Introduction
Materials and Methods
Characteristics of Rootstocks
Cultivation of Grafted Melons in a Greenhouse
Measurements and Analysis
Statistical Analysis
Result and Discussion
Introduction
Most of melon (Cucumis melo L.) in Korea has been cultivated consecutively in a greenhouse for purposes of early harvest, stable production and quality improvement (Lee et al., 2009; MIFAFF, 2012). However, the continuous and intensive cultivation in the same greenhouse causes the increase of pathogen density and salt accumulation in soil, resulting in the incidence of soilborne diseases and crop yield reduction. Fusarium wilt by Fusarium oxysporum Schltdl., gummy stem blight by Didymella bryoniae, and Phytophthora blight by Phytophthora capsici are the most common and damaging soilborne diseases of cucurbit crops worldwide (Cohen et al., 2002; Davis et al., 2008b; Heo et al., 2001).
Monosporascus root rot and vine decline (MRR/VD), caused by the soilborne pathogen Monosporascus cannonballus Pollack & Uecker, is also a serious melon disease. The incidence of MRR/VD has been reported in many regions around the world such as the US, Israel, Japan, Korea, and so on (Cohen et al., 2000; Heo et al., 2001). The symptoms include the wilting and collapse of vines late in the season, often only days or a few weeks before harvest, resulting in poor quality melons with small size, lower sugar content, and soluble solids (Martyn, 2007). Losses due to MRR/VD fluctuate year to year from 10 to 25% of the crop, although individual fields may suffer 100% loss (Martyn and Miller, 1996).
Short-term crop rotation, soil solarization, and fungicides have been ineffective in controlling MRR/VD (Martyn and Miller, 1996). Preplant fumigation with methyl bromide is the best option for controlling MRR/VD. However, the production and use of methyl bromide were curtailed because it was recognized as ozone-depleting chemical. Screening accessions for resistance to MRR/VD or the development of cultivars with resistance or tolerance to MRR/VD have been continued (Fita et al., 2009).
Grafting has been widely used in order to reduce con-tinuous cropping injuries, together with cropping rotation, the use of resistant cultivars, and soil disinfestation (Lee et al., 2010). Melons are generally grafted onto Cucurbita rootstocks to manage soilborne pathogens. When melons were grafted onto the Cucurbita rootstocks, the incidence of MRR/VD on grafted melon was significantly lower than on nongrafted plants (Cohen et al., 2000; Davis et al., 2008b).
However, the fruit quality of grafted melons is influenced by the combination of scion and rootstock. When melons and oriental melons were grafted onto vigorous squash interspecific hybrid such as ‘Shintozwa’ (Cucurbita maxima D. × C. moschata D.), preharvest internal decay or breakdown and abnormal fermentation occurred in the fruit. Cucurbita rootstocks reportedly also caused the decrease of the sugar content and the increase of vitrescence (a physiological disorder in which fruit flesh appears water-soaked) in the fruit (Davis et al., 2008b).
Melons grafted onto melon rootstocks are expected to have less horticultural problems related to scion-rootstock compatibility (King et al., 2010). Accordingly, grafting onto resistant melon rootstock may improve the resistance without reducing fruit quality.
This study evaluated effects of melon rootstocks with MRR/VD and Fusarium wilt on the tolerance to MRR/VD, yield and fruit quality of grafted melons under greenhouse conditions.
Materials and Methods
Characteristics of Rootstocks
The genotypes of melon rootstocks are listed in Table 1. These rootstocks were collected from Turkey, Egypt, India, Uzbekistan, and Japan, and screened for their resistance to both MRR/VD and Fusarium wilt (Lee et al., 2012; Moon et al., 2009; Park et al., 2013). Melon rootstocks of R1 to R6 showed the resistance to MRR/VD in the greenhouse evaluation. R1, R2, R3, and R4 were highly resistant to MRR/VD. R5 and R6 were moderately and slightly resistant to MRR/VD, respectively. ‘Homerunstar’ (Syngenta Seeds Co., Ltd.) which belongs to the inodorus group of C. melo and susceptible to MRR/VD was used as scion. Non-grafted ‘Homerunstar’ and plants grafted onto squash interspecific hybrid ‘Shintozwa’ (Nongwoo Bio Co., Ltd.) rootstock served as controls.
Rootstocks and scions were sown into the 72-cell plugtrays (W 280 × L 540 × H 45 mm, Bumnong Co., Ltd., Jeongeup, Korea) filled with commercial growing substrate (Biomedia, Seminis Vegetable Seeds, Inc.). They were grown on the bench in a greenhouse.
Root-removed one cotyledon splice grafting method was used according to the procedure described by Lee et al. (2010). Grafted seedlings were placed in the 32-cell plugtrays filled with new substrate (W 280 × L 540 × H 63 mm, Bumnong Co., Ltd., Jeongeup, Korea). Plants were watered daily except the period during healing and acclimatization. After grafting, plants were healed and acclimatized in the tunnel covered with double-layered plastic film and shade cloth in the greenhouse for one week (Lee et al., 2010). A nutrient solution (electric conductivity 1.5 dS・m-1 with ‘Hanbang’ for seedling, N-P-K-Ca-Mg = 8.0-2.4-2.4-4.8-1.6 me・L-1, Coseal Co., Ltd., Gunsan, Korea) was applied and the application frequency was determined depending on growth stage (RDA, 2008).
Cultivation of Grafted Melons in a Greenhouse
Two experiments were conducted in unheated polyethylene greenhouses. In two experiments, tolerance to MRR/VD of melons grafted onto different rootstocks was evaluated in greenhouse (243 m2) infested with M. cannonballus.
In the first experiment (summer cultivation), 34-day-old grafted and 27-day-old non-grafted melon seedlings were transplanted on May 30, 2012. In the second experiment (autumn cultivation), 27-day-old grafted and 18 day-old non-grafted melon seedlings were transplanted on August 13, 2012. Three double-rows (1.2 × 26 m, rows were 0.5 m apart in double-row) were made and each double-row was mulched with black plastic film prior to planting. Three rows of drip irrigation tapes were placed under the plastic mulch in each double-row. Melon transplants were grown between drip irrigation tapes (0.5 m between plants within row) at a plant density of 1.31 plants/m2. The experiment was arranged in a randomized complete block design with three replicates. Each replicate consisted of ten plants except rootstocks with low emergence percentage or graft-take.
Irrigation and additional fertilizer application was carried out using standard procedures for melon cultivation (RDA, 2005). Additional fertilizer was applied depending on the growth conditions through drip irrigation system. Ammonium sulphate and potassium sulfate were applied before fruit set (one time in the summer cultivation and three times in the autumn cultivation, respectively). After fruit set, ‘Hanbang’ for watermelon cultivation (N-P-K-Ca-Mg = 12- 3-5-6-3 me・L-1, Coseal Co., Ltd., Gunsan, Korea) was applied (two times in the summer cultivation and three times in the autumn cultivation, respectively). Melon was trained vertically by staking. Manual cross-pollination was conducted for fruit set. One fruit per plant was set on lateral vines between 10th to 12th nodes of main vine, with removal of other lateral vines. After fruit set, the apical bud of main vine was removed on 10th nodes above fruit-bearing node. Air temperature and relative humidity data inside the greenhouse were recorded at an interval of 30 minute using a data logger (TR-72U thermo recorder, T&D Co., Matsumoto, Japan).
Measurements and Analysis
The Monosporascus wilt incidence and survival rate of melons grafted onto different rootstocks were recored during the experiments. Mature fruits were harvested from 30 July to 6 August 2012 (61 to 68 days after transplanting in the summer cultivation) and from 20 to 30 October 2012 (61 to 68 days after transplanting in the autumn cultivation) according to the date of pollination. Nine mature fruits per treatment were harvested except the treatments in which the fruits could not be harvested by the incidence of disease. The length, width, and weight of fruit were measured. Each fruit was sliced into two (longitudinal), and the flesh thickness at the middle of the slice was measured using a caliper gauge. From the middle of the slice, three pieces of fruit flesh were cut off, with the core and peel removed. The remaining flesh of each piece was used for measuring fruit firmness with a texture analyzer (TAPlus texture analyzer, Lloyd Materials Testing, Bognor Regis, UK) equipped with a 5 mm-diameter round-head plunger. Another set of each piece was squeezed to extract the juice for measuring total soluble solids (TSS) using refractometer (GMK-701R, G-Won Hitech., Co., Ltd., Seoul, Korea) and titratable acidity using titrator (TitroLine Easy, Schott Instruments GmbH, Mainz, Germany).
The other set of each piece was frozen in a freezer (Ultra low temperature freezer, ilShinBioBase, Dongduchun, Korea) with a tempertrature of -75°C and dried in a freeze dryer (PVTFD10R, ilShinBioBase, Dongduchun, Korea). Then the mineral content of fruits was measured. Dried fruits were powdered and 500 mg of this was hydrolyzed with the solution of HNO3:HClO4 = 3:1. Total nitrogen (T-N) was measured by Kjeldahl method using nitrogen analyzer (KeltecTM 8400 Analyzer, Foss, Hilleroed, Denmark). Potassium, calcium, and magnesium were measured using atomic absorption spectrometer (AA-6800F, Shimadzu Kyoto, Japan). Phosphorus was measured by vanadate method, using spectrophotometer (UV-3150, Shimadzu, Kyoto, Japan).
Statistical Analysis
All data were statistically analyzed by analysis of variance using SAS version 9.1 (SAS Institute Inc., Cary, NC, USA) software. Duncan’s multiple range test was performed at p ≤ 0.05 on each of the variables measured.
Results and Discussion
|
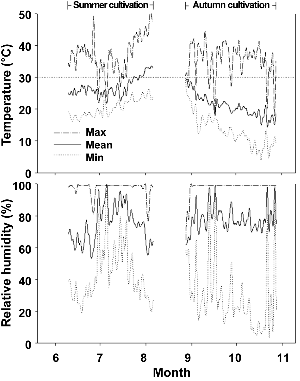
Fig. 1. Changes in daily maximum (max), mean, and minimum (min) air temperature during the summer and autumn cultivations. |
Grafting has been utilized as an efficient tool for improving disease resistance, yield, and quality of plants (Davis et al., 2008b; King et al., 2010, Lee et al., 2010). Grafting in melons has been reported to improve the resistance to Fusaruim wilt (Bletsos, 2005), MRR/VD (Cohen et al., 2000), root-knot nematodes (Sigüenza et al., 2005). Melons are generally grafted onto the same species (C. melo L.) or Cucurbita rootstocks. It was recently reported that grafting onto Cucurbita rootstocks improved the resistance of both Fusarium wilt and gummy stem blight and yield (Crinò et al., 2007). However, the fruit quality of melons reportedly decreased by grafting onto vigorous squash interspecific hybrid such as ‘Shintozwa’ (Cucurbita maxima D. Í C. moschata D.).
The MRR/VD is recently one of the most devasting diseases in the production of melons. In this study, the tolerance to MRR/VD, yield, and fruit quality of melons grafted onto melon rootstocks were evaluated under greenhouse conditions, for high disease resistance and less problems related to fruit quality or scion-rootstock compatibility.
In the summer cultivation of the melons, the average daily maximum, mean and minimum temperatures were 37.4, 27.0 and 20.4°C, respectively (Fig. 1). Even though the temperature of the autumn cultivation was lower than that of the summer cultivation, the maximum temperature often rose above 30°C in daytime during the cultivation period. M. cannonballus is common in hot and semiarid areas and the optimum growth temperature is 30 to 35°C (Martyn and Miller, 1996). High soil tempereature under cultural conditions such as plastic mulch, plastic tunnel, or glasshouse melon culture induces the growth and infection of M. cannonballus in otherwise temperate zone (Uematsu et al., 1992). The MRR/VD is more severe under stress conditions such as heat and drought (Martyn and Miller, 1996).
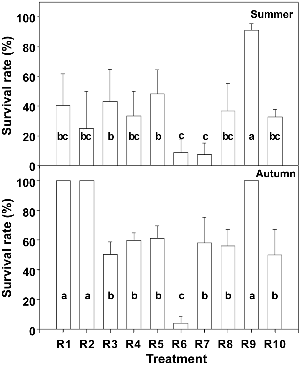
The incidence of wilting and the severity of disease differed depending on growing season and rootstsocks. In the summer cultivation, wilting by M. cannonballus had occured in melons grafted onto R7 susceptible to MRR/VD since around 40 days after transplanting (Fig. 2). Wilting symptom spreaded to those grafted onto other rootstocks and some of these died before harvest. Whereas non-grafted melons (R10) showed lower survival rate (33%), ‘Shintozwa’ (R9) rootstock showed higher surviral rate (91%). The survival rates of grafted melons grafted onto melon root-stocks differed depending on the rootstocks (average 30%), ranging from 48% (R4) to 8% (R7). The survival rates of melons grafted onto R6 and R7 were lower than that of non-grafted seedlings.
In the autumn cultivation, the symptoms of MRR/VD progressed more slowly and the survival rate of plants was higher than those in the summer cultivation in which the temperature in the greenhouse was higher. The wilting symptom had appeared in non-grafted melons (R10) around 35 days after transplanting. It occurred consecutively in R6, R7, R8, and ‘Shintozwa’ (R9). ‘Shintozwa’ (R9) and R1, R2 of which wilt incidence appeared late showed less severe disease symptom and all of these survived.
The results are similar to those reported by Cohen et al. (2005). They reported that significant differences in MRR/VD incidence were found between growing seasons, and disease reduction and the beneficial effect of grafting on yield were more pronounced in the moderate-temperature spring than in the high-temperature autumn. In this study, the lower temperature in the autumn cultivation resulted in longer growing season. The days from flowering to harvest and growing season in the autumn cultivation was approximately 10 days longer than those in the summer cultivation (data not shown).
Martyn and Miller (1996) reported that late-maturing varieties expressed less severe disease symptoms than early-maturing varieties. Moreover, they reported that the progression of symptoms of MRR/VD is affected by fruit load and maturity stage. Fruit removal reduced leaf stomatal conductance and increased root growth, thus enabling the plants to survive (Pivonia et al., 2002). The lighter fruit (average 1.2 kg) in the autumn cultivation was also con-sidered to be one factor associated with slower progression and less severity of disease symptoms (average 1.6 kg in the summer cultivation).
|
Fig. 2. The survival rate of melons grafted onto different rootstocks grown in the greenhouse infested with M. cannonballus in the summer and autumn cultivations. |
The melons grafted onto ‘Shintozwa’ (R9) rootstock exhibited less severe disease symptom and the higher survival rate in both seasons. This disease reduction of ‘Shintozwa’ (R9) rootstock was reported to be attributed to its vigorous large root system (Cohen et al., 2000). In the summer cultivation, grafting onto melon rootstocks did not result in the increase of survival rate compared to non-grafted melons, even though there were differences in the progression of wilting symptoms.
Unlike the summer cultivation, melons grafted onto R1 and R2 in the autumn cultivation exhibited higher survival rate. In the autumn cultivation, R1 and R2 were resown owing to poor emergence. The melons grafted onto R1 and R2 were transplanted at younger stages and their days to flowering took a little bit longer than others. It has been reported that the age of seedlings affected the growth of plant after transplanting and younger transplanted seedlings had greater root growth (Mishra and Salokhe, 2008). Although the size of root system of rootstocks in the field was not evaluated in this study, it is considered that the seedling age may affect the root growth and this would contribute to overcoming MRR/VD.
Table 2 showed the fruit size, fruit mass, and yield of melon influenced by the rootstock in both seasons. In summer cultivation, melon grafted onto ‘Shintozwa’ (R9) rootstock had the heaviest fruits, followed by R4, R5, and R8. Melons grafted onto ‘Shintozwa’ (R9) and R4 yielded 15% and 13% respectively more than non-grafted melons. ‘Shintozwa’ (R9) rootstock also had the heaviest fruits and yielded highest in autumn cultivation even though the fruit size, fruit mass, and yield of melon in autumn cultivation were lower than in the summer cultivation. The yield of R1 followed.
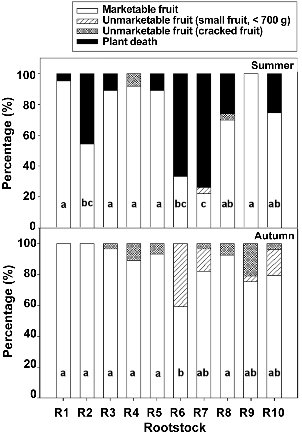
In the summer cultivation, melons grafted onto ‘Shintozwa’ (R9), R1, R3, R4, and R5 showed higher percentages of marketable fruit, while R2, R6, and R7 showed lower percentages than non-grafted melons (Fig. 3). The reduction of marketable fruit was caused by plant death, disease incidence, and small fruits below 0.7 kg or cracked fruits, depending on rootstocks. Marketable yield was highest in ‘Shintozwa’ (R9), followed by R4, R5, and R1.
In the autumn cultivation, melon grafted onto R1, R2, R3, R4, R5, and R8 showed higher percentages of marketable fruit. The percentage of marketable fruit of ‘Shintozwa’ (R9) in the autumn cultivation was lower than in the summer cultivation, due to the reduction of marketable fruits by fruit craking. The marketable yield was highest in R1, followed by ‘Shintozwa’ (R9), R2, R3 and R5. Even though the survival rate and percentage of marketable fruits in the autumn cultivation was higher than in the summer cultivation, the yield in the autumn cultivation was lower than that in the summer cultivation because of lighter fruit.
The flesh thickness, firmness, total soluble solids (TSS) contents, titratable acidity (TA), and TSS/TA ratio of melon grafted onto different rootstocks varied with the growing season (Table 3). The fruit flesh in the summer cultivation was thicker (average 4.0 cm) than that in the autumn cultivation (average 3.5 cm) and had no significant differences among rootstocks. The fruit flesh of melons grafted onto R1 was the thickest in the autumn cultivation. The fruits in the summer cultivation were firmer (average 9.6 N) than those in the autumn cultivation (average 6.2 N) and melons grafted onto ‘Shintozwa’ (R9) were the softest. The melons grafted onto ‘Shintozwa’ (R9) in the summer cultivation had the highest TSS. In the autumn cultivation, no significant differences among rootstocks were observed for firmness, TSS, and TSS/TA ratio.
|
Fig. 3. The percentage of marketable or unmarketable fruits of melons grafted onto different rootstocks in the summer and autumn cultivations. |
It has been reported that grafting influences the fruit maturity and its quality such as sugar and acids contents, and juice pH (Davis et al., 2008a). There are many conflicting reports on the changes in fruit quality by grafting (Davis et al., 2008a; Lee et al., 2010). Grafting onto squash interspecific hybrid rootstocks reduced fruit firmness (Zhao et al., 2011). The TSS of melons grafted onto squash interspecific hybrid rootstocks was also reported to be reduced or similar to non-grafted melons (Crinò et al., 2007; Zhao et al., 2011).
In this study, melons grafted onto ‘Shintozwa’ (R9) rootstock had the lowest firmness and highest TSS in the summer cultivation. Rouphael et al. (2010) reported that the decrease of photosynthesis may be related to low fruit quality with a high photosynthate demand by the heavy fruit load. Although the photosynthesis of melons grafted onto different rootstocks was not examined in this study, the lower TSS of melon fruits grafted onto melon rootstocks or non-grafted melon fruits could be due to lower photo-synthesis by the wilting and death of melon plants by M. cannonballus.
|
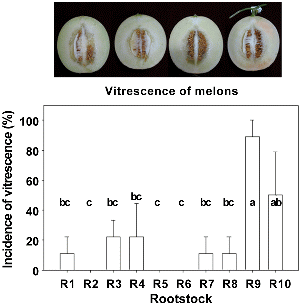
Fig. 4. The incidence of vitrescence in the fruit of melons grafted onto different rootstocks in the aututmn cultivations. |
Grafting was also reported to increase vitrescence in melon (Rouphael et al., 2010). In this study, the softening and vitrescence incidence in melon fruits mainly appeared in the autumn cultivation (Fig. 4). In the summer cultivation, the only melon fruits with lowest hardness grafted onto ‘Shintozwa’ (R9) showed the vitrescence. Meanwhile, the incidence of vitrescence in the autumn cultivation increased and was highest in the melons grafted onto only ‘Shintozwa’ (R9). The melons grafted onto melon rootstocks had fewer incidences of vitrescence than those of ‘Shintozwa’ (R9) (89%) or non-grafted melons (50%). The vitresecence did not appear in R2, R5, and R6.
Low calcium level reportedly induced the softening and vitrescence incidence in melon fruits (Johnstone et al., 2008; Madrid et al., 2004; Serrano et al., 2002). Rouphael et al. (2010) reported that the incidence of the physiological disorder would be closely associated with the rootstocks. Likewise, the vigorous rookstock such as squash interspecific hybrid tend to absorb more nitrogen and less calcium into the fruits, thus resulting in the incidence of premature internal decay in oriental melons.
In this study, mineral contents of melon fruits were also influenced by rootstocks and varied with the growing season (Table 4). Phosphorus content in the fruit in the summer cultivation was higher than in the autumn cultivation. Calcium and magnesium contents were higher in the autumn cultivation. Phosphorus, calcium, and magnesium contents of the fruits grafted onto ‘Shintozwa’ (R9) were lower than those grafted onto melon rootstocks or non-grafted melons.
Accordingly, the vitrescence incidence may be related to fruit softening. It would be caused by lower calcium content, considering melons grafted onto ‘Shintozwa’ (R9) with lowest calcium content showed more virescence incidence. However, the calcium content in fruits was higher in the autumn cultivation than in the summer cultivation. Other factors besides calcium would be related to vitrescence incidence.
Cohen et al. (2005) reported that the disease prevention achieved by grafting depends not only on the contribution of the rootstock but also on the response of the scion to the disease. In general, the results of this study indicate that even though melon rootstocks had shown high levels of resistance to both MRR/VD and Fusarium wilt in the greenhouse evalution (Lee et al., 2012), grafting using these melon rootsctoks could not result in disease reduction as grafting onto ‘Shintozwa’ (R9) did. However, it was confirmed that grafting onto melon rootsctoks such as R1, R3, R4, and R5 yielded marketable fruits as much as ‘Shintozwa’ (R9), especially in autumn season. Grafting onto melon rootstocks also had no reduction of fruit quality such as low calcium content, fruit softing, and vitrescence. The results in this study suggest that grafting onto the melon rootstocks may increase the marketable yield without the reduction of fruit quality.