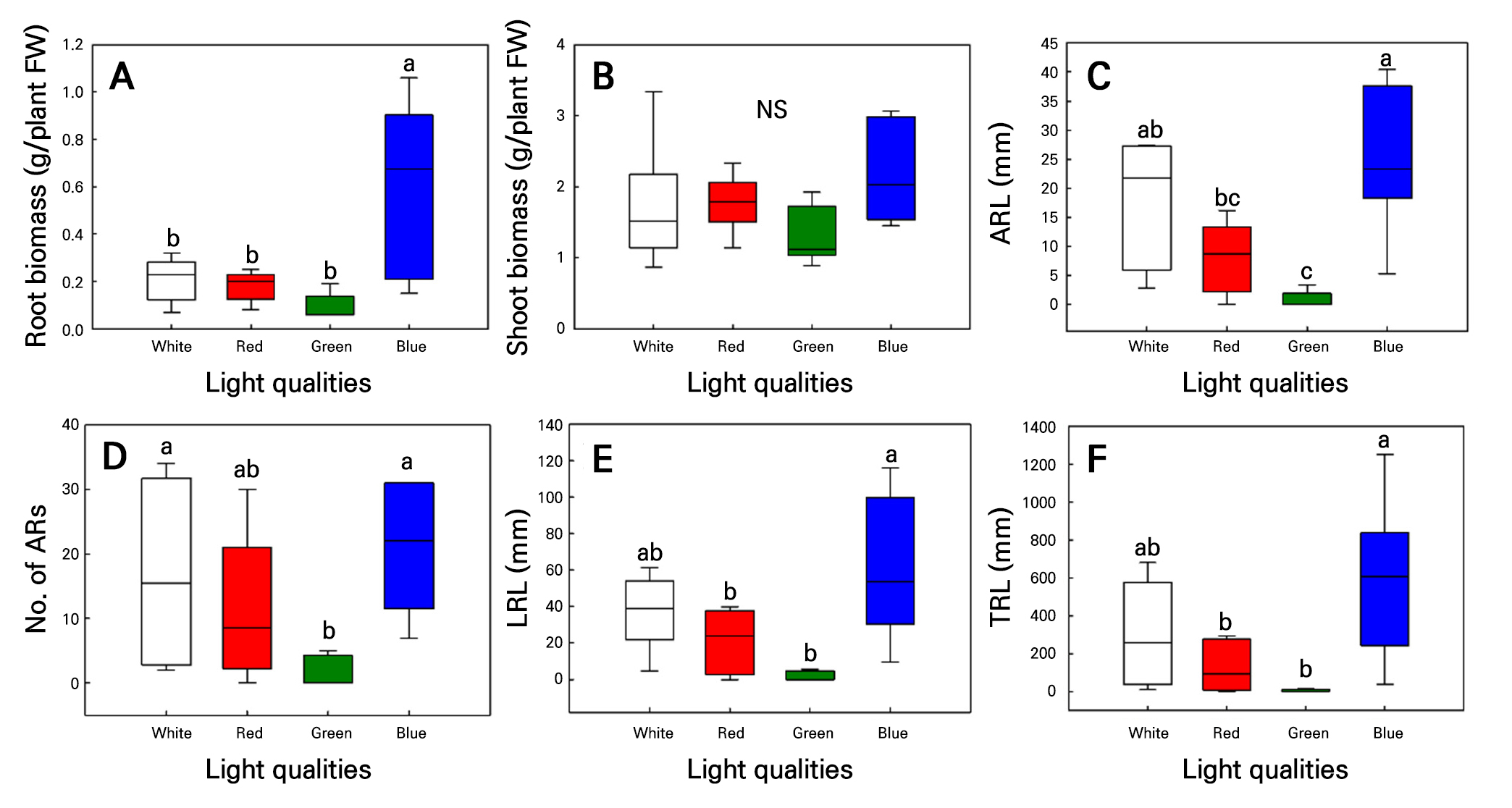
Fig. 5.
Root biomass (A), shoot biomass (B), average adventitious root length (ARL; C), adventitious root number (ARN; D), longest root length (LRL; E), and total adventitious root length (TRL; F) in hemp (C. sativa L. ‘IT342810’) shoot cuttings grown under different light conditions for 21 d. Different letters (a–c) indicate significant differences among the light treatments (Tukey’s test; p < 0.05; n = 6).
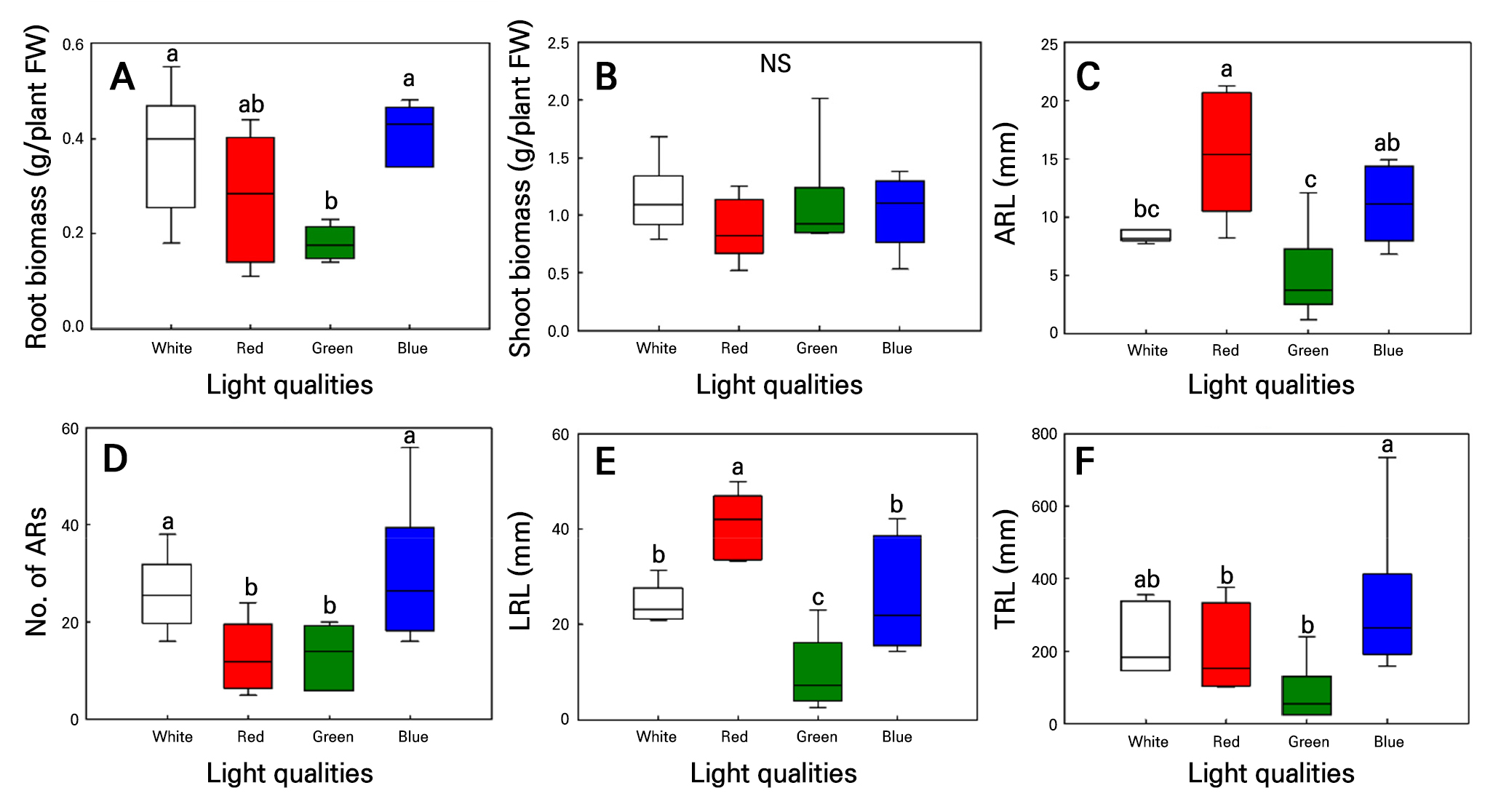
Fig. 6.
Root biomass (A), shoot biomass (B), average ARL (C), ARN (D), LRL (E), and TRL (F) in hemp (C. sativa L. ‘IT342811’) shoot cuttings grown under different light conditions for 21 d. Different letters (a–c) indicate significant differences among the light treatments (Tukey’s test; p < 0.05; n = 6).
Growth under different RH conditions
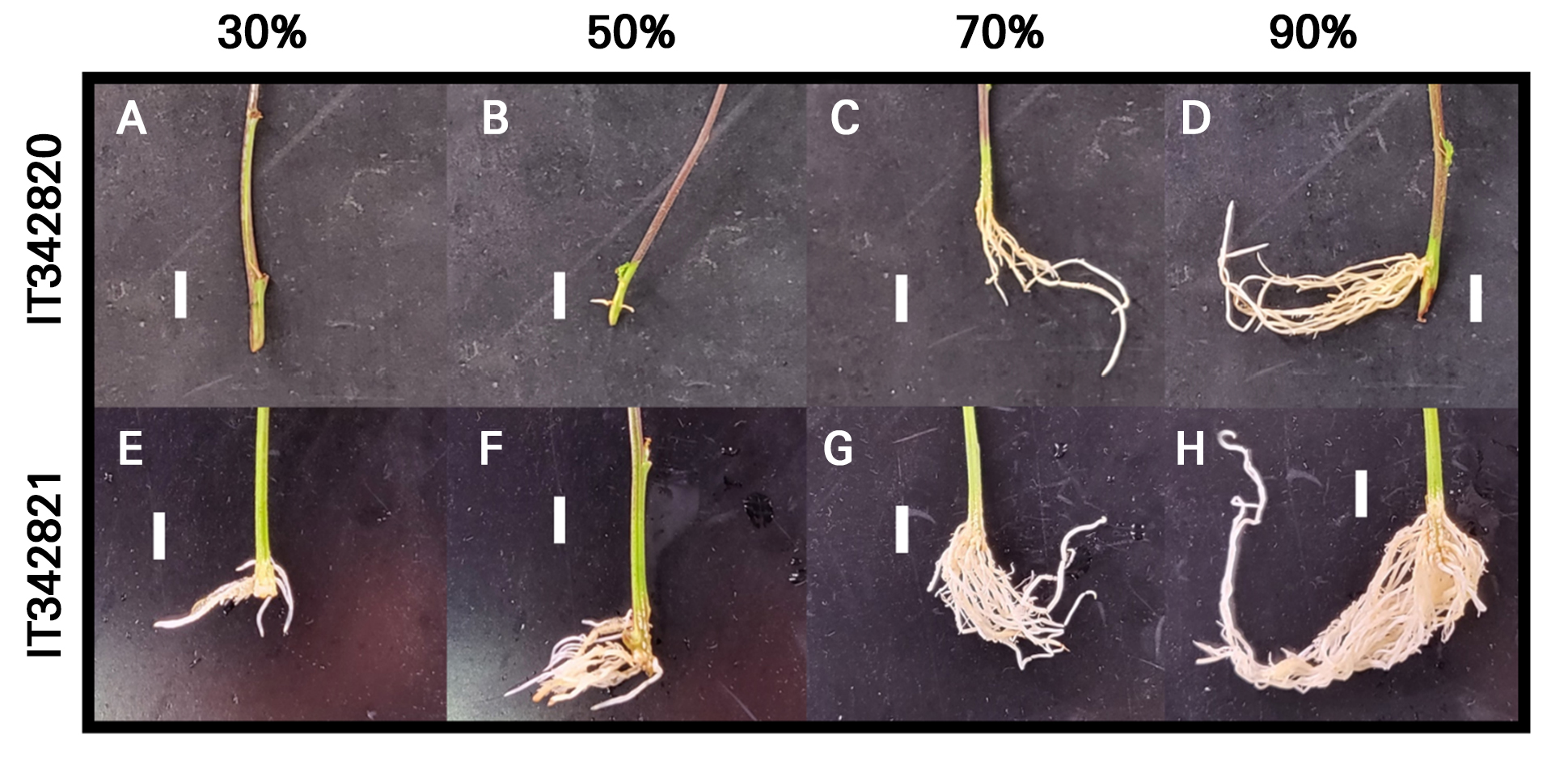
The RH experiment performed over 21 d revealed that root-related growth parameters increased with an increase in RH in both the C. sativa L. ‘IT342810’ and ‘IT342811’ cultivars (Fig. 7). For C. sativa L. ‘IT342810,’ RH 90% resulted in the highest level of growth, with RFW of 0.09 ± 0.02 g/plant, SFW of 1.3 ± 0.29 g/plant, ARL of 29.6 ± 7.02 mm, ARN of 7.1 ± 0.79 roots, LRL of 55.9 ± 9.16 mm, and TRL of 214.9 ± 82.8 mm. No significant differences were observed between the 70 and 90% RH conditions, revealing a tendency to saturate from 70% RH (Fig. 8). Similarly, for C. sativa L. ‘IT342811’, RH 90% resulted in the highest amount of growth, with RFW of 1.24 ± 0.14 g/plant, SFW of 1.97 ± 0.18 g/plant, ARL of 41.3 ± 4.86, ARN of 38 ± 7.12 mm, LRL of 91.6 ± 12.4 mm, and TRL of 145.7 ± 14.2 mm. Significant differences were observed in RFW, LRL, and TRL between the 70 and 90% RH conditions, with growth parameters showing an increasing trend with an increase in the RH (Fig. 9).
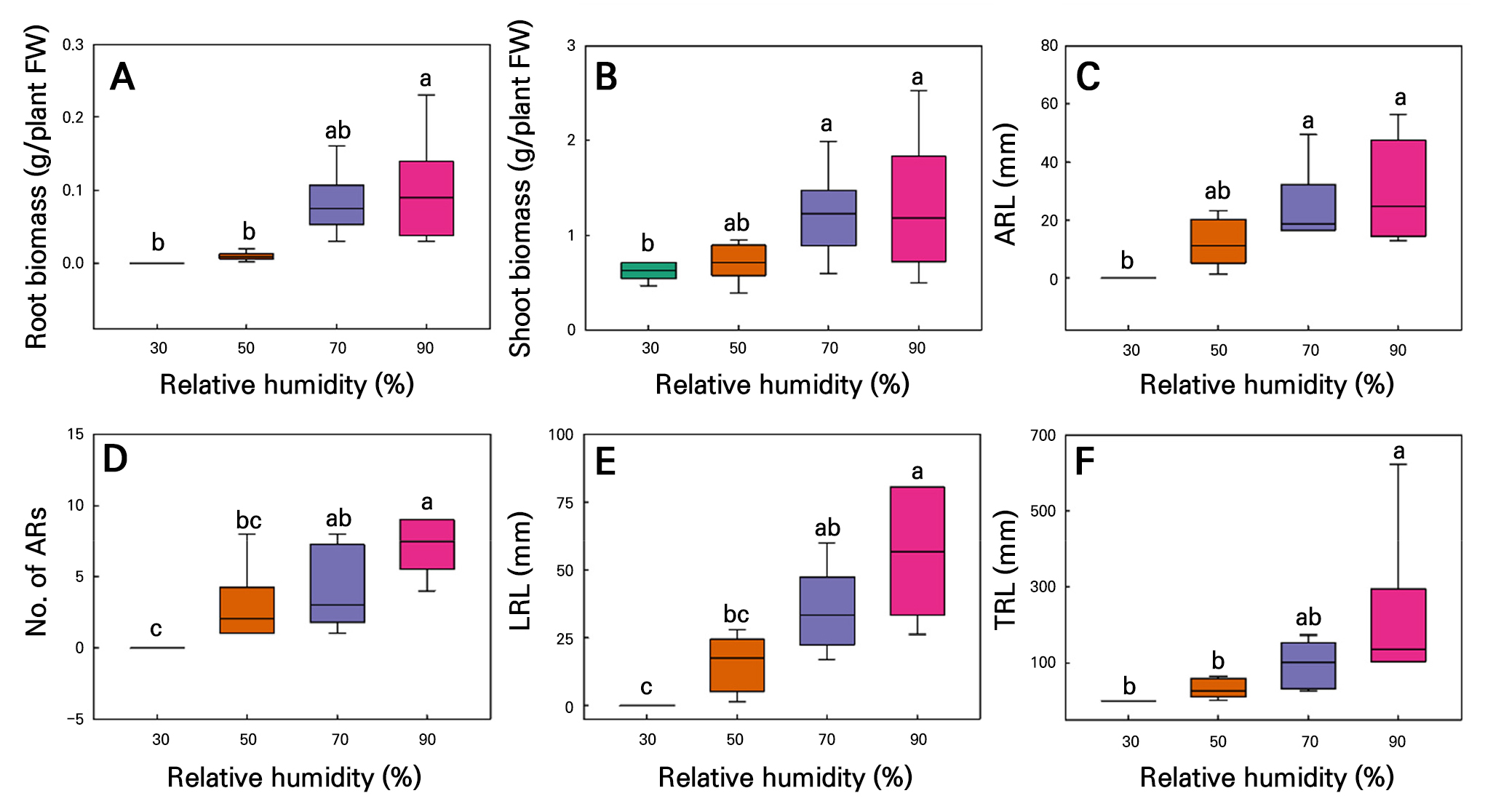
Fig. 8.
Root biomass (A), shoot biomass (B), average ARL (C), ARN (D), LRL (E), and TRL (F) in hemp (C. sativa L. ‘IT342810’) shoot cuttings cultured under different relative humidity conditions for 21 d. Different letters (a–c) indicate significant differences among the relative humidity treatments (Tukey’s test; p < 0.05; n = 6).
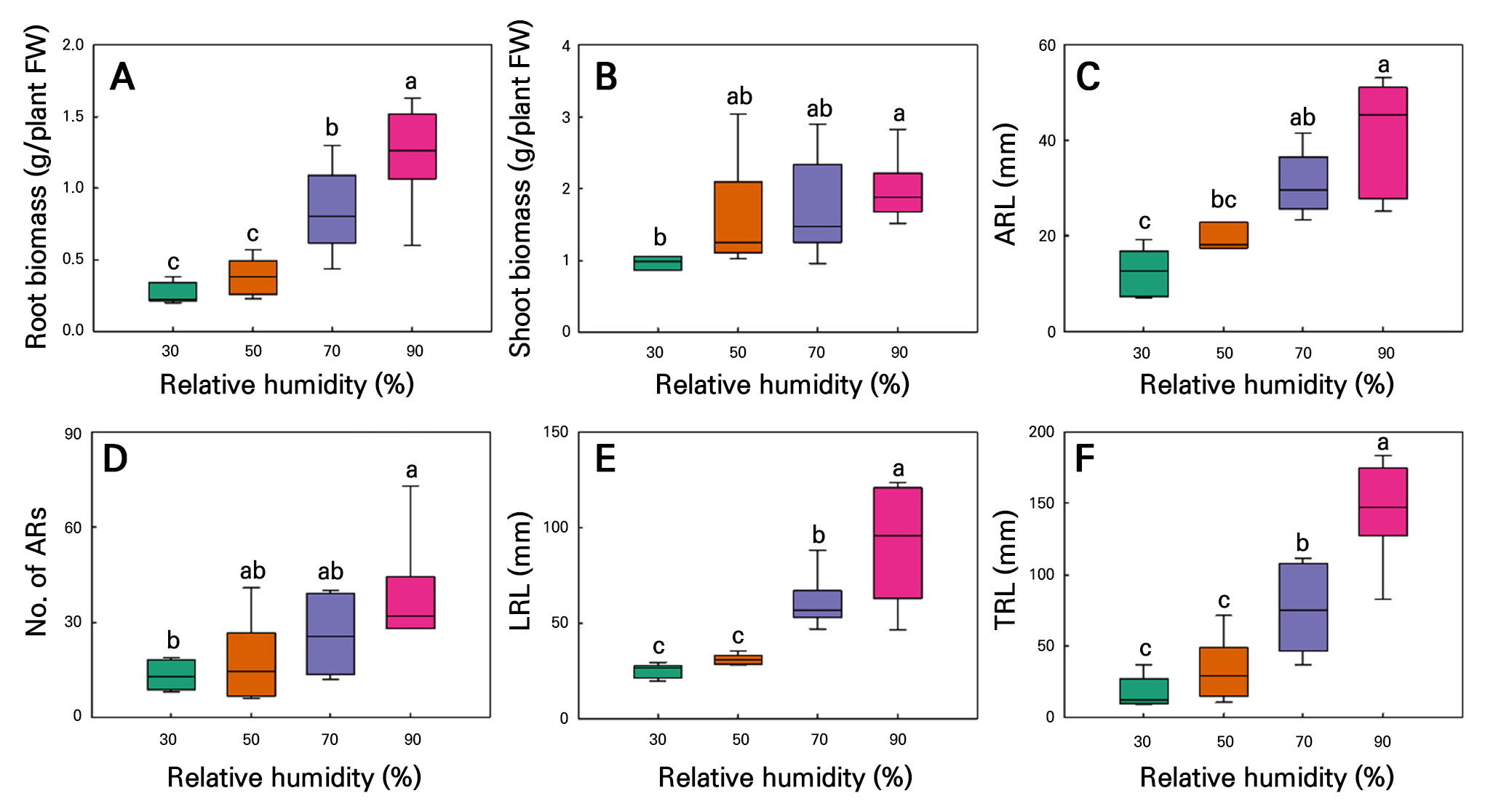
Fig. 9.
Root biomass (A), shoot biomass (B), average ARL (C), ARN (D), LRL (E), and TRL (F) in hemp (C. sativa L. ‘IT342811’) shoot cuttings grown under different relative humidity conditions for 21 d. Different letters (a–c) indicate significant differences among the relative humidity treatments (Tukey’s test; p < 0.05; n = 6).
Growth under different temperature conditions
AR formation was inhibited at low temperatures but increased as the temperatures increased (Fig. 10). For C. sativa L. ‘IT342810,’ all root growth parameters were highest at 30°C, with RFW of 0.29 ± 0.06 g/plant, SFW of 1.78 ± 0.22 g/plant, ARL of 43.6 ± 9.57 mm, ARN of 8.33 ± 2.62 roots, LRL of 77.7 ± 15.6 mm, and 288.6 of 87.9 mm. No root growth was observed at 15°C. SFW was not significantly affected by the temperature. Growth increased with the temperature, with no significant differences between 25 and 30°C; a similar trend was evident in other parameters, showing saturation at 25°C (Fig. 11). For C. sativa L. ‘IT342811,’ grown over 21 d at 15–30°C, 25°C resulted in the greastest amount of growth, with RFW of 0.57 ± 0.88 g/plant, SFW of 1.62 ± 0.17g/plant, and ARL of 51.4 ± 14.5 mm. ARN, LRL, and TRL were highest under 30°C, with corresponding values reaching 18.1 ± 3.39 mm, 80.6 ± 8.72, and 649.1 ± 115.8 mm. There were no significant differences across the 20–30°C range in the RFW, SFW, ARL, and LRL outcomes. RFW, ARL, and LRL decreased sharply at 15°C and reached saturation at 20°C (Fig. 12A, 12C, and 12E). In contrast, no significant differences were found in SFW (Fig. 12B). ARN and TRL gradually increased with rising temperatures, reaching their highest values at 30°C (Fig. 12D and 12F).
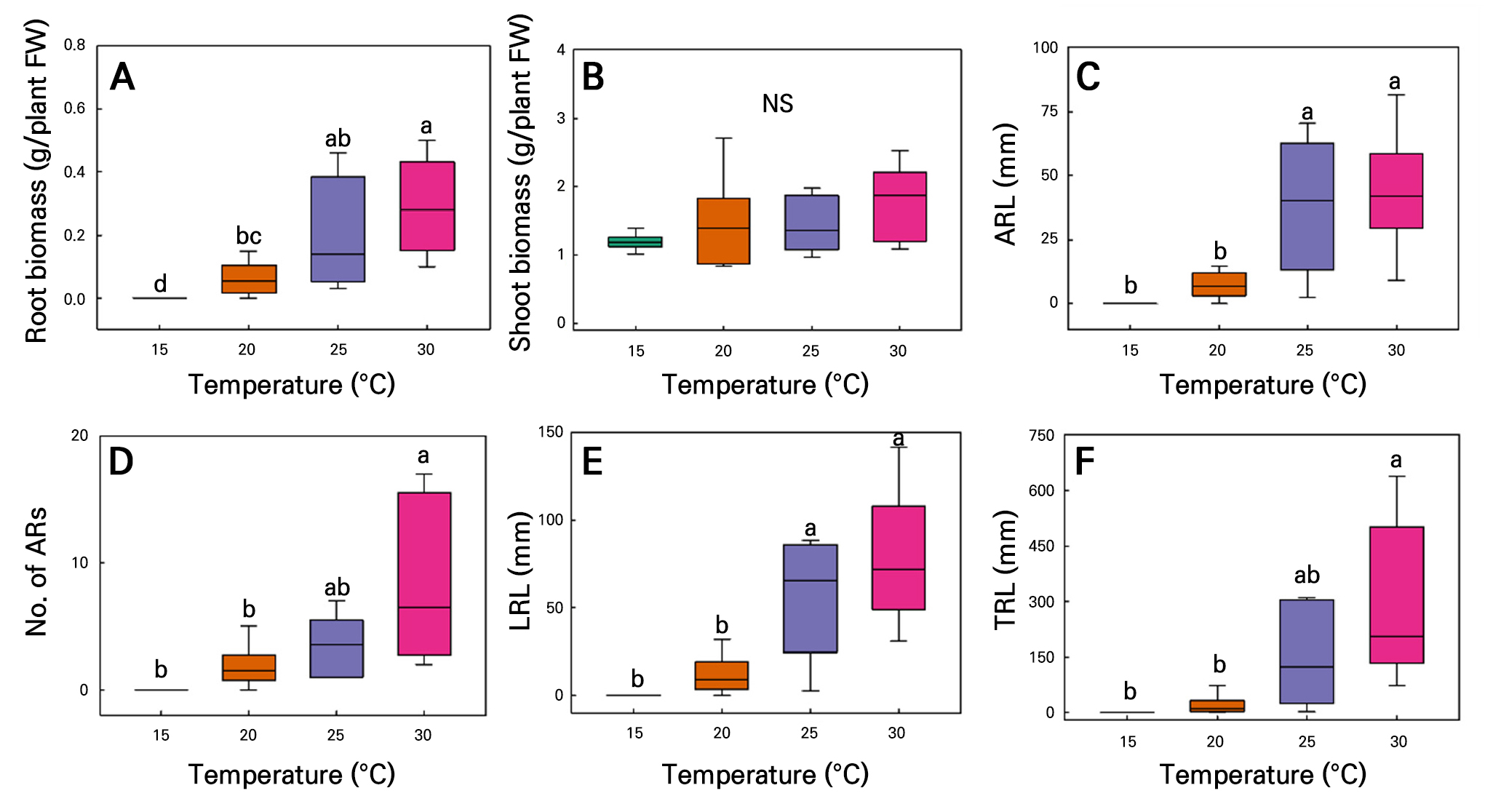
Fig. 11.
Root biomass (A), shoot biomass (B), average ARL (C), ARN (D), LRL (E), and TRL (F) in hemp (C. sativa L. ‘IT342810’) shoot cuttings grown under different temperature conditions for 21 d. Different letters (a–c) indicate significant differences among the temperature treatments (Tukey’s test; p < 0.05; n = 6).
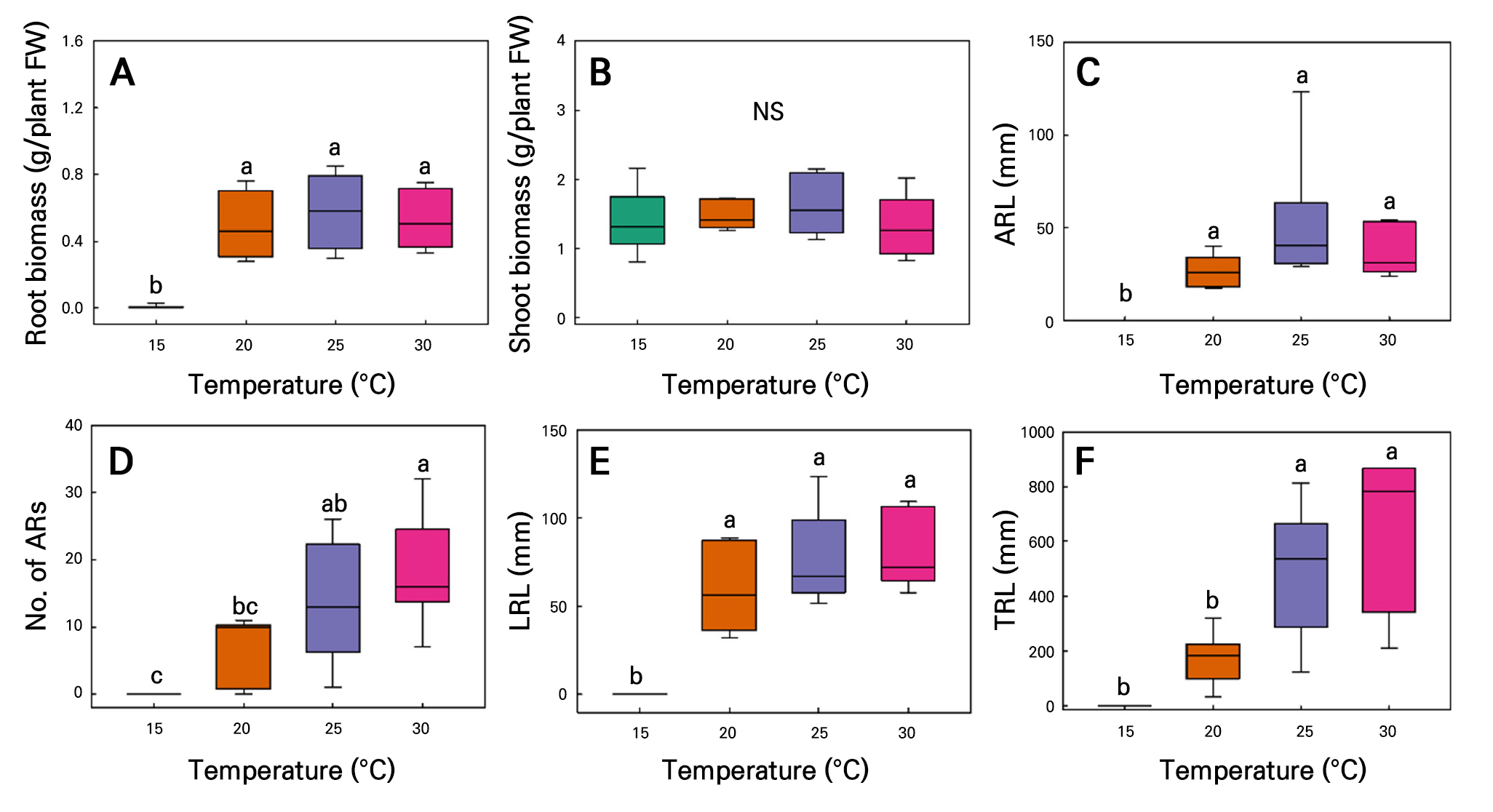
Fig. 12.
Root biomass (A), shoot biomass (B), average ARL (C), ARN (D), LRL (E), and TRL (F) in hemp (C. sativa L. 'IT342811’) shoot cuttings grown under different temperature conditions for 21 d. Different letters (a–c) indicate significant differences among the temperature treatments (Tukey’s test; p < 0.05; n = 6).
Discussion
Blue light induces adventitious rooting
The C. sativa L. ‘IT342810’ cultivar exhibited the highest values for all growth indicators. On the other hand, the C. sativa L. ‘IT342811’ cultivar did not show a specific light condition that was superior for all indicators. However, RFW, ARN, and TRL were notably higher under blue light. Additionally, both cultivars showed the lowest values for all growth indicators under green light. Therefore, it can be inferred that both cultivars experienced better underground development under blue light, whereas root growth was suppressed under green light. Blue light promotes the expression of LBD genes associated with lateral root formation in Arabidopsis thaliana (Gil et al. 2020). Additionally, CRY1, which is activated by blue light, plays a critical role in root initiation. However, green light inhibits CRY1 activity, thereby negating the beneficial effects of blue light and inhibiting root formation (Canamero et al. 2006; Folta and Maruhnich 2007). Additionally, in ‘IT342811’, ARL and LRL were higher under red light, and RFW did not show a significant difference compared to blue light. This suggests that ‘IT342811’ responded positively not only to blue light but also to red light. Red light promotes root development by reducing the concentration of phenolic compounds, such as 3,4-dihydroxybenzoic and ferulic acids, inhibiting root formation (Wu and Lin 2012). White light also resulted in higher values for certain indicators in both ‘IT342810’ and ‘IT342811’. The white light used in this experiment contained all wavelengths of the spectrum but had a relatively high proportion of green, blue, and red wavelengths (Fig. 1). Previous research has shown that blue and red light together exerts synergistic effects, leading to better root induction than monochromatic light (Iacona and Muleo 2010). However, considering that the white light used in this study contained the highest proportion of green wavelengths, which do not significantly contribute to root formation, it is likely that the high green light content limited root induction in ‘IT342810’ and ‘IT342811’ compared to monochromatic blue or red light. The results indicate that blue light was most effective for ‘IT342810’, as it exhibited the highest values across all indicators. Similarly, ‘IT342811’ showed the highest RFW under blue light, suggesting that blue light is also beneficial for its growth. However, because ‘IT342811’ also exhibited high underground growth under red light, further studies investigating the optimal ratio of blue and red light for this accession are necessary.
High RH promotes adventitious rooting
As RH increased, indicators for both cultivars tended to increase. However, ‘IT342810’ showed no significant differences among all indicators between 70% and 90% RH, whereas ‘IT342811’ exhibited significant differences in RFW, LRL, and TRL between 70% and 90% RH. Although the optimal RH for rooting varies by species, a high RH generally enhances adventitious rooting. Typically, a high RH reduces transpiration and water loss, maintains the high turgor pressure necessary for cell expansion and AR formation, and prevents dehydration (Loach 1988). For example, roses (Rosa hybrida Hortorum) exhibit high rooting rates at 90–100% RH due to reduced transpiration and the optimal internal water balance (De Hoog 2001). A high RH also upregulates OsRAU1, a gene similar to AUX1, thereby enhancing indole-3-acetic acid transport to the roots and promoting rooting (Chhun et al. 2007). ‘IT342810’ did not form roots at 30% RH, whereas ‘IT342811’ was able to root under this condition, though its growth parameters were the lowest. Poor rooting under low RH conditions is primarily due to excessive transpiration caused by insufficient humidity. Low relative humidity can lead to increased transpiration, causing greater water loss and potentially resulting in water stress, which may negatively affect root development (Kuwagata et al. 2012) Therefore, low RH conditions should be avoided when propagating ‘IT342810’ and ‘IT342811’ cuttings. Specifically, maintaining an RH range of 70–90% is essential for ‘IT342810’, while an optimal RH level of 90% is recommended for ‘IT342811’.
Specific temperatures enhance adventitious rooting
Adventitious rooting in both ‘IT342810’ and ‘IT342811’ cultivars was inhibited at 15°C (Figs. 11 and 12), confirming that 15°C is a suboptimal temperature for root development in these cultivars. Furthermore, in the ‘IT342810’ cultivar, 25°C appears to act as a temperature saturation point, with no significant variation observed in root-related parameters beyond that temperature. A similar phenomenon is noted for the ‘IT342811’ cultivar at 20°C. Inhibition of root growth at low temperatures can be due to many factors, such as reduced soil moisture, water uptake and transport issues, water potential levels, stomatal conductance, and leaf evaporation rates, all of which inhibit overall plant growth and AR formation (Flexas et al. 1999). In addition, the temperature affects phloem transport, which is important for the distribution of sugars, proteins, and hormones in plants (Taiz and Zeiger 2002). Auxin, a hormone produced in the shoot, is distributed via polar transport in the phloem (Bhalerao et al. 2002) and plays a critical role in root initiation by promoting cell division in the root pericycle and stimulating adventitious and lateral root formation (Bishopp et al. 2011). However, at low temperatures, phloem transport slows down, hindering plant growth (Ray and Savage 2021). These findings suggest that root development is a temperature-dependent process, and below a certain threshold, certain fundamental physiological functions required for root initiation may be restricted. In conclusion, the threshold temperature for adventitious root formation in both cultivars is 15°C. The optimal temperature range for root growth in ‘IT342810’ is 25–30°C, while for ‘IT342811,’ it is 20–30°C. These results highlight the differences in temperature sensitivity among hemp cultivars concerning adventitious root formation, emphasizing the need to consider cultivar-specific variations when determining the optimal temperature range for root development.
Conclusions
In conclusion, this study provides key data with which to maximize the productivity of Korean hemp cultivars during indoor cultivation. The results here indicate that adventitious root development was most active in the ‘IT342810’ cultivar under blue light, 70–90% relative humidity, and temperatures of 25–30°C, while the ‘IT342811’ cultivar exhibited optimal root formation under red and blue light, 90% relative humidity, and temperatures of 20–30°C. These findings suggest that promoting adventitious root formation can directly influence plant growth and survival, ultimately reducing the number of discarded plants and lowering production costs. While this study focused on the environmental factors affecting root formation in cuttings, it did not examine the combined effects of multiple environmental variables. Therefore, future research should build on these findings by conducting more comprehensive studies that analyze the interactive effects of various environmental conditions, further refining optimal cultivation strategies for different hemp cultivars.