Introduction
Material and Methods
Plant Materials and Acclimation Conditions
Development of Nutrient Solution
Treatment
Measurement
Data analysis
Results
Survival Rate according to EC Level of Nutrient Solution
Growth Characteristics according to Nutrient Solution and EC Level
Change of EC Level of Nutrient Solution by Acclimation Stage
Mineral Contents according to Nutrient Solution and EC Level
Discussion
Conclusion
Introduction
Dwarf apple rootstocks ‘M9’ have a long juvenile period and self-incompatibility, and are typically produced by vegetative propagation methods, such as mound layering (Kim et al., 1998; Lim et al., 2015). However, the apple plantlets produced by vegetative propagation methods are easily infected with viruses from mother plants. Viruses such as Apple mosaic virus (ApMV), Apple stem grooving virus (ASGV), Apple stem pitting virus (ASPV), and Apple chlorotic leaf spot virus (ACLSV) can
reduce fruit yields from 9% to 67% and are a major problem for orchards (Cembali et al., 2003). Meijneke et al. (1973) reported that apple trees not infected with an apple virus showed improved growth (19-23%), fruit quality (11%), and fruit yield (31%). Kim et al. (1998) also reported that tree vigor of virus-infected ‘M9’ apple trees was only 30% of that of virus-free ‘M9’. For these reasons, demand for virus-free apple plantlets has recently increased and studies on their production have been increasing globally. Particularly, in vitro culture methods using an apical virus-free meristem has been used to investigate virus-free apple plantlet production (Walkey and Webb, 1968; Theiler-Hedtrich and Baumann, 1989; Chatenet et al., 2001).
Compared with normal ex vitro conditions, the in vitro environment has unique conditions such as high relative humidity, low CO2 concentration, low light intensity, and photomixotrophic cultivation provided by sugar-supplemented medium. Plants cultivated in these benign environmental conditions must go through an acclimation period to adapt to stressful ex vitro conditions (Kozai, 1991; Kadleček et al., 2001; Hazarika, 2006; Pospíšilová et al., 2007). In particular, water-related environmental factors heavily influence in vitro apple plantlets (IAP) survival. Apple plantlets propagated in vitro with RH close to 100% have underdeveloped stomatal function and exposure to an ex vitro environment with relatively low RH can result in severe water loss through excessive transpiration (Shackel et al., 1990; Díaz-Pérez et al., 1995). Moreover, although water lost by transpiration could be supplemented by water absorption from the roots (Jackson et al., 2000), IAP typically have poor initial root growth (James and Thurbon, 1979) and the poorly-formed vascular connection between the shoot and root (Díaz-Pérez et al., 1995; Hazarika, 2003) make water transport difficult for these plantlets. A lack of defense mechanisms against water stress is another problem (Dixon and Paiva, 1995; Sgherri and Navari-Izzo, 1995; Blokhina et al., 2003; Mittler, 2002; Oh et al., 2009), which decreases plantlet survival rate. Therefore, we have conducted a previous study to improve the acclimation rate of IAP through the precise control of environmental factors such as RH, temperature, and volumetric water contents of rhizosphere in a closed-type plant production system (Ko et al., 2018).
Closed-type plant production systems like plant factories adopt hydroponic methods for plant cultivation, where water and essential minerals are supplied to plants through nutrient solution. The minerals dissolved in the water are absorbed through various transport proteins located in root cell membranes. The absorption process is divided into passive transport, which is facilitated by the concentration gradient, and active transport, which occurs against the concentration gradient using energy from ATP hydrolysis. Passive transport is possible if the mineral concentration in the nutrient solution is high, but if not, it needs to be absorbed through active transport (Taiz and Zeiger, 2006). Steiner (1980) suggested that each plant species has a specific mineral absorption rate, and if we supply minerals to that ratio, the conserved energy that is not used for mineral absorption will have a positive impact on growth. In other words, when the minerals are outside an optimum concentration, plants can divert energy from absorbing the desired minerals to growth processes. This concept has been already demonstrated in our previous study using Crepidiastrum denticulatum (Park et al., 2016). Meanwhile, excessively high mineral concentration in a nutrient solution would decrease the water potential and impair water uptake into the roots, which would further deteriorate the water status of IAP (Romero-Aranda et al., 2001). Jacobs and Timmer (2005) suggested that dramatic changes in pH, EC, and ion availability for outplanted forest tree seedlings may inhibit root growth and function by causing specific ion toxicities, as well as decrease the water potential. EC sensitivity depends on the plant species, age of the root system, and soil moisture availability. Thus, the proper EC level should be maintained to optimize a seedling’s nutrient availability and minimize root damage. In the hydroponic systems of a closed-type plant production system, the environment is easy to control and the nutrient solution containing the optimum mineral absorption rate for IAP can be easily administered, improving their growth and acclimation. However, there is currently no study for the development of a nutrient solution specific to apple plantlets.
Therefore, this study was conducted to develop a NAP based on the optimum mineral absorption rate for apple plantlets and to determine the proper concentration of nutrient solution for promoting not only stable plantlet acclimation, but also growth in a closed-type plant production system.
Materials and Methods
Plant Materials and Acclimation Conditions
Virus-free apple plantlets (dwarf apple rootstock ‘M9’) were propagated by apical meristem culture in in vitro conditions with a Murashige and Skoog (MS) medium containing 0.5 mg·L-1 BA, 30 g·L-1 sucrose, and 8.4 g·L-1 agar for ten weeks. Subsequently, they were plated on rooting MS medium (supplemented with 0.5 mg·L-1 IBA, 30 g·L-1 sucrose, and 8.4 g·L-1 agar) for ten weeks in 620-mL bottles with a ventilation filter equipped with a rubber cap (diameter 40 mm). The culture bottles containing the IAP were placed on shelves of two airtight growth chambers (DS-52G3P; DS-51GLP, Dasol Scientific, Hwaseong, Korea), and the RH was maintained at 90% by partially opening the rubber caps. This stage was the pre-acclimation (PA) stage and lasted for one week before transplanting the plantlets to ex vitro conditions. After the PA process, the IAP were washed with tap water to remove the medium fragments and were transplanted to deep flow technique (DFT) systems, which were most effective for IAP acclimation and growth in our previous study (Ko et al., 2018). The DFT system was supplied with sufficient air through a ceramic air diffuser (diameter 150 mm). The environmental conditions of the growth chambers during the six-week acclimation period were as follows: air temperature, 25°C; photosynthetic photon flux density (PPFD), 60 µmol·m-2·s-1, irradiated by fluorescent lamps; light period of 16 h; RH, decreased stepwise from 90% to 80%, and 60% at two-week intervals.
Development of Nutrient Solution
To develop the NAP, the plantlets acclimated under previously described conditions were cultured in Hoagland nutrient solution (HN, pH 6.0, EC 1.2 dS·m-1) (Hoagland and Arnon, 1950) for six weeks. For mineral analysis, healthy apple plantlet shoots were collected at 6 weeks of transplanting and the shoots were dried for 72 h at 70°C in an oven (FS-420, Advantec Co., Tokyo, Japan). Six whole-dried apple plantlet shoots were finely ground simultaneously using a mill (Tube Mill control, IKA, Wilmington, NC, USA), and about 1 g was taken four times from each sample.
To analyze all the mineral content except N, the dried samples were digested using a wet digestion method as described by Havlin and Soltanpour (1980) with a minor modification. Each sample was digested with 20 mL of 70% nitric acid (HNO3) at 125°C. H2O2 was added to the nitric-perchloric digests, and the sample was heated at 200°C on a heating block (OD-98-002P, ODlab, Seoul, Korea). After cooling for 24 h, the solution was adjusted to 110 mL by adding triple distilled water and filtered through 125 mm quantitative filter paper (Quantitative ashless, Hyndai Micro, Seoul, Korea). The contents of K, S, P, Ca, Mg, Cu, Fe, Zn and Mn were measured with an ICP-OES spectrophotometer (Optima 7300 DV, Perkin Elmer, Waltham, MA, USA).
The total N contents in the apple plantlet shoots was analyzed by the Kjeldahl method (Kjeldahl, 1883; Martre et al., 2003) with a minor modification using a total N distillation apparatus (TKN-EW1080, DSlab, Seoul, Korea). About 0.5 g of the finely ground apple plantlet shoots, 10 g of CuSO4-K2SO4, and 50 mL of H2SO4 were added into the Kjeldahl flask, and the solution was heated at 180°C for about 30 min on the heating block. After the solution became a clear blue color, it was heated at 200°C for about 40 min. The samples were cooled and the volume made up to 200 mL with triple distilled water in a volumetric flask. The decomposed solution (10 mL) was distilled with 50 mL of 45% NaOH in the Kjeldahl flask and the end of the cooling tube was immersed in an Erlenmeyer flask containing 50 mL of 2% boric acid solution with an indicator. The Kjeldahl flask was heated until the Erlenmeyer flask was filled up to 70 mL, and then titrated with 0.05 N H2SO4.
Based on the cation and anion (Fig. 1A) ratios obtained from the mineral analysis, NAP was developed for apple plantlet (Table 1). NAP contained a sufficient amount of micronutrients (Cu, Fe, Zn, Mn, B and Mo) that ordinary plants need (data not shown).
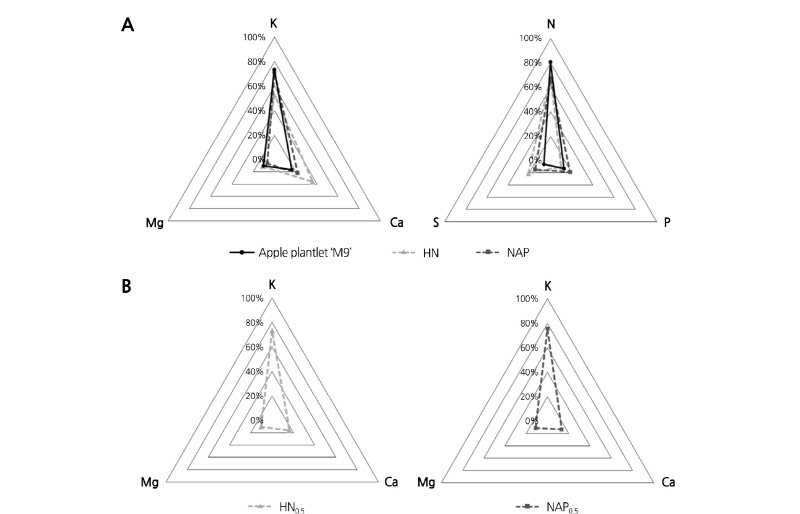
Fig. 1. Cation (K, Mg, Ca) and anion (N, P, S) compositions of in vitro propagated apple ‘M9’ plantlets, Hoagland nutrient solution (HN), and nutrient solution for apple plantlets (NAP) (A) and cation composition of in vitro apple plantlets (IAP) cultured in HN and NAP with EC 0.5 dS·m-1 at 6 weeks after transplanting (B).
Table 1. The macro-element compositions of HN and NAP
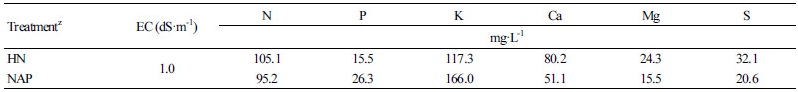 | |
zHN: Hoagland nutrient solution, NAP: nutrient solution for apple plantlets. | |
Treatment
We conducted an experiment to verify IAP survival rate according to concentration of HN (EC: 1.0, 2.0 and 3.0 dS·m-1) and NAP (EC: 0.5, 1.0, 2.0 and 3.0 dS·m-1). Based on this result, we treated both nutrient solutions with three concentrations (EC: 0.5, 1.0, and 2.0 dS·m-1) to determine the proper EC level in another experiment. According to our previous study, IAP started growing at two weeks after transplanting and the growth rate rapidly increased after four weeks (Ko et al., 2018). Thus, two types of EC level controls were additionally applied to investigate the response to change in the rhizosphere EC level according to acclimation stages. Some of the NAP treatments with EC level of 0.5 dS·m-1 were increased to 2.0 dS·m-1 at two (NAPex2) and four weeks (NAPex4) of transplanting each. The pH of all nutrient solutions was 6.0. The pH and EC were measured using a pH and EC meter (Multi 3430 SET F, WTW, Weilheim, Bavaria, Germany). The nutrient solutions were replaced every two weeks.
Measurement
Survival rate
Survival rate was represented as the cumulative percentage of survived IAP after transplanting to ex vitro for six weeks.
Growth characteristics
Stem and root lengths were measured by digital Vernier calipers (NA530-300S; Bluebird, Seoul, Korea). Fresh shoot and root weights were measured using an electronic scale (SI-234; Denver Instrument, Denver, CO, USA). The samples were subsequently dried in an oven (VS-1202D3; Vision Scientific, Daejeon, Korea) at 70°C for 72 h for measuring dry matter. Total leaf area was measured using a leaf area meter (LI-3100C; LI-COR, Lincoln, NE, USA). Roots were scanned by a scanner (HP Officejet Pro 8610, HP Development Company, Palo Alto, CA, USA) for root surface area, which was estimated by a Java-based image processing program (ImageJ, National Institutes of Health, Bethesda, MD, USA).
Mineral contents
Mineral contents of the IAP shoots were analyzed to verify the effects of mineral composition and EC level of HN and NAP on their growth at 6 weeks of transplanting. Nine plant essential elements (K, S, P, Ca, Mg, Cu, Fe, Zn and Mn) were measured with the same method used for NAP development as described above.
Data analysis
The estimates of each parameter were based on five replications per treatment. An analysis of variance (ANOVA) was performed, and treatment means were compared using Duncan’s Multiple Range Test (DMRT). All statistical analyses were performed with the SAS program (SAS 9.4; SAS Institute, Cary, NC, USA).
Results
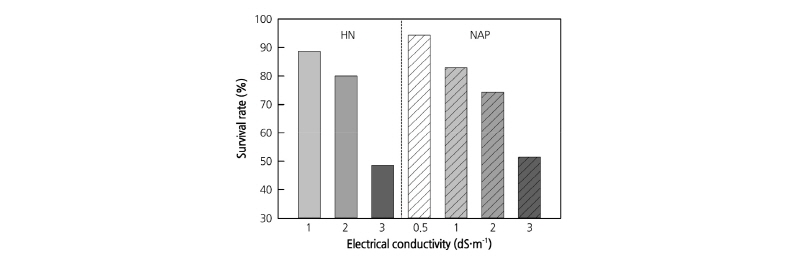
Survival Rate according to EC Level of Nutrient Solution
The survival rates tended to decrease with increasing EC level in both HN and NAP at six weeks after transplanting (Fig. 2). Survival rate of NAP treatment with EC level 0.5 dS·m-1 was the highest at 94.3%, while EC level 3.0 dS·m-1 of HN and NAP showed the lowest values of 48.6% and 51.4%, respectively. Based on this result, the concentrations of the nutrient solutions in the experiment to determine proper EC level were set at 0.5, 1.0 and 2.0 dS·m-1.
Growth Characteristics according to Nutrient Solution and EC Level
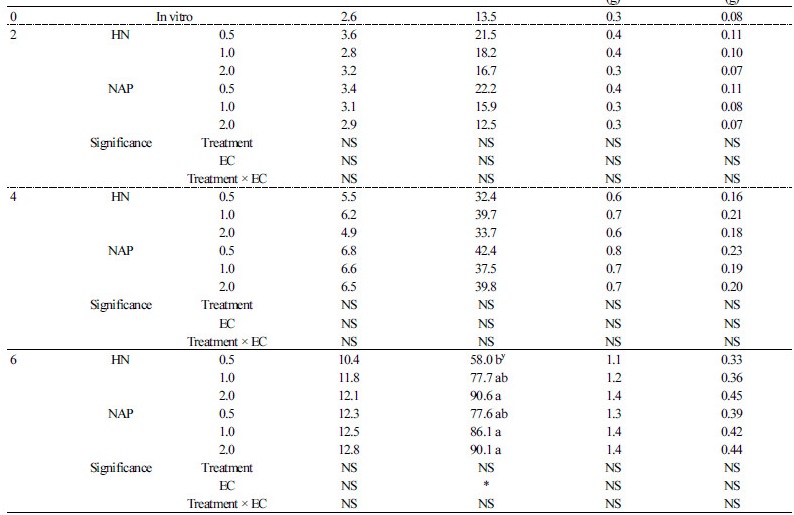
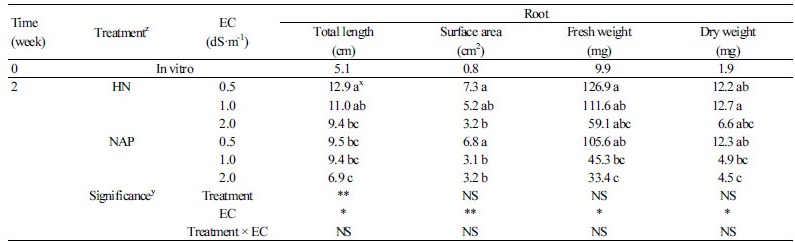
There were no significant differences in all shoot growth parameters except the total leaf area at six weeks after transplanting, but the tendency was different according to the EC level in each growth stage (Table 2). At two weeks of transplanting, both the HN and NAP treatment tended to have a high numerical value for all shoot growth at a low concentration of EC 0.5 dS·m-1. At four weeks after transplanting, the shoot growth of HN and NAP treatment were numerically high for EC 1.0 and EC 0.5 dS·m-1, respectively. However, a relatively low concentration of nutrient solution had a positive effect on the shoot growth until four weeks after transplanting. All the shoot growth parameters of EC 2.0 dS·m-1 treatment tended to be high at six weeks after transplanting and total leaf area was significantly higher in EC 2.0 dS·m-1. Root growth showed a marked difference compared to shoot growth at all growth stages (Table 3). Similar to shoot growth, all the root growth parameters at two weeks after transplanting were significantly high at the low concentration of EC 0.5 dS·m-1 in both nutrient solution treatments. On the other hand, although the root length of HN was significantly higher than that of NAP at two weeks after transplanting, there were no significant differences in all the other root growth parameters. There were no significant differences according to EC at four weeks after transplanting, but it showed the lowest numerical value at the low concentration of EC 0.5 dS·m-1, contrary to the tendency observed at the initial stage for two weeks. Root surface area and root dry weight of NAP at four weeks after transplanting were significantly higher than those of HN. Furthermore, all the root growth parameters of NAP at six weeks after transplanting were significantly higher than those of HN. Surface area and fresh and dry weight of roots with NAP0.5 at six weeks of transplanting were 1.8, 2.7, and 2.7 times higher than HN0.5, respectively. We could see a clear difference in root growth between the two treatments (Fig. 3A). On the other hand, the root growth of HN and NAP in six weeks after transplanting had different tendencies according to EC level. At six weeks after transplanting, the root growth of HN showed a tendency to increase as the EC level increased similar to shoot growth, whereas the root growth of NAP tended to decrease with increasing EC level.
Change of EC Level of Nutrient Solution by Acclimation Stage
The growth rates from 2-4 weeks and 4-6 weeks after transplanting were measured to confirm the change of growth speed according to the increasing EC level at each acclimation stage (Fig. 4). The growth rates of total leaf area, root surface area, shoot dry weight, and root dry weight between two-four weeks of NAPex2 were higher than that of NAPex4, which did not change the EC level. Like NAPex2, all the growth rates between four-six weeks of NAPex4 also showed increasing tendency when EC level was increased, and they were 2.1, 2.2, 2.1, and 3.0 times higher than growth rates between two-four weeks of NAPex2, respectively. However, at 4 weeks after transplanting, the growth rates of NAPex2 was significantly lower, and all the growth parameters of NAPex4 were significantly higher than that of NAPex2. Fig. 3B shows a marked difference between shoot and root growth.
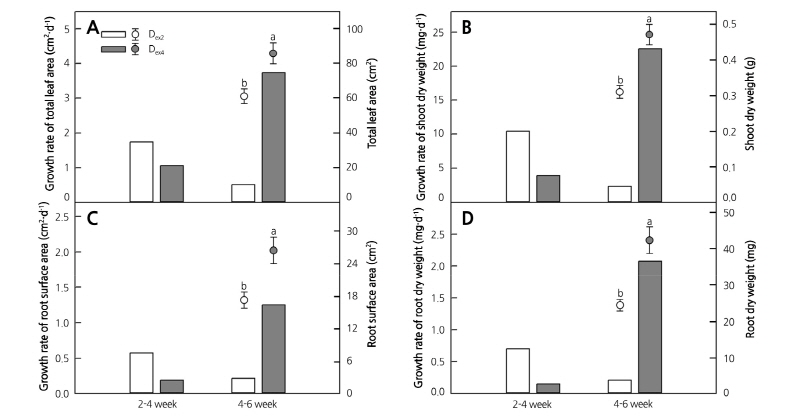
Fig. 4. Growth rate (bars) of total leaf area (A), shoot dry weight (B), root surface area (C), and root dry weight (D) and those values at 6 weeks after transplanting (circles) of in vitro-propagated apple plantlets according to EC change of nutrient solution for apple plantlets (NAP) from 0.5 dS·m-1 to 2.0 dS·m-1 at 2 (NAPex2) and 4 (NAPex4) weeks after transplanting each. Different letters indicate a significant difference at p ‹ 0.01 (n = 8).
Mineral Contents according to Nutrient Solution and EC Level
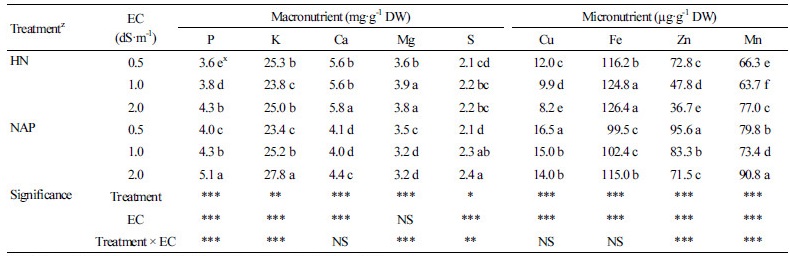
Mineral contents of shoots of IAP acclimated for six weeks showed significant difference according to mineral composition and concentration of nutrient solutions (Table 4). Ca and Mg contents were significantly high in IAP with HN treatment, which contained a high ratio of Ca and Mg. Similarly, P and K contents were significantly high in IAP with the NAP treatment, which contained a high ratio of P and K. Although there was some difference in the mineral composition between the two treatments, the cation ratio remained almost constant. The cation ratio differences of K, Ca, and Mg contents in IAP with HN or NAP were only 2.3%, 2.9%, and 0.8%, respectively (Fig. 1B). However, the cation ratio differences of the mineral contents between the two nutrient solutions were 18.5%, 14.1%, and 4.3%, respectively (Fig. 1A). In addition, the cation ratios of both treatments are almost the same with that of IAP measured for NAP development (Fig. 1). P and Ca tended to increase with increasing EC level in both nutrient solutions. K and S showed this tendency only in NAP treatment and Mg did not show any tendency according to EC level. On the other hand, all the micro elements except Fe were significantly higher in NAP than HN. Cu and Zn showed the highest contents in the nutrient solution of EC 0.5 dS·m-1; conversely, Fe and Mn showed the highest contents in the nutrient solution of EC 2.0 dS·m-1.
Discussion
We have developed a NAP that is adapted to the mineral absorption rate of IAP. The NAP was compared with HN at several EC levels to evaluate its effects for IAP acclimation and growth and to determine a proper EC level. The two nutrient solutions of HN and NAP did not make any significant difference in the shoot growth of IAP during six weeks of acclimation, but the root growth of IAP was significantly increased by NAP at four weeks after transplanting. These results suggest that the NAP treatment, which is adjusted to the mineral absorption rate of IAP, would facilitate the mineral absorption through root passive transport proteins, and more energy could be supplied directly into IAP root growth instead of active transportation (Steiner, 1980; Taiz and Zeiger, 2006). In addition, the weak roots of IAP require time to adapt to chemical environment changes, such as EC, pH, and ion availability of the rhizosphere. This aspect of mineral composition for NAP would have a positive effect on the root growth by contributing to the acclimation of the rhizosphere environment (Jacobs and Timmer, 2005). In our previous study, we found that IAP exposed to ex vitro conditions inevitably suffered water stress due to their anatomical and physiological abnormalities. Furthermore, the root growth tends to start earlier than the shoot growth under the environmental conditions, causing excessive water stress at the initial acclimation stage (Ko et al., 2018). Typically, plant shoots respond more sensitively to drought than the roots (Sharp and Davies, 1989). IAP shoots were also reported to be inhibited by root-to-shoot signaling in drought stress conditions (Gowing et al., 1990). These findings suggest that the IAP, which are under water stress at the initial acclimation stage, improved water absorption by preferentially growing the root part rather than the shoot part, where transpiration occurs. This may support the evidence that the effects of NAP treatment on growth promotion was more prominent in root growth than shoot growth.
At the initial acclimation stage of two weeks after transplanting, nutrient solution with a low EC level showed positive effects on IAP shoot and root growth. The IAP have weak roots and a poorly formed vascular connection between the shoot and root at the initial acclimation stage; therefore, they have difficulty in water uptake (James and Thurbon, 1979; Díaz-Pérez et al., 1995; Hazarika, 2003). Furthermore, water lost by transpiration cannot be supplemented by water absorption through their roots (Jackson et al., 2000). That is, nutrient solution with a high EC concentration at the initial acclimation stage decreases the water potential in the rhizosphere and makes water absorption difficult, not only hurting plant growth, but also survival rate (Romero-Aranda et al., 2001; Jacobs and Timmer, 2005). After the initial acclimation stage, the root growth in HN treatments tended to increase with increasing EC levels at six weeks after transplanting, which might be the result of absorbing more minerals after adapting to the high EC levels. In contrast, NAP showed a tendency to decrease with increasing EC. It is thought that different mineral composition might be the reason for this, although further study is necessary to confirm this hypothesis.
In our previous study, the growth of IAP started at two weeks after transplanting and then rapidly increased after four weeks (Ko et al., 2018). Thus, we increased the EC level of NAP from 0.5 dS·m-1 to 2.0 dS·m-1 at two weeks (NAPex2) and four weeks (NAPex4) to investigate IAP’s response to EC level changes at specific acclimation stages. The growth rate of both NAPex2 and NAPex4 tended to increase after increasing EC levels, and NAPex4 showed greater increase compared to NAPex2. In addition, the growth rates from four to six weeks of NAPex2 was significantly decreased, resulting in significantly higher values in all the growth parameters of NAPex4 compared to NAPex2. The leaf water potential of IAP under EC 1.0 dS·m-1 of HN tended to decrease until three weeks after transplanting (Ko et al., 2018), which implies that the IAP were under water stress. Environmental changes, such as the rapid changes in the rhizosphere EC level during the initial three weeks after transplanting, can cause intense drought stress and inhibit IAP growth (Jacobs and Timmer, 2005). In particular, woody plants and forest tree seedlings are known to be sensitive to high EC levels and damage can quickly occur (Landis et al., 1989; Maynard et al. 1997). Therefore, increasing EC levels should be performed after at least four weeks after transplanting.
The mineral content analysis on IAP acclimated for six weeks in both HN and NAP showed the mineral absorption rate from both nutrient solutions was maintained to some extent (Fig. 1B), although there were significant differences in the absorbed content for several minerals. For mineral uptake and utilization, the ability of the plant to absorb minerals is more crucial than the mineral content of the soil, so long as the soil does not have a specific mineral deficiency (Collander, 1941; Dykyjová, 1979; Steiner, 1980). IAP cultivated under both HN and NAP transported all the minerals that we analyzed in this study with a balanced rate, resulting in no significant difference in shoot growth between treatments. Although we did not examine N content of IAP at six weeks after transplanting, no significant increase in shoot growth resulted from HN with higher N content than NAP, implying IAP absorbed N equally, regardless of the nutrient solution treatment. In contrast, the significantly higher root growth in NAP compared to HN indicated that NAP improved the absorption rate of IAP and stimulated root growth. These results support that the mineral composition in NAP allows the plant to use passive transport instead of active transport, letting it conserve energy and improve root growth (Steiner, 1980). On the other hand, P positively affects root development (Römer and Schilling, 1986; Chabot et al., 1998) and K is known to elongate the root apex by increasing intercellular osmotic potential (Taiz and Zeiger, 2006). Although the root growth promoting effect may be a result of the high P and K ratio of NAP, the difference in the absorbance of these minerals is not large, and efficient mineral absorption from NAP seems to be more influential.
Conclusion
We have conducted studies to improve the acclimation of IAP through environmental control in closed-type plant production systems where plant essential minerals must be artificially supplied with nutrient solution. Until now, there has been no study to develop a nutrient solution for apple plantlets that improve its acclimation.
Growth and mineral contents of NAP treatments were compared with HN at several EC levels to confirm the effects of the NAP and to determine the proper EC concentration. In summary, the low concentration of nutrient solution improved IAP growth at the initial stage of acclimation and a high concentration of nutrient solution began to show a positive effect on IAP growth in the later acclimation stage. In addition, root growth was significantly promoted by NAP at four weeks after transplanting. Therefore, if NAP is supplied at the proper EC level according to the acclimation stage, it will alleviate their poor root development and promote acclimation. In conclusion, our research has developed a unique nutrient solution for fruit trees such as apple plantlets and will help the acclimation and growth of in vitro-propagated plantlets.