Introduction
The Huangguan pear (Pyrus pyrifolia Nakai) is widely grown in southern China and is an important fruit throughout the country. The Huangguan pear is popular for its creamy flesh and sweet and fragrant flavor; however, the fruit quality deteriorates rapidly when improperly stored. Pear quality depends largely on the soluble sugar components of the fruit. Under improper storage conditions, the sugar and acid contents of the fruits decrease, resulting in an undesirable sugar/acid ratio with a worsened taste. The quality and shelf life of vegetables and fruits are affected by many factors, including enzymatic browning, texture deterioration, loss of sugars and water, microbial growth, and senescence processes (Sathe et al., 2007). Fruits undergo a series of biochemical and physiological changes after harvest,which accelerate the senescence process and lead to color change, flavor deterioration, softening, and weight loss (Nunez- Gastelum et al., 2015; Yoo et al., 2016). Cold temperatures and treatments with edible coatings are widely used to slow the senescence processes and increase the postharvest shelf life of stored fruit for many species (Brummell, 2006; Lara et al., 2004).
Edible coatings are increasingly popular because of environmental considerations and trends toward the use of convenience foods (Ozden and Bayindirli, 2002). A variety of coating treatments are used, including calcium compounds, polysaccharides and proteins (Alzamora et al., 2005; Mihindukulasuriya and Lim, 2014; Pierro et al., 2007). Calcium, which is known for its ability to reduce or delay parasitic and/or physiological disorders in fruits and vegetables, shows promise as a treatment for maintaining product firmness and/or controlling storage rot when applied as either organic or inorganic salts (Alzamora et al., 2005). Postharvest calcium applications maintain cell turgor and tissue firmness, and delay membrane lipid catabolism, extending the storage life of fresh fruits (Luna-Guzman and Barrett, 2000; Picchioni et al., 1998). In a series of studies on leaf abscission and tissue senescence, Aghdam et al. (2012) and Wang et al. (2011) reported that Ca2+ treatments inhibited or slowed these processes. Based on these findings, we previously conducted a series of studies that revealed that calcium could remarkably inhibit the occurrence of brown spots and reduce the incidence of spoilage in Huangguan pears (Kou et al., 2015).
Polysaccharide-based coatings are expected to be good oxygen barriers due to their tightly packed and ordered hydrogenbonded network structure (Yang and Paulson, 2000). Pullulan, an extracellular, nontoxic, edible biopolysaccharide produced by Aureobasidium pullulans, has numerous applications in food (Leathers, 2003). In recent decades, more attention has been paid to pullulan, including its physical, chemical, and film-forming properties, such as ductility, resistance to air, its ability to reduce water loss and respiration rates, and its capacity to delay the maturation of fruit (Wu et al., 2009). Pullulan has been successfully used as a hydro-colloid coating for strawberries (Trevino-Garza et al., 2015). Pullulan-based coating treatments can effectively maintain high levels of CO2 within the fruit while substantially reducing internal O2 levels, and can retard enzymatic browning, maintain firmness, decrease weight loss, inhibit microbial growth and restrict the respiration rate of the fruit during cold storage (Wu et al., 2009; Wu and Chen, 2013), thus extending the shelf-life of the fruit. Previous research on the application of pullulan in pears focused mainly on its prevention of postharvest disease and its effects on other physiological indexes during storage (Kou et al., 2013; Kou et al., 2015; Robiglio et al., 2011); however, to date, there are few studies into its effect on sugar metabolism in fruit.
Sucrose, glucose and fructose are synthesized in photosynthetic source tissues and are among the most important sugars in fruits. Sucrose is synthesized under catalysis by sucrose synthase (SS; Enzyme Commission number: EC 2.4.1.13) or sucrose phosphate synthase (EC 2.4.1.14), and can be degraded either by SS or neutral or acid invertase (EC 3.2.1.26) (Li et al., 2012), which convert sucrose into glucose and fructose. AIV is located in the vacuole or cell wall and plays an important role in the hydrolysis of sucrose (Hatch and Glasziou, 1963). SS reversibly catalyzes sucrose synthesis and degradation; although this enzyme catalyzes a freely reversible reaction, with reported △G values ranging from -1.4 to -4.7 kJ·mol-1 for the sucrose synthesis reaction (Geigenberger and Stitt, 1993), its main physiological function is thought to be the cleavage of sucrose in sink organs to supply carbohydrates for the generation of uridine diphosphate glucose (UDPG) and the possible storage of energy via the uridine diphosphate (UDP) activation of the hexose moiety (Verma et al., 2011). In many species, SS was also found to modulate the sink strength of plants (Carlson et al., 2002; Moscatello et al., 2011).
Several fruits have been used to study the effects of edible coatings on these ripening characteristics (Aghdam et al., 2012;Hanani et al., 2012; Raese and Drake, 2000); however, their effects have not been reported for the Huangguan pear. Two coatings, calcium chloride and pullulan, were therefore evaluated for their impacts on sucrose metabolism and the activity of related enzymes, as well as the additional regulation of AIV gene expression during long-term cold storage, in order to determine whether these factors could result in better postharvest handling for Huangguan pears.
Materials and Methods
Plant materials and postharvest treatments
‘Huangguan pear’ (Pyrus pyrifolia Nakai) fruits were harvested in the Shenyuwang orchard in Gao Cheng city, He Bei province, China, and transported to the laboratory within 2 h. The pears were selected on the basis of uniform color, size, and hardness, and were free of any visible physical defects or fungal infections. They were then randomly divided into three groups, with 300 fruits in each group. The first group (control) was immersed in deionized H2O. The second and third groups of fruit were immersed in 2% CaCl2 for 15 min or 1% pullulan for 5 min, respectively, and then naturally air-dried for 24 h at 20°C (Kou et al., 2013). The fruits were individually wrapped in polyethylene foam nets and packed into cartons with ventilation holes, before being subjected to cold storage at 0°C with approximately 95% relative humidity (RH). The fruits were stored for seven months, with random subsamples of each group taken monthly.
Whole fruits were used for the respiration test. For other chemical and physiological measurements, an approximately 2-cm-thick section of flesh tissue was removed using a razor blade and peeled. All samples were frozen with liquid nitrogen and stored at -80°C for further analysis.
Determination of firmness, TA and TSS
Measurements of flesh firmness (expressed in kg·cm-2) were carried out on the peeled fruit using a Fruit Pressure Tester (GY-1, Hangzhou Huier Instrument equipment co., Zhejiang, China) with an 8-mm probe. The flesh was liquidized and filtered, then the TA, expressed as a percentage of malic acid, was measured using a 0.1 M sodium hydroxide solution with phenolphthalein as an indicator. TSS was measured using a VR-113 handheld refractometer (Beijing Vision Photoelectric Technology Co., LTD, China). Each experiment was repeated at least three times.
Measurement of respiration rate
The respiration rate was measured in a controlled environment at 20°C and 80-85% RH using the closed system method described by Caled et al. (2012), with a slight adjustment. The respiration rate was measured by the amount of CO2 evolved by the fruits per unit of fresh weight (FW). Ten fruits from each treatment were enclosed in the chamber for 30 min with a gas flow rate of 0.5 L·min-1. The effluent air was connected to a GXH-3051 Infrared Gas Analyser (Institute of Junfang Scientific Instrument; Beijing, China), and %CO2 was detected. The respiration rate was calculated according to the formula:
where V, C, W and T are the gas flow rate (0.5 L·min−1), %CO2, fruit weight (kg) and temperature (°C), respectively. The respiration rate was expressed as mg CO2·h−1·kg−1 FW.
Determination of sugars
The composition of sugars in the fruit was determined according to the method described by Lopez-Gomez et al. (1992), with some modifications. Briefly, seven to ten fruits were sampled for each group. The flesh (4 g) was pulverized in a mortar under liquid N2, diluted in 20 mL of ultrapure water, then filtered twice through a 0.45-μm Millipore filter. An aliquot of 20 μL was injected into the high performance liquid chromatography (HPLC) system (Agilent 1200, Agilent Technologies, California, USA), which was equipped with a quaternary pump and a refractive index detector. The mobile phase constituted acetonitrile-water (7:3, v/v) at a flow rate of 1.0 mL·min−1 at 25°C. The HPLC system was used to integrate peak areas and determine the relative amounts of sugars in comparison to the external standard solution calibrations for chromatographically pure sucrose, glucose and fructose, which were purchased from Sigma Co. (New Jersey, USA).
Enzyme extraction and assays
This method was modified from Nielsen et al. (1991). A 1 g sample of frozen flesh powder was added to 5 mL of extraction buffer, which contained 300 mM HEPES-NaOH buffer (pH 7.5), 10 mM MgCl2, 1 mM EDTA, 0.1% (w/v) bovine serum albumin, 2.5 mM DL-dithiothreitol, 0.5% (w/v) polyvinylpolypyrrolidone (PVPP) and 10 mM ascorbate. This extract was purified by centrifugation (15,000 × g, 15 min, 4°C) and then immediately used for enzyme assays.
AIV activity was assayed following the protocol of Verma et al. (2011) by using the UV-VIS spectrophotometer. Using 1.0 mL of reaction mixture containing 200 μL of 0.5 M sucrose, 600 μL of 0.2 M sodium acetate buffer (pH 5.5) and 100 μg of the prepared desalted enzyme sample. The reaction mixtures were incubated at 37°C for 1 h, after which the assay was stopped by adding 100 μL of 3 M Tris base and boiling for 10 min. The NI assay was conducted in a 0.1 M sodium phosphate buffer at pH 7.0, following the same method as described above for AIV.
The activities of SS and SPS enzymes were assayed using the desalted extract, as described by Verma et al. (2011). For the SS assay, 50 μL of extract and an equal volume of reaction mixture, containing 4 mM UDPG, 0.06 M fructose, 15 mM MgCl2 and 0.1 M phosphate buffer (pH 8.0), were incubated for 30 min at 37°C and boiled for 10 min. After cooling, the reaction mixture volume was adjusted to 1 mL using ultrapure water, added to 5 mL of cooled anthracenone reagent (0.14 g anthracenone with 100 mL of 30% H2SO4) and boiled for a further 10 min. Next, the mixture was rapidly cooled in ice water and placed in the dark for 20 min. Color development was measured at 630 nm by using UV-VIS spectrophotometer. SPS was assayed as described above, except that the reaction mixture contained 4 mM UDPG, 0.06 M fructose-6-P, 15 mM MgCl2 and 0.1 M phosphate buffer (pH 8.0). The experiments were conducted at 0-4°C.
The enzyme activity was expressed as μmol·min−1·mg-1 sample of either glucose (for the AIV and NI assay), sucrose (SS assay) or sucrose-P (SPS assay) produced. The chemicals, including UDPG, fructose-6-P, PVPP, ascorbate, were purchased from Sigma Co.(New Jersey, USA).
RNA extraction, cDNA synthesis and expression analysis of AIV
Total RNA was extracted using the method referred to by Doyle et al. (1987). The cDNA was synthesized using a First Strand cDNA Synthesis kit (Beijing TransGen Biotech, Co., Ltd., China) and amplified by semi-quantitative RT-PCR. The specific primers for the AIV gene (AB334115) were as follows: sense, 5’-TGATTCACTGGCTCTACCTCC-3’; antisense, 5’-ACGCACTCCCACATACCC-3’. The RT-PCR was performed for 35 cycles (94°C for 1 min, 57°C for 1 min, 72°C for50 s), which produced an amplified fragment of 430 bp. The size of the amplified band was checked by running the PCR product on a 1% agarose gel containing 0.01% ethidium bromide. Actin (CN938023) was used as an internal constitutively expressed control, using the following primers: sense, 5’-CAGTGGTCGTACAACTGGTAT-3’; antisense, 5’-AGGTAGCTCATAGCTCTTCTC- 3’.
Statistical analysis
All of the experiments were performed in triplicate, and the data were statistically evaluated using a one-way analysis of variance (AVOVA) in SPSS version 20.0 (IBM Analytics, New York, USA). Significant differences were compared using Duncan’s new multiple range tests, with a probability of p <0.05 and p <0.01 considered to be significant and highly significant, respectively. Correlations between the data obtained were run using a standard Pearson correlation.
Results
Quality attributes
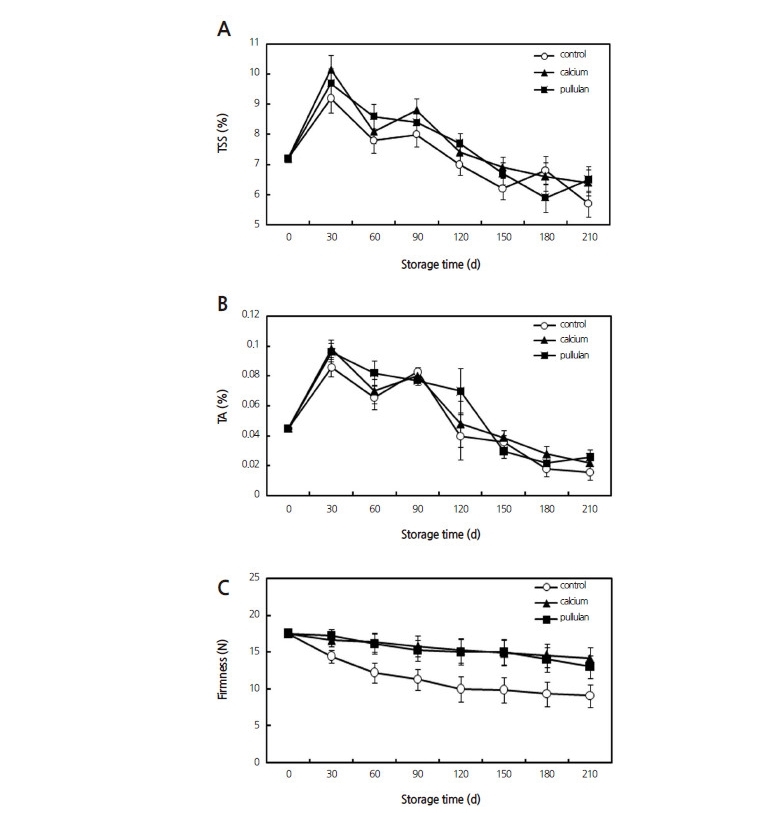
In the pullulan- and calcium-treated fruits, the changes in TSS (Fig. 1A) and TA (Fig. 1B) were similar during the storage stage, increasing to a maximum in the first month and then decreasing. Both treatments increased the TA and TSS contents in comparison with the control, and both the TA and TSS contents of pullulan-treated fruit were slightly higher than that of the calcium-treated group at the end of the storage period. During storage, the firmness of the pears decreased rapidly in the first two months in the control group, while softening was impaired by both coating treatments (Fig. 1C).
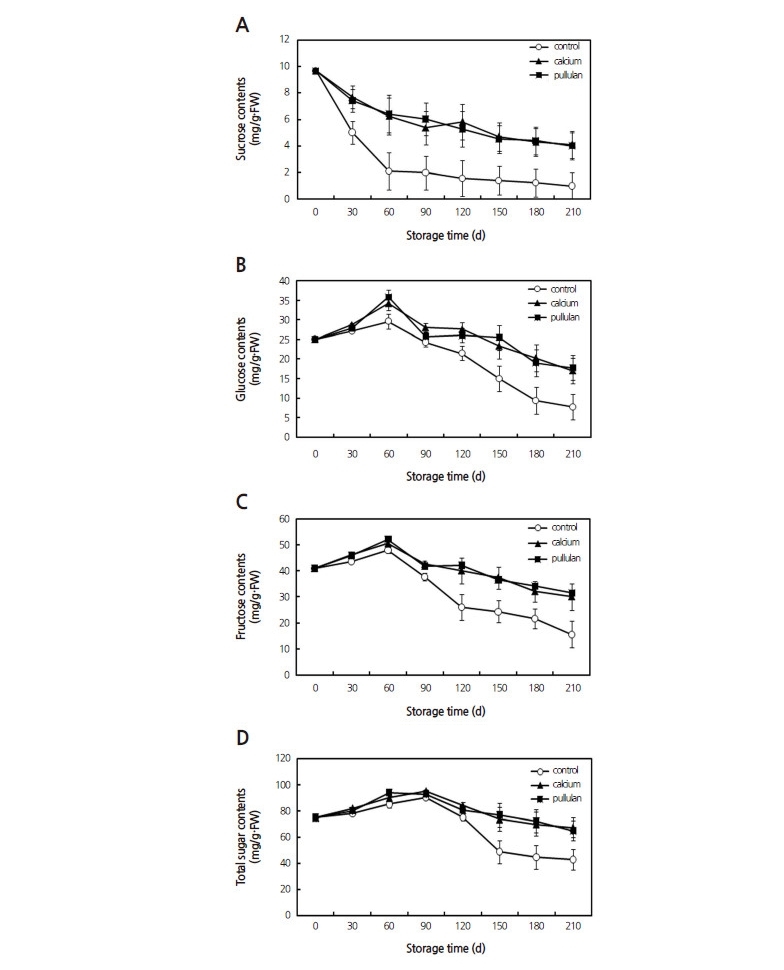
The sucrose content of the uncoated samples was found to decline sharply during storage, with a faster loss observed in the control fruits ( p <0.05; Fig. 2A). The contents of fructose and glucose, as well as the total sugars of the three groups, increased initially then decreased. The treated pears maintained higher contents of fructose, glucose and total sugars (Fig. 2B, Fig. 2C and Fig. 2D) than the uncoated samples ( p <0.05).
Respiration rate
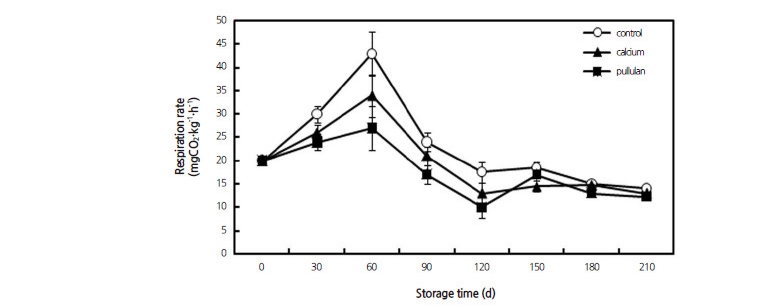
Both the pullulan and calcium chloride treatments significantly inhibited the postharvest fruit respiratory rate (Fig. 3), although the respiration rates in all fruit treatments followed a similar trend throughout storage. A typical respiratory climacteric emerged in the second month after harvest. The respiratory peak decreased by 36.3% ( p <0.01) and 18.5% ( p < 0.05) in pullulan and calcium-treated fruit, respectively, compared with the control (43.227 mg CO2·kg−1·h−1 FW).
Enzyme activities
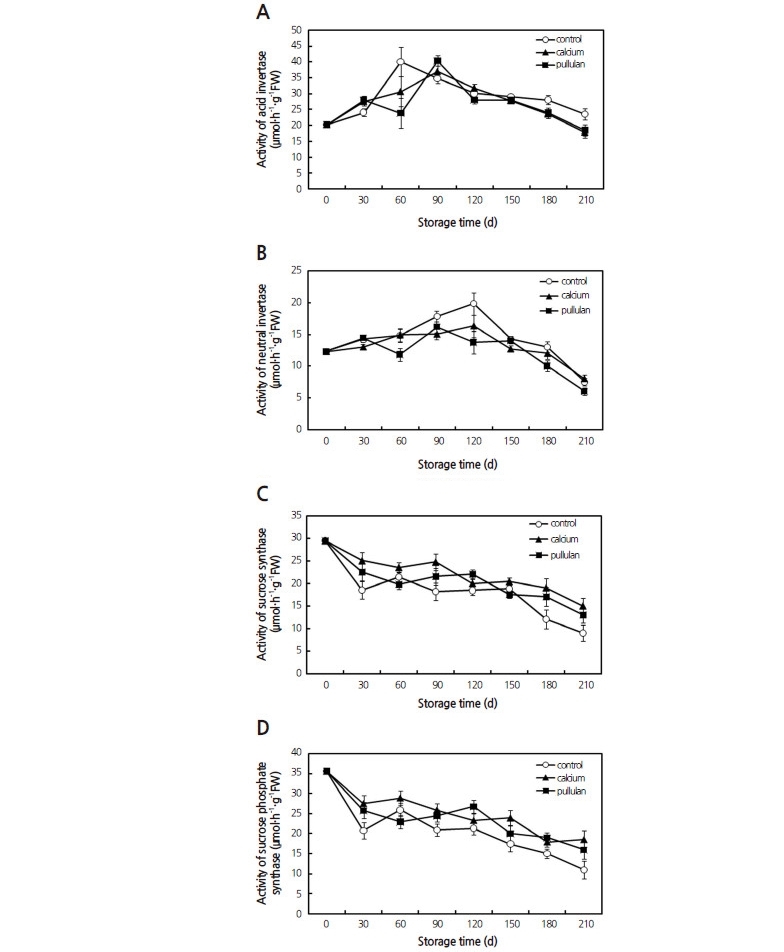
As shown in Fig. 4A and Fig. 4B, the changes in AIV and NI activity during storage were similar for all treatments. AIVactivity was found to be higher than NI activity throughout the storage period. The AIV activity of the control group reached its maximum 60 d earlier than in the treatment groups. It was notable that both calcium and pullulan inhibited AIV activity for the subsequent storage time ( p <0.05), further delaying the consumption of carbon sources. Additionally, both treatments significantly inhibited NI activity by 13.8% (calcium) and 15.8% (pullulan) compared with that of the control set ( p <0.05), but they did not affect the time of peak activity.
correlation between SS and SPS activity was observed ( p <0.01). Interestingly, calcium and pullulan had a qualitative promotional effect on the activities of SS and SPS throughout the storage period, with a stronger effect in the calcium treatment than the pullulan ( p <0.05).
AIV gene expression
To observe possible transcriptional responses to exogenous calcium and pullulan application, the level of AIV expression was also analyzed at representative time points of 30 d, 90 d, 150 d and 210 d of storage. A specific 430 bp AIV product was amplified (Fig. S1). The expression levels of the AIV gene of both the control and treatment groups were highest at 90 d and then decreased until the end of storage (Fig. 5). These observations were in accordance with the changes in the AIV activities (Fig. 4A); however, the change in AIV activity did not correspond to the expression of the AIV gene during cold storage upon treatment with calcium or pullulan (Fig. 4A and Fig. 5C). There were no significant differences in AIV expression between the treatments and the control group at any of the time points.
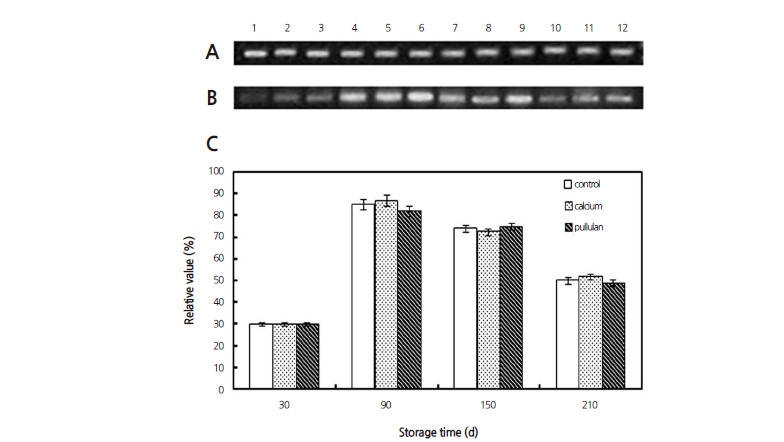
Fig. 5
Changes in the expression of the AIV gene in Huangguan pears subjected to different treatments during cold storage. The expression level of the control gene Actin is displayed in (A). The relative amount of AIV gene expression is presented in (B). In A and B, lanes 1-3, 4-6, 7-9 and 10-12 are samples taken at 30 d, 90 d, 150 d and 210 d of storage, respectively. Lanes 1, 4, 7 and 10 are the expression levels of the control group; lanes 2, 5, 8 and 11 are the samples from the calcium treatment group; and lanes 3, 6, 9 and 12 are samples from the pullulan treatment group. The relative expression levels of AIV are displayed in C. Error bars represent SD (n=3).
Discussion
In this study, the two treatments significantly inhibited the loss of TSS and TA contents in the Huangguan pear during cold storage, as well as preventing fruit softening. TSS content can approximately reflect the amount of fruit carbohydrates and can be used as a measurement of ripeness and related storage shelf-life. TA can be consumed as a respiration substrate or transformed into salts or other substances (Parker et al., 2015), which is consistent with our finding that the TA was formed in the early stages of fruit storage and significantly increased to the respiratory climacteric, peaking at 30 d and then gradually decreasing. Calcium and pullulan inhibited the respiration rate, metabolism and deterioration of fruit, resulting in a delayed loss of TSS and TA. Fruits soften during storage, and this is a major factor that often dictates shelf-life. Flavor change associated with fruit ripening also represents a major characteristic, along with softening, for the determination of fruit quality (Makkumrai et al., 2014). This study found that softening was delayed during cold storage in Huangguan pears treated with calcium chloride and pullulan. Wong et al. (1994) reported that calcium ions (Ca2+) were involved in the regulatory function of many enzymes in cellular and physiological processes, and it is known to play an essential role in the structural maintenance of membranes and cell walls (Zheng et al., 2014). Preharvest and postharvest treatments with calcium salts have been effective in controlling several physiological disorders, reducing the incidence of fungal pathogensand maintaining fruit firmness (Bakshi et al., 2005). In contrast, the retardation of softening in the pullulan-treated pears may be ascribed to a reduction in water loss, as pullulan-based coatings provide a mechanical barrier (Wu et al., 2016). No significant differences were observed between the calcium and pullulan treatments in any of the characteristics examined in the stored Huangguan pears.
The behavior of individual sugars during storage could be explained in terms of sucrose metabolism (Gerhardt et al., 1987). Several postharvest treatments that delay the aging process allow fruits to maintain higher levels of sugar; however, the presence of saccharolytic enzymes can be associated with a decrease in the sucrose concentration (Hasperue et al., 2014). It is expected that sucrose degradation, catalyzed by NI, AIV and SS, became predominant to satisfy the carbohydrate requirement for respiration (Zhu et al., 2013).
Previous research has suggested that edible coatings could reduce the respiration rate of fruits and vegetables (Mahfoudhi and Hamdi, 2014; Ozden and Bayindirli, 2002). Many edible coatings have excellent selective permeability to O2 and CO2 (Elsabee and Abdou, 2013). Oxygen is the key factor that causes oxidation, inducing several unwanted food changes such as odor, color and flavor, as well as nutrient deterioration. When fruits and vegetables receive less oxygen, the production of the ripening-accelerating compound ethylene is disrupted and the physiological loss of water is minimized, enabling them to remain firm, fresh and nutritious for longer periods of time (Dhall, 2013). Coatings providing a proper oxygen barrier can therefore help in improving food quality and extending food shelf life (Sothornvit and Pitak, 2007). Bai et al.(2002) reported that gaseous exchange in fruits occurs by permeation through the skin or diffusion through open pores (e.g., stomates, lenticels, stem, and blossom scars). Our study showed that both of the coating treatments partially or fully suppressed the respiration rates of pears, which was probably due to pore blockage (Zapata et al., 2008). The respiration rate of pullulan-treated fruit was lower than that of the calcium-treated group, possibly because pullulan has a characteristic resistance to air. This result is similar to that obtained with other edible coatings (Esteban et al., 2011; Hanani et al., 2012). Clearly, the coating treatments contributed to the reduction of vital ripening activities, thus maintaining the quality of Huangguan pears during storage. Respiration converts carbohydrates to supply the energy and other substrates for the fruit; thus, there was a correlation between the sugar content and the respiration rate.
In addition to their effects on respiration, edible coating treatments of fruits and vegetables have been reported to slow their metabolism (Park et al., 1994; Trevino-Garza et al., 2015; Wu et al., 2016), indicating that the activities of enzymes involved in the metabolism of sugar may be suppressed by the application of edible coatings. This could lead to the observed inhibition of hexose degradation in pears during storage; the present study showed that sucrose in the control group decreased remarkably in the first 60 d, which was concomitant with an increase in AIV activity. The AIV activity in the stored Huangguan pears increased at first and then decreased slowly, a pattern which was also reported in broccoli florets (Coupe et al., 2003). Both of the coatings postponed peak in AIV activity. One proposed hypothesis is that calcium and pullulan preserved the stored Huangguan pears by inhibiting AIV activity. NI is also involved in the cleavage of sucrose, and its activity pattern throughout the storage stage of the coated and control groups was similar to the reported trends observed in loquat fruit during cold storage (Shao et al., 2013).
SPS activity may play an important role in sucrose accumulation (Botha and Black, 2000; Shi et al., 2012), as it catalyzes the synthesis of sucrose for the generation of UDP. SS decomposes sucrose for the generation of UDPG and the possible storage of energy via a UDP activation of the hexose moiety (Bahaji et al., 2015). The higher activities of SS and SPS may balance the sucrose content and provide energy for the fruit during storage, which was in accordance with our results. Additionally, SS is thought to favor the generation of UDPG from sucrose for the formation of cell wall materials. Calcium is known to influence the structural stability of the fruit tissue as well as enzyme activity, and is an important chemical component in the organs of fruits and vegetables. Exogenous calcium is hypothesized to promote cell wall formation, which could lead to an increase in SS activity, as was observed in table grapes (Ciccarese et al., 2013). The activity levels of SS and SPS were correlated, indicating a synergistic effect of these two enzymes on sucrose concentration that requires further investigation. Membrane integrity plays an important role in the prevention of sugar metabolism by separating substrates from enzymes through intracellular compartmentalization (Boonsiri et al., 2007). Our results indicated that treatment with a coating of with 2% CaCl2 or 1% pullulan promoted the conversion to sucrose by maintaining SS and SPS activities, and delayed the hydrolysis of sucrose by inhibiting the activities of NI and AIV, therefore significantly inhibiting hexose degradation and resulting in higher sucrose levels in the stored pear fruits.
Zrenner et al. (1996) reported that the soluble acid invertase gene (INV-19) was involved in reducing sugars during cold storage in potato tubers. Itai and Tanahashi (2008) investigated the expression of two AIV genes (PpAIV1 and PpAIV2) in Japanese pears during storage, and suggested that PpAIV1 could be responsible for the observed sucrose degradation. In the present study, the Huangguan pear AIV gene expression increased rapidly and decreased gradually during storage, with the highest expression level at 90 d, which corresponds to the changes in the AIV activities. There were no significant differences between the coating-treated group and the control group, which was also consistent with their AIV activities.These differences in results may be caused by the varying requirements for energy and substrates in different species, and as a result, the function and expression of AIV need further investigation. Treatments that prohibit sugar metabolism seem to be associated with a low respiration rate, low AIV and NI activity, and high SS and SPS activities.
In conclusion, treatments with 2% calcium or 1% pullulan inhibit the activities of the sucrose-cleaving enzymes AIV and NI and promote SS and SPS activities, thus postponing the consumption of sucrose and maintaining the hexose contents in the Huangguan pears. These effects were probably associated with the film-forming properties of the treatments, which could reduce the respiration rate by establishing an oxygen barrier through the skin of the fruit. No statistically significant differences between the two treatment groups were found; hence, we concluded that both 2% calcium and 1% pullulan coatings could maintain fruit quality and prolong the storage life of Huangguan pears.
Conflict of interest: The authors declare no conflict of interest.