Introduction
Material and Methods
Germplasm
Evaluation of Plants in Response to Chilling Temperatures
Data Analysis
DNA Extraction, PCR, and Restriction Enzyme Analysis for Genotype Screening
Notation of Plastidic Types
Results
Chilling Response
Genotype Analysis
Discussion
Conclusion
Introduction
Low temperatures limit plant growth (Gomes et al., 2000) and can cause significant economic losses in vegetable crop species worldwide (Synder and Abreu, 2005). Temperatures near freezing disrupt plant growth in cold-sensitive plants causing stunting and injury (Thomashow, 1999). In fact, plant exposure to non-freezing temperatures below 12°C often causes Chilling injury in many tropical and sub-tropical plants, especially when grown in temperate climates (Saltveit and Morris, 1990). For instance, cotton (Gossypium hirsutum L.), rice (Oryza sativa L.), maize (Zea mays L.), tomato (Solanum lycopersicum L.), and cucumber (Cucumis sativus L.) are not considered Chilling tolerant (Jackman et al., 1988). SuCh species suffer a loss of productivity due to photo-inhibition resulting from a disruption of photosynthesis and associated metabolic pathways (Hetherington and Smillie, 1989).
There has been historic researCh interest in Characterizing the plant response and meChanisms associated with tolerance to Chilling temperatures (Smallwood and Bowles, 2002). Plant growth and acclimation to sub-optimal temperatures has been well documented in a number of species (Hughes and Dunn, 1996; Thomashow, 1999). Response of plants to low temperature stress, as well as signal transduction that promote or suppress gene function depends on a plant’s ability to perceive Chilling temperatures. In some species, non-freezing low temperature plant response is related to drought and salinity tolerance (Smallwood and Bowles, 2002).
Chilling temperatures (above 0°C) often affects cell mem-brane fluidity, enzyme activity, and the stability of nucleic acids (e.g., RNA and DNA secondary structures) and enzyme activity (Smallwood and Bowles, 2002). Chilling temperatures can affect enzyme-modulated RNA and DNA transcription, translation, and cellular metabolism (Huner et al., 1998). For instance, polyunsaturated acyl Chains of membrane phospholipids increase with decreasing temperatures, resulting in Changes in membrane fluidity (Nishida and Murata, 1996). Disruptions in these plant processes initiated by low tem-peratures can lead to cellular energy imbalances (Nishida and Murata, 1996) and subsequently to various negative plant responses suCh as dehydration resulting from decreased water absorption and translocation by roots (Huner et al., 1998; Smallwood and Bowles, 2002).
Chilling resistance is an important trait that is not present in modern U.S. processing cucumber (C. sativus L.) cultivars (Saltveit and Morris, 1990). Chilling temperatures (0-12°C) often cause severe economic damage to cucumber foliage and fruit during field and greenhouse production (Chung et al., 2003b; Staub and Wehner, 1996). Chilling tolerance in cucumber has both nuclear (Kozik and Wehner, 2008) and maternal (Chung et al., 2003b, 2007; Gordon and Staub, 2011) genetic components. A single dominant gene controls Chilling tolerance in the nucleus (Ch; Kozik and Wehner, 2008) and multiple factors condition maternal control (i.e., at least two Chloroplast candidate genes; Chung et al., 2007) of Chilling response in cucumber. The interaction between nuclear (Kozik and Wehner, 2008) and cytoplasmic (Chung et al., 2007) factors during Chilling has been described in U.S. processing cucumber (Gordon and Staub, 2011). Plant damage is dependent on the interplay of these genetic factors (Gordon and Staub, 2011) and the intensity and duration of Chilling temperatures (Chung et al., 2003b; Smeets and Wehner, 1997).
The Chloroplast [e.g. ‘Chipper’ (tolerant) and line Gy-14 (susceptible)] and nuclear [e.g. line NC76 (tolerant)] Chilling tolerance of several U.S. processing type cucumbers has been Characterized, but little is known regarding Chilling response in other market types (e.g. fresh market). Although Asian, European, and U.S.A. cucumber market types are genetically different (Horejsi and Staub, 1999), and Chilling tolerance has been reported in Korean cucumber germplasm (Kuk and Shin, 2007), the strength of their response compared to Chilling tolerant U.S. processing cucumber types is not known.
Therefore, a study was designed to compare the Chilling response of several previously identified susceptible and tolerant U.S. and Asian (Korean, Japanese and Chinese) cucumber lines and to determine Chloroplast marker/Chilling phenotype associations in Korean fresh market cucumber. This comparative analysis will allow for strategic integration of Chilling tolerance genes into elite Korean germplasm through plant improvement.
Materials and Methods
Germplasm
Chilling tolerant and susceptible cucumber germplasms used in this study were obtained from different sources including the U. S. Department of Agriculture (USDA), Agricultural ResearCh Service, Cucumber Breeding Project, Madison, WI (U.S. processing cucumber types), and Asian fresh market types from the Kurume Vegetable Breeding Company, Fukuoka, Japan, Tianjin Chunrui Vegetable Growers Co-operative, Tianjin, Peoples Republic of China, and various local vegetable seed companies (Table 1). Of the 16 germ-plasms used herein, 12 accessions were of Korean origin, two originated from the U.S.A. [line NC76 (Chilling tolerance conditioned by nuclear gene, Ch; Kozik and Wehner, 2008), ‘Chipper’ (Chilling tolerance conditioned by maternal factors; Chung et al., 2007)], one from China (‘Dongguan’ Chilling tolerant; genetic basis of tolerance unknown; Ali et al., 2013) and one from Japan (‘Suiseifushinari 2-go’ unknown Chilling response; Ali et al., 2013). The Korean accessions used were selected either because of their known Chilling injury (CI) tolerance according to seed companies (Ali et al., 2013) or their importance to the Korean horticulture industry. The phenotypic response to Chilling temperatures (4°C) and nuclear and Chloroplast genotype of line NC76 and ‘Chipper’ were previously classified by Chung et al. (2003b), Kozik and Wehner (2008) and Gordon and Staub (2011), and were used herein as standards for comparison.
Evaluation of Plants in Response to Chilling Temperatures
Seeds were sown in plug pots (50 cm × 50 cm) in a greenhouse at Dongguk University, Seoul, Korea containing Sunshine mix # 4 - aggregate plus (SunGro Horticulture, Bellevue, WA, U.S.A.). Seeds were germinated at 22 (light)/18 (dark)°C, under a 9 hours photoperiod at a light level of 176 μmoles・m-2・s-1 PPF supplied by cool-white fluorescent and incandescent lamps. Relative humidity (RH) was held at 50%, and emerged seedlings were watered one time daily and with soluble fertilizer (20N:09P:20K, TeChnigro: SunGro Horticulture) weekly.
Chilling treatments were performed under controlled environment conditions according to Chung et al. (2003b). When first true leaves were fully opened with no remaining adaxial leaf curl, plants were subjected to a Chilling treatment of 4°C (Experiment 1) and 1°C (Experiment 2 and Experiment 3) for 8 (08:00 to 16:00) and 9 (08:00 to 17:00) hours, respectively (immediate and constant). These Chilling tem-perature regimes were Chosen according to the historic precedence of Chilling Challenge at 4°C to cause damage to seedlings (Chung et al., 2003b; Gordon and Staub, 2011; Smeets and Wehner, 1997) and the lack of knowledge of the response of known tolerant (at 4°C) germplasm at 1°C (e.g. ‘Chipper’; Chung et al., 2007). In eaCh experiment, plants were arranged in a completely randomized design (CRD).
The light level was 149 mmoles・m-2・s-1 PPF during Chilling treatment where RH was measured at ~ 80%. This provided a uniform physiological age for Chilling damage assessment. During Chilling treatment seedlings were watered at the soil level only to the extent of eliminating dryness and were provided the standard watering on conclusion of the Chilling protocol. After Chilling, plants were returned to pretreatment conditions and leaf damage was quantified by visual rating for 5 days after the Chilling treatment. Chilling damage on the first true leaves was scored from 1 to 5 as shown in Fig. 1, where a damage rating (DR) of 1 = no damage, 2 = slight damage, 3 = moderated damage, 4 = advanced damage, and 5 = severe damage. This rating score sCheme was modified from the 0-9 scale of Smeets and Wehner (1997) since the visual damage observed herein was comparatively less pronounced.
Data Analysis
Visual ratings data from the three experiments were subjected to analysis of variance (ANOVA) using a complete randomized design. Chilling temperature/experiments were evaluated separately for assessment of treatment (entry) effects using Statistix 8.1 statistical analysis package (Analytical Software, Tallahassee, FL, USA). Entry means and standard deviations were calculated and mean difference tests were performed using least significant (LSD) tests to group germplasm responses (P< 0.05).
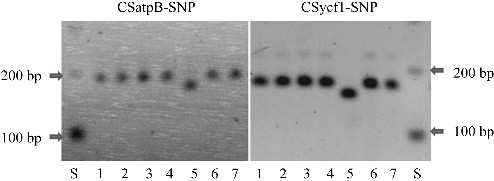
DNA Extraction, PCR, and Restriction Enzyme Analysis for Genotype Screening
To determine Chloroplast marker/Chilling phenotype associa-tions in Korean fresh market cucumber, two sdCAPS Chloroplast markers, CSatpB-SNP and CSycf1-SNP developed by Ali et al. (2013) were employed to define the Chloroplast genotype designations of ‘NacdongChungjang’ (phenotypically Chilling susceptible) and ‘JanghyungnaChap’ (phenotypically Chilling tolerant) (phenotypic Chilling response designated by this study). Likewise, these markers were also employed for genotypic Characterization of Chilling susceptible cucumber cultivars, ‘Joeunbaegdadagi’, ‘Sinjungpum’, ‘White’, ‘Dongguan’, and Chilling tolerant line NC76 according to Ali et al. (2013).
Total DNA was extracted according to Chung et al. (2003a). All PCR reagents were obtained from the Solgent Corporation (Seoul, South Korea) and restriction enzymes used for experimentation were purChased from NEB (IpswiCh, MA, USA). EaCh 15 µL reaction volume contained 4.0 mM of MgCl2, 0.3 mM of dNTPs, 15 ng of DNA, 0.45 µM of primers, polymerase buffer and 0.2 units of Taq DNA polymerase. All amplifications were conducted using the following cycling profile: 94°C for 5 min; 40 cycles of 94°C for 30 s, 50°C for 60 s, 72°C for 60 s; 72°C for 6 min, and then followed by an indefinite holding at 4°C. For CSatpB-SNP, Sau96I and for CSycf1-SNP, RsaI REs were applied to PCR products according to a manufacturer’s protocol.
The digested PCR products were electrophoresed in 1.5% agarose gels in 0.5X TBE buffer (0.045 M Tris-borate and 1.0 mM EDTA pH 8.0) for 3 hours at approximately 170 V, and then gels were stained with ethidium bromide (0.5 mg・mL-1 in TBE) and banding patterns were captured with a digital camera (WGD-30, Daihan Scientific, Seoul, South Korea). Gels were scored in accordance with their expected band sizes.
Notation of Plastidic Types
Accessions were given the plastid Chilling sensitivity designation of s for susceptible and t for tolerant. These Chilling sensitivities are presented as prefixes in describing plastid status and are related to the ‘Chipper’ (Ch) standard Chloroplast (e.g., tCh signifies that an entity is tolerant and is genotypically similar to ‘Chipper’) according to Gordon and Staub (2011). Additionally, the annotation, - and --, is used to designate plastid and nuclear genotypic constitution, and, thus, a putative Chilling-tolerant accession is designated as t/ChCh, whereas a putative Chilling-susceptible accession is annotated as s/ChCh. In contrast, line NC-76 whiCh possesses a Chilling-tolerant nuclear genome is denoted as ChCh in a homozygous state. Uncertainty in classification is annotated by a ?.
Results
Chilling Response
In general, the response of seedlings to Chilling at 4°C (Experiments 1; mean DR of accessions taken collectively = 1.8) was more pronounced than at 1°C (Experiments 2 and 3; mean DR of accessions taken collectively = 1.1) (Table 1). For example, seedlings of ‘NacdongChungjang’ were more damaged when Challenged at 4°C [mean DR = 3.8 (Experiment 1)] than at 1°C [mean DR = 1.5 and 1.7 (Experiment 2 and Experiment 3, respectively)]. Likewise, the damage to ‘Dongguan’ seedlings at 1°C [mean DR = 1.2 and 1 (Experiment 2 and Experiment 3, respectively)] was less than at 4°C [mean DR = 2.8 (Experiment 1)]. Similarly, ‘Eunsungbaegdadagi’ seedlings were more affected by Chilling at 4°C [mean DR = 2.5 (Experiment 1)] than at 1°C [mean DR = 1.4 and 1 (Experiments 2 and 3, respectively)].
In Experiment 1, where the Chilling Challenge was 4°C, the seedling damage to plants of line NC76 was relatively low (mean DR = 1), whiCh was in concordance with reports by Kozik and Wehner (2008) (Table 1). In Experiment 1, Chilling damage in NC76 (mean DR = 1) and Korean cultivar JanghyungnaChap (mean DR = 1) seedlings was similar to U.S. ‘Chipper’ (mean DR = 1.1), SaeronChungjang (mean DR = 1.1), and ‘Hangangmatbaegdadagi’ (mean DR = 1.2). The rankings of these germplasms in Experiment 1 were generally the same as the US tolerant lines and therefore these must be considered as Chilling tolerant. In contrast, the Chilling response of Korean ‘NacdongChungjang’ (mean DR = 3.8), ‘Dongguan’ (mean DR = 2.8), ‘Eunsungbaegdadagi’ (mean DR = 2.5), ‘Sinjungpum’ (mean DR = 2.3), ‘White’ (mean DR = 2.2) and ‘Joeunbaegdadagi’ (mean DR = 2.1) were comparatively high, and these germplasms must be considered as Chilling susceptible. Plants of ‘NacdongChungjang’ were severely affected by Chilling at 4°C and must be considered the most susceptible germplasm evaluated.
In Experiment 2, line NC76 (mean DR = 1), ‘SaeronChungjang’ (mean DR =1), ‘GlorysamChug’ (mean DR = 1) and ‘Joeun-baegdadagi’ (mean DR = 1) and in Experiment 3, ‘Saeron-Chungjang’ (mean DR = 1), ‘Heungnongbaegdadagi’ (mean DR = 1), line NC76 (mean DR = 1), ‘Chipper’ (mean DR = 1), ‘Suiseifushinari 2-go (mean DR = 1), ‘Dongguan’ (mean DR = 1), ‘Hangangmatbaegdadagi’ (mean DR = 1), and ‘JanghyungnaChap’ (mean DR = 1) were relatively tolerant to Chilling at 1°C (Table 1). Data taken collectively over experiments indicate that ‘SaeronChungjang’, ‘Chipper’, line NC76, ‘Hangangmatbaegdadagi’, and ‘JanghyungnaChap’ were the most Chilling tolerant germplasms examined. In contrast, ‘NacdongChungjang’ was uniformly susceptible to Chilling at 1°C.
Genotype Analysis
Genotypic differences among the germplasms examined were detected by Chloroplast marker analysis. For instance, in this study, genotypic differences were detected between two US cucumbers (line NC76 and ‘Chipper’ (Chilling tolerant) and two Korean cultivars [‘NacdongChungjang’ (Chilling susceptible) and ‘JanghyungnaChap’ (Chilling tolerant)] (Table 1 and Fig. 2). Various Chilling tolerant and susceptible phenotypes were detected among all Korean, Japanese, and Chinese cucumbers tested in this study. However, these possessed same susceptible-associated banding morphotypes of Chilling as Chilling susceptible cucumber (i.e., ‘GY14’) as defined by Chung et al. (2007), Ali et al. (2013), and the results obtained in this study (Table 1).
Discussion
Chilling damage is irreversible when tomato (Solanum lycopersicum L., cv. Abunda) plants are Challenged at 6°C for several weeks under low light (60-100 µmol/quanta/m2/s) (Brüggemann et al., 1992). At suCh low Chilling tempera-tures, inhibition of Photosystem I (PSI) may be impaired due to interaction of active oxygen species with reduced electron acceptor in PSI (Sonoike, 1996a, 1996b). SuCh photo-inhibitive impairment of Photosystem I (PSI) can also be detected at Chilling (0-10°C), where plant damage is visible (Kudoh and Sonoike, 2002). In our study, cucumber seedlings exposed to Chilling temperatures (4°C) manifested more damage when compared to seedlings Challenged at 1°C (Table 1). Although this result is counter intuitive, it might be partially attributed to temperature-dependent metabolic disruptions in a plant’s photosynthetic apparatus (Brüggemann et al., 1992; Kudoh and Sonoike, 2002; Sonoike, 1996b), and/or to Chilling and post-Chilling environ-ments (Smeets and Wehner, 1997). Variation in plant response may occur when temperatures approaCh freezing (1-2°C) since photosynthetic systems and associated metabolic activities are often disrupted depending on genotype and Chilling environment (Hetherington and Öquist, 1988; Hetherington and Smillie, 1989). Impaired photosynthetic and metabolic systems can, however, be reconstituted to “normal” activity under elevated post-Chilling temperatures (> 20°C) (Hetherington and Öquist, 1988). Therefore, we suggest a hypothesis that photosynthetic metabolism in Chilling susceptible cucumber plants tested in this study could have been impaired following a short period of Chilling stress (e.g. 9 hours) with temperatures close to 0°C (i.e., 1°C). However, Chilling at higher temperatures (i.e. 4°C to 6°C) may not substantially disrupt photosynthetic metabolism, but could cause damage to the photosynthetic apparatus. High light intensity during Chilling could cause more severe Chilling damage when compared to little or no light during the Chilling (Sonoike, 1998). This may also provide a partial explanation for the observed relationship between Chilling damage level and plant reaction (i.e., photosynthetic meta-bolism) after Chilling Challenge. Thus, plants Chilled at 1-2°C temperatures may be less affected (i.e., lower visible damage ratings) when compared to Chilling at 4 to 6°C. This hypothesis is supported by the fact that Chilling response in cucumber is associated with Chilling temperature (intensity and duration) and light (intensity and duration) (Kozik and Wehner, 2008; Smeets and Wehner, 1997; Staub and Wehner, 1996).
Gordon and Staub (2011) found that, when subjected to a Chilling treatment of 5.5 hours (08:00 AM to 13:30 PM) at 4°C (immediate and constant at an irradiance of 270 μmoles・m-2・s-1 PPF), line NC76 demonstrated an intermediate Chilling response compared with ‘Chipper’. The Chilling response of reciprocal F1 progeny heterozygous for the nuclear Chilling resistance factor (Ch) contributed from line NC76 (paternal parent) was similar to their maternal (t/ChCh) and (s/ChCh) parent lines. Moreover, in re-introgression crossing, the re-introduction of the original plastid type (i.e., the s and t types) in F1 progeny restored the Chilling response to that equal to parental Chilling tolerant ‘Chipper’. However, in Gordon and Staub (2011) study, NC76 was not used as a maternal parent. Therefore, although Gordon and Staub assumed that NC76 possessed the s plastid type according to Kozik and Wehner (2008), its response to the Chilling conditions described by Chung et al. (2003b) and Gordon and Staub (2011) has not been investigated. These and other results led them to conclude that under those Chilling conditions, that: 1) line NC76 and ‘Chipper’ were genotypically ?/ChCh and t/ChCh, respectively; 2) tolerant individuals t/ChCh (i.e., ‘Chipper’) would not be Chilling tolerant under the experimental conditions described by Kozik and Wehner (2008); 3) the maternal plastid contribution is more predictive than the nuclear contribution in determining a plant’s Chilling phenotype, and; 4) the effect of Ch on Chilling response is negligible in cross- progeny under the Chilling conditions applied by Gordon and Staub (2011).
The 12 Korean and two US, one Japanese, and one Chinese cucumber germplasms demonstrated differing Chilling responses to the Chilling temperatures (1 and 4°C) imposed herein (Table 1). In this study, although Chilling response varied widely, some Korean market type cucumber germ-plasms were classified as Chilling susceptible (‘Nacdong-Chungjang’, ‘Eunsungbaegdadagi’, ‘Sinjungpum’, and ‘White’) and tolerant (‘SaeronChungjang’, ‘Hangangmatbaegdadagi’, and ‘JanghyungnaChap’) based on Chilling injury ratings (Table 1).
The role of the Chloroplast CSatpB-SNP and CSycf1-SNP marker loci associated with genetic control of Chilling injury in cucumber (Ali et al., 2013; Chung et al., 2007) in regulating specific bioChemical pathways has not been defined. Never-theless, their phenotypic responses were generally typical of that of the historic tolerant Chilling controls, U.S. processing type ‘Chipper’ and line NC76 (Chung et al., 2007; Gordon and Staub, 2011).
All Chilling tolerant Korean cucumbers examined in this study did not possess marker-Chilling associated Chloroplast genotypes related to Chilling tolerance (Table 1). Thus, these phenotypically Chilling tolerant cucumbers may possess nuclear (e.g., Ch as in NC76; Kozik and Wehner, 2008) and/or unique maternal (i.e., other than the two identified in ‘Chipper’; Chung et al., 2003b) factors that condition Chilling tolerance. Given previous reports (Chung et al., 2003b; Gordon and Staub, 2011; Kozik and Wehner, 2008), the Chilling genotypes of susceptible and tolerant germplasms are provisionally designated as s/ChCh and t/ChCh or s/Ch_, respectively. These hypothesized genotypic designations can be confirmed by examination of cross-progeny derived from controlled crossing between Chilling susceptible and tolerant accessions (Gordon and Staub, 2011). Comparative analysis of Chilling response in reciprocal backcross cucumber lines derived from Chilling susceptible and tolerant lines can be used to access both plastid and nuclear factors (Gordon and Staub, 2011). The Chilling response in cross- progeny persists and remains constant over multiple backcross generations in whiCh increasing dosages of paternal nuclear alleles were introduced into the progeny. A determination of the mode of inheritance to Chilling injury in Korean cucumber could be accomplished by comparing the Chilling response of reciprocal F1, F2,and/or BC2 progeny (e.g., reciprocal ‘NacdongChungjang’ × ‘JanghyungnaChap’ matings) as demonstrated by Gordon and Staub (2011).
Understanding the response of cucumber seedlings to various Chilling temperatures (Chung et al., 2003b ; Kozik and Wehner, 2008) and the relationship of nuclear and plastid factors conditioning Chilling tolerance (Gordon and Staub, 2011) is important when designing breeding strategies for the development of seedling tolerance to Chilling. For instance, the effect of Ch on Chilling response in cucumber germplasm is negligible when seedlings are Challenged at 4°C under the Chilling regime used by Gordon and Staub (2011; see above). The Ch nuclear factor may, however, be important in Chilling tolerance at other Chilling conditions (Kozik and Wehner, 2008). Under a Chilling Challenge of 4°C and 8 hours as examined by Chung et al. (2003b), tolerant plastid factors presumably would provide adequate protection to cucumber plants in the seedling stage under field conditions (Chung et al., 2003b; Gordon and Staub, 2011; data presented herein). Traditional backcrossing might be further augmented by the deployment of marker-assisted selection during backcrossing. Simple, yet effective, marker- assisted backcross introgression strategies may be effective for the introduction of plastomes and nuclear factors using the Chloroplast markers identified by Chung et al. (2003b) and Ali et al. (2013). SuCh strategies have been effective in the introgression of yield and quality component traits in cucumber (Fazio et al., 2003; Fan et al., 2006).
Conclusion
We have provided herein the comparative analysis of the Chilling response of Korean cucumber varieties and previously documented Chilling tolerant U.S. cucumber lines. The Chloroplast identity (susceptible vs. tolerant) of these cultivars was also Characterized through SNP-marker geno-typing, whiCh could be beneficial for breeding Chilling tolerant cucumber varieties.