Introduction
Materials and Methods
Site Description
Bacterial Strains and Culture Conditions
Auxin Analysis in Soil
Field Experiments
Soil Nutrient Content and Nutrient Uptake by Plants
Chlorophyll Content Analysis
Statistical Analysis
Results
Auxin Detection and Nutrient Content in Soil
Dry Weight and Nutrient Uptake of C. japonica Seedlings
Chlorophyll Content of C. japonica Seedlings
Discussion
Introduction
Camellia japonica, a broad-leafed evergreen woody species, is one of the most important flowering trees in southern coastal areas of Korea. C. japonica blooms in the winter, creating a pleasant display. Unfortunately, the natural habitats of this Korean Camellia species have been severely affected byhuman activities such as road construction and illegal seedling collection. The Korean government has a growing interest in restoring high quality seedling production of the ornamental plant C. japonica in coastal areas (Park, 2016).
Generally, land reclamation refers to the expansion of land over sea and coastal wetlands by filling the area with large amounts of soil. These reclaimed lands are often low in nutrients such as nitrogen and phosphorus, which are essential for plant growth and development (Sohn et al., 2010). Costly chemical fertilizers are needed to obtain high yield and quality (Dobereiner, 1997). Fertilizers create environmental problems such as leaching of nitrate into ground water, surface runoff of nitrogen and phosphorus, and eutrophication of aquatic ecosystems (Prunty and Greenland, 1997; Adesemoye and Kloepper, 2009). As an alternative to chemical fertilizers, plant growth-promoting rhizobacteria (PGPR) can be used as bio-fertilizer in organic farming practices (Mohamed and Babiker, 2012). Rhizobacteria from the genus Bacillus have been used as PGPR to improve plant growth by supplying nutrients to plants (Abbasi et al., 2011). Bacillus can provide fixed atmospheric nitrogen, solubilize inorganic phosphate, and help the host plant produce phytohormones (Dobereiner, 1997; Abbasi et al., 2011). Auxin produced by Bacillus can stimulate root development and prevent the production of ethylene, a plant growth - inhibiting hormone, by promoting the production of bacterial 1 - aminocyclopropane - 1 - carboxylate (ACC) deaminase. This root development may in turn reduce the effects of environmental stresses, including salt and drought stress (Glick et al., 1998; Patten and Glick, 2002; Suzuki et al., 2003).
Saemangeum is an estuary tidal flat that is slated by the government of South Korea for establishment of a coastal plant community, to create natural landscapes and protect against land desertification in coastal areas. Saemangeum represents the largest land reclamation project in the southwest coast of Korea (Cho, 2007). The project will create 28,300 km2 of reclaimed land and a lake as large as 11,800 km2 by constructing the world’s longest (33.9 km) sea dike (Lie et al., 2008). It is scheduled to include agricultural land, industrial land, city parks, and an arboretum, among other things (Ryu et al., 2010). However, ornamental plant seedlings in Saemangeum reclaimed coastal land have low growth rates, mainly because of salt stress (Sohn et al., 2010).
Soil salinity is a major problem for plants in reclaimed coastal lands. Elevated Na+ levels in the soil solution can drive water out of the cells, reducing cell turgor, leaf area, and consequently reducing photosynthetic activity, growth and yield (Parida and Das, 2005; Rojas - Tapias et al., 2012; Qiu et al., 2003; Koyro, 2006). Salinity-induced osmotic stress can reduce water uptake by plant roots and induce stomatal closure in shoots, reducing evaporation and overall water transport (Rojas - Tapias et al., 2012). In addition, salinity can cause ionic stress due to high concentrations of potentially toxic salt ions within plant cells (Kohler et al., 2009). In recent years, a new biocontrol approach was developed to protect plants from salt stress by growing them in the presence of PGPR (Yue et al., 2007).
Several studies have demonstrated the beneficial effects of PGPR on nutrient uptake and photosynthesis in plants (Esitken et al., 2006; Aslantas et al., 2007). PGPR are important for managing ornamental plant growth because they have beneficial effects on soil conditions, nutrient availability, tree growth, and yield. However, there are no data on the effect of PGPR on cultivation of the ornamental plant C. japonica in reclaimed coastal land. The objective of this study was to determine whether inoculation with Bacillus licheniformis MH48 could promote nutrient uptake in C. japonica seedlings in high salinity and low nutrient environments, for the purpose of producing high quality seedlings in the Saemangeum reclaimed coastal land of Korea.
Materials and Methods
Site Description
Saemangeum is an estuarine tidal flat on the coast of the Yellow Sea in South Korea that lays at the mouths of the Dongjin and Mangyeong Rivers on the coast of Jeollabuk - do (Fig. 1). A dam was added by the government of South Korea to the Saemangeum Seawall Project so that an estuarine tidal flat could be converted to either agricultural or industrial land. The dam was completed in April 2006. The project of filling in the estuary began in 1991, but the project was slowed down by a series of court actions of by environmentalists. The completed seawall is approximately 33 km long and replaced a coastline that was once more than 100 km long. After the estuary is completely filled, an area of about 400 km2 will be added to the Korean peninsula, making it one of the biggest land reclamation projects in history. The soil is classified as silt loam with a slope of 0 - 2% (Korea Soil Information System, 2016) and has low phosphate content and high salinity. Soil salinity can also have a harmful effect on the growth of trees, as many trees can be killed or suffer from severe physiological disorders such as necrosis of leaves, arrested shoot growth, and leaf dropping due to salt stress (Sohn et al., 2010). The climate in Saemangeum is characterized by a total annual rainfall of 1,141 mm and annual average temperature of 13°C from January to December 2014 (Korea Meteorological Administration, 2016).
Bacterial Strains and Culture Conditions
The bacterial strain B. licheniformis MH48, a rhizobacterium, was isolated from an experimental site in the Saemangeum coastal area (Park, 2016). To prepare inoculum, B. licheniformis MH48 was cultured in broth media (0.15% urea ((NH2)2CO), 0.04% potassium phosphate monobasic (KH2PO4), 0.03% potassium chloride (KCl), 0.1% organic compost, and 0.22% sugar) and incubated at 30°C for 5 days.
Auxin Analysis in Soil
To determine whether auxin was produced by B. licheniformis MH48, soil samples were taken from 0 to 30 cm depth in 1 week after bacteria inoculation to plant pots and in control plant pots without bacteria inoculation, respectively. Soil samples were completely dried at room temperature and sifted with a 2 mm sieve. The presence of auxin in soil was determined according to Sarwar et al. (1992) with minor modifications. Each dried soil sample (10 g) was placed in a flask and treated with 19 mL of 0.2 M phosphate buffer (pH 7), 7 mL of 5% trichloroacetic acid, and 4 mL of 0.5 M calcium chloride. The flasks were covered with parafilm and incubated in a shaking incubator (at 150 rpm) for 5 min. After the incubation, the solution was filtered through Whatman filter paper No. 2. The soil filtrate was then partitioned with acidic ethyl acetate (3 : 1 v / v). The ethyl acetate fraction were separated from the aqueous fraction, reduced in volume, and dissolved in 2 mL methanol. Auxin was measured by high-pressure liquid chromatography (Shimazu LC - 10Avp HPLC system; Shinmadzu, Kyoto, Japan) using a C18 reversed - phase column (Frankenberger and Brunner, 1982). Acetonitrile and 50 mM KH2PO4 (pH 3; 30 / 70) were used for the mobile phase at a flow rate of 1 mL·min-1 (Patten and Glick, 2002). The presence of auxin in soil was determined by comparing its retention time and standard peak size.
Field Experiments
Field experiment was conducted by using a randomized complete block design by cutting furrows to 5 m width × 5 m height. The following two treatment groups with three replicates per treatment were used in this experiment : (1) control (without bacteria) ; (2) B. licheniformis MH48 inoculation. Generally, land reclamation in coastal areas filling the sea with large amounts of soil for plant cultivation to alleviate salinity conditions. Study sites had filling sandy soil with 30 cm. Seedlings of C. japonica s (4 year old at height of 50 - 60 cm) were planted with 70 cm × 70 cm spacing into each treatment in July 2014, with a total of 54 seedlings per treatment (3 replicates per treatment, 18 seedlings per replicate). One month after planting, 20 L B. licheniformis MH48 (cultured as described above) was poured into the soil adjacent to the roots. Control seedlings were not treated with any bacteria.
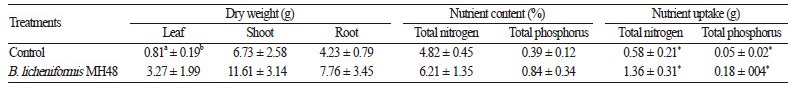
Soil Nutrient Content and Nutrient Uptake by Plants
Soil samples were taken from 0 to 30 cm depth at the end of each experiment in March 2015 to analyze total nitrogen and phosphorus content. Soil samples were oven-dried at 105°C for 24 h. After drying, soil samples were sifted through a 2 - mm sieve. To determine total nitrogen and phosphorus uptake by C. japonica seedlings after inoculation with B. licheniformis MH48, dry weight and nutrient content of seedlings samples were measured. Three seedlings from each treatment were randomly sampled in March 2015. Leaves, shoots, and roots were separated and rinsed with deionized water. Their dry weight was recorded after oven-drying at 65°C for 48 h. The samples were then pulverized and filtered through a 30 - mesh screen followed by total nitrogen and phosphorus content analyses. Nutrient uptake of C. japonica seedlings was calculated using the following formula (Liu et al., 2013): Nutrient uptake (g) = (dry weight (g) × nutrient content (%)) / 100.
Total nitrogen content in soils was determined using the Kjeldahl method (Mulvaney, 1996) after wet digestion with H2SO4. Total nitrogen content of seedlings was analyzed using an elemental analyzer (Variomax CN Analyzer, Elemental, Germany) with thermal conductivity detector (TCD) after combustion at high temperature (1200°C) with nitrogen and helium gas. Total phosphorus content in soil and seedling samples was determined using an ICP - OES (Optima 8300,PerkinElmer, USA) after digestion in hydrochloric acid : nitric acid (3 : 1 v / v) and heating in a Microwave Oven (MARS Xpress, CEM corporation, USA).
Chlorophyll Content Analysis
Chlorophyll content of C. japonica seedlings was measured once per month from January 2015 to March 2015 using a chlorophyll meter (SPAD - 502plus, Minolta, Japan). Mean chlorophyll content of a total of fifty leaves was measured. The unit of chlorophyll content was indicated by the SPAD - value.
Statistical Analysis
Statistical analyses were carried out using the software package SPSS version 21 (IBM SPSS Statistics, USA). Soil nutrient (total nitrogen and total phosphorus) and seedling data (dry weight, nutrient content, nutrient uptake, and chlorophyll content) were analyzed using Student’s t - test (95% confidence) to evaluate the difference between inoculation with B. licheniformis MH48 and control (without bacteria inoculation).
Results
Auxin Detection and Nutrient Content in Soil
Auxin in soils was detected by HPLC analysis (Fig. 2). The extracted fraction of inoculated soil filtrate had a sharp peak with the same retention time (5.507 min) as that observed for the auxin standard in HPLC. Auxin was not detected in the control soil filtrate without bacteria inoculation (Fig. 2).
Fig. 2.
HPLC chromatograms of auxin standard (dotted line), control soil extracts without bacterial inoculation (double dotted line), and extracts from soil inoculated with B. licheniformis MH48 (solid line)..
Total nitrogen and phosphorus content of soils with inoculation of B. licheniformis MH48 was significantly higher ( p < 0.05) than the control without bacterial inoculation (Fig. 3). The average total nitrogen content of soils inoculated with B. licheniformis MH48 was 1.3 g·kg-1, 2.2 - fold higher than the control. The application of B. licheniformis MH48 also resulted in a 20.0 - fold increase in total phosphorus content in the soil when compared to the control.
Fig. 3.
Effect of inoculation with B. licheniformis MH48 on total nitrogen (A) and total phosphorus (B) content in soil. Error bars represent standard deviation. *indicates significantly different based on a Student’s t -test ( p < 0.05).
Dry Weight and Nutrient Uptake of C. japonica Seedlings
Treatment with B. licheniformis MH48 had no significant effect ( p > 0.05) on leaf, shoot, or root dry weight or nutrient content (total nitrogen and total phosphorus) of C. japonica seedlings (Table 1). Bacterial inoculation also did not significantly stimulate ( p > 0.05) seedling growth. Although not statistically significant, the root dry weight of seedlings with inoculation of B. licheniformis MH48 showed a tendency to be higher (1.8 fold) than the control without bacterial inoculation (Table 1).
Total uptake of nitrogen and phosphorus in C. japonica seedlings was significantly higher ( p < 0.05) after treatment with B. licheniformis MH48 when compared to the control (Table 1). The average total nitrogen uptake of seedlings with and without inoculation of B. licheniformis MH48 was 1.36 g and 0.58 g, respectively. The average total phosphorus uptake of seedlings with inoculation of B. licheniformis MH48 and control seedlings were 0.18 g and 0.05 g, respectively.
Chlorophyll Content of C. japonica Seedlings
The average chlorophyll content in leaves of C. japonica seedlings grown in soil inoculated with B. licheniformis MH48 was significantly higher ( p < 0.05) than in leaves of control seedlings (Fig. 4). The average chlorophyll content (SPAD value) in leaves of C. japonica seedlings in the treatment with inoculation of B. licheniformis MH48 was 64.6.
Discussion
Soil salinity is a limiting factor for plant growth in the Saemangeum reclaimed coastal land (Sohn et al., 2010). In this study, nutrient uptake and chlorophyll content of C. japonica seedlings were lower in the absence of bacterial inoculation, likely due to salt stress (Table 1 and Fig. 4). Our results agree with those of Mohamed and Gomaa (2012) and Rojas - Tapias et al. (2012) showing that soil salinity can decrease nutrient uptake and photosynthetic activity in plants. Salt stress is likely associated with decreased photosynthesis due to partial stomatal closure (Mohamed and Gomaa, 2012; Rojas - Tapias et al., 2012).
High - salinity conditions often reduce seedling nutrient uptake due to competition between Na and nutrients such as nitrogen and phosphorus (Mohamed and Gomaa, 2012). Phosphorus and nitrogen are essential nutrients for plant growth and development (Orhan et al., 2006). Recently, a new biocontrol approach was developed to supply nutrients and protect plants from salt stress in soils by treating seedlings with PGPR (Orhan et al., 2006; Yue et al., 2007; Rojas - Tapias et al., 2012). Bacillus species can increase the total content of nitrogen and phosphorus in soils via mechanisms such as atmospheric nitrogen fixation and solubilization of phosphorus by organic acid exudation (Rodriguez and Fraga, 1999; Vazquez et al., 2000; Beneduzi et al., 2008). Our study suggests that inoculation with B. licheniformis MH48 can increase total nitrogen and phosphorus content in soils, as well as nitrogen and phosphorus uptake by seedlings.
Although the root growth of seedlings after inoculation with B. licheniformis MH48 was not significantly different fromcontrol seedlings without bacterial inoculation, root dry weight was 1.8 times higher (Table 1). This may be due to the formation of fine roots such as lateral roots and root hairs in seedlings with bacterial inoculation. Root hairs play an important role in nutrient uptake (Leitner et al., 2010). It has been reported that auxin secreted by bacteria may directly promote the development of fine roots, especially root hairs, by stimulating cell elongation and division (Lee and Cho, 2013; Salazar-Henao et al., 2016). Auxin may also indirectly promote root growth, by increasing the activity of bacterialACC deaminase, which prevents the production of growth-inhibiting levels of ethylene (Aslantas et al., 2007). In the present study, B. licheniformis MH48 led to auxin accumulation in the soil (Fig. 2), but did not significantly promote the growth of C. japonica seedlings compared to the control. Thus, auxin produced by B. licheniformis MH48 may stimulate fine root development and alleviate salt stress in seedlings, resulting in higher rates of nutrient uptake by the roots. Chlorophyll content of C. japonica seedlings was also significantly increased by the inoculation of B. licheniformis MH48 (Fig. 3). This may result from higher nutrient uptake, particularly nitrogen, required for chlorophyll biosynthesis (Beneduzi et al., 2008). Several studies have reported that nitrogen-fixing PGPR can increase chlorophyll content in plants (Beneduzi et al., 2008; Rojas - Tapias et al., 2012). Our study suggests that auxin produced by B. licheniformis MH48 may improve nutrient uptake in C. japonica seedlings due to fine root development and salt-stress alleviation, which in turn increases chlorophyll content under high-salinity conditions (Fig. 4).