Introduction
Materials and Methods
Isolated Microspore Culture
Germination of Microspore-derived Embryos
Determination of Ploidy Level
Bud Pollination
Plant Materials for Glucosinolates Analysis
Analysis of Glucoraphenin and Glucoraphasatin by HPLC
Purification of Glucoraphenin and Glucoraphasatin by Al2O3-column and prep-HPLC
Statistical Analysis
Results and Discussion
서 언
Radish (Raphanus sativus L.) is a major vegetable crop in Korea, China, Japan, and Southern Asia. Both the root and leaf tissues of radish are used as the main ingredient of “Kimchi”, a healthy fermented food native to Korea. Increasing the content of the health-promoting compounds in radish, including vitamin B and C and glucosinolates (GSLs), is a common goal in the development of new varieties (Curtis, 2003, 2011), which leads to medicinal and functional value of the plant.
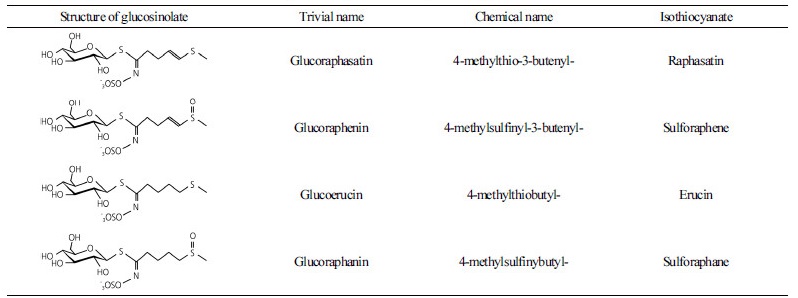
GSLs are a group of sulfur- and nitrogen-containing secondary metabolites in Brassica crops (Fahey et al., 2001; Barillari et al., 2005). Over 160 different GSLs have been reported in diverse plants and are
classified as aliphatic, indole, and aromatic GSLs depending on their precursor amino acids. Previous reports suggested that GSLs have potential anticancer activity (Heber, 2004; Grubb and Abel, 2006; Higdon et al., 2007). The composition of GSLs is influenced by species, variety, developmental stage, and plant tissue (Fahey et al., 2001). Recent studies have also shown that glucoraphenin (GRE) and glucoraphasatin (GRH) are abundant and unique in radish (Barillari et al., 2005; Han et al., 2015). These two GSLs have a double bond on their side chain compared with glucoerucin (GER) and glucoraphanin (GRA) in broccoli (Table 1) (Barillari et al., 2005; Barillari et al., 2007; Montaut et al., 2010). The chemopreventive activity of isothiocyanate (ITC) by hydrolysis of GSLs has been previously reported (Dawson et al., 1993; Saavedra et al., 2010). Sulforaphene and raphasatin, derived from GRE and GRH, respectively, have been shown to inhibit the growth of tumor cells and have antioxidant activities (Barillari et al., 2007; Montaut et al., 2010).
To improve the yield and health-promoting potential of radish plants, conventional breeding using mass selection, combined mass pedigree selection, and bud pollination have been used. However, these breeding practices are labor-intensive and time-consuming. The doubled haploid (DH) plants method via isolated microspore culture (IMC) a faster and more effective method for developing homozygous parental lines. DHs are very powerful in that genetic variations can be generated within a near isogenic background and provide homozygous lines in one or two generations (Babbar et al., 2004; Forster et al., 2007; Ferrie and Caswell, 2011; Ferrie and Möllers, 2011; Bhatia et al., 2017). Numerous factors can influence the success of embryogenesis including genotype and the condition of the donor plant, the developmental stage of the microspore, and the culture environment (Smýkal, 2000; Babbar et al., 2004; Seguí‐Simarro and Nuez, 2008; Ferrie and Möllers, 2011). A highly efficient IMC system for the generation of embryogenesis and DH plants has been reported in various Brassica species (Custers, 2003; Da Silva Dias, 2003; Ferrie, 2003). Although some attempts of tissue culture have been reported in radish plants, this species is considered one of the most difficult species to regenerate among Brassica crops (Takahata et al., 1996; Curtis, 2003; Chun and Na, 2011; Chun et al., 2011; Han et al., 2014).
The objective of this study was to generate near isogeneic lines of radish plants by IMC and to examine the variation of GSL content within the DH population. Furthermore, GRE and GRH were purified to near homogeneity from the radish tissues and were used as standards for quantitative analysis from the DH lines.
Materials and Methods
Isolated Microspore Culture
R. sativus L. cv ‘Taebaek’ (Monsanto Co., Seoul, Korea) were used as donor plants for IMC. Seeds were sown in 32 hole plugs. When the seedlings were 4 weeks old, they were placed in a cold room at 4.0 ± 1.0°C under a 16/8 h (light/dark) photoperiod for 4 weeks to induce flower bud differentiation. After cold treatment, the plants were transferred to a greenhouse to induce bolting and flowering.
Flower buds of 4.0 ± 0.5 mm in length with a longer stigma than the floral leaf were collected. The surface of the flower buds was sterilized with 1% sodium hypochlorite for 15 min followed by rinsing three times with sterile deionized water. The buds were then ground in a mortar containing Nitsch and Nitsch (NLN) liquid medium (Lichter, 1982) supplemented with 10% sucrose. A microspore suspension was filtered through 45 μm nylon screens. The suspension was centrifuged three times at 100 × g for 15 min each. The microspores were suspended in 1/2 NLN medium supplemented with 13% sucrose. Then, 3 mL of microspore suspension was placed in a 60 × 15 mm sterile petri dish supplemented with 1% activated charcoal. The petri dish was incubated at 32.5°C for 48 h prior to maintenance at 25.0°C in darkness. After 2 weeks of culture, the petri dish was transferred to a gyratory shaker and agitated at 70 rpm with a 18/6 h (light/dark) photoperiod at 25°C. The embryo yield was examined 4 weeks after culture.
Germination of Microspore-derived Embryos
The cotyledonary embryos were transferred to 1/4 Murashige and Skoog (MS) medium (Murashige and Skoog, 1962) and supplemented with 3% sucrose and 7.5% agar at pH 5.8. All microspore derived embryos were incubated at 20 ± 2.0°C under a 16/8 h (light/dark) photoperiod to induce shoot and root regeneration. The shoots were transferred to MS medium supplemented with 3.75% agar for rooting. Rooted embryos in solid medium were rinsed with sterile deionized water and transferred to autoclaved artificial soil. The regenerated plants were grown in a sealed chamber with polyethylene film for acclimatization at 20 ± 2.0°C under a 16/8 h (light/dark) photoperiod. After 2 and 6 d, the humidity in the chamber was gradually lowered by making small holes in the polyethylene film. After 10 d plants generated by IMC were transferred to pots and grown in a greenhouse.
Determination of Ploidy Level
The ploidy status and DNA content of the regenerated plants was determined by flow cytometry (Cyflow Ploidy Analyser; Sysmex Pactec GmbH, Görlitz, Germany). Young leaves (5 mm2) of radish plants with diploid status (2n = 18) were used as the control. Young leaves of the DH lines were individually chopped in a 60-mm glass petri dish containing 400 µL of extraction buffer (Solution A in the CyStain UV Precise P Kit; Sysmex Pactec GmbH). Then, 1.6 mL of 4, 6-diamidino-2- phenylindol staining buffer (Solution B of the same kit) was added. The suspension was filtered through a 30 µm nylon mesh (CellTrics, Sysmex Pactec GmbH). For each sample, 2,500-5,000 nuclei were analyzed by flow cytometry using a device equipped with an HBO-100 mercury lamp.
Bud Pollination
The DH lines selected by flow cytometry analysis were grown for 6 weeks in a greenhouse. The plantlets were placed in a cold room at 4.0 ± 1.0°C under 16/8 h (light/dark) photoperiod for 4 weeks. After inducing floral bud differentiation, plants were kept in a greenhouse to produce flowers. Mature buds of the regenerated radish plants were pollinated by bud pollination to produce seeds. At the beginning of flower bud opening, mature pollen was directly placed onto the stigma. Pods were harvested 50 d after bud pollination and dried in shade. The seeds were harvested and kept dry until use.
Plant Materials for Glucosinolates Analysis
The eight-seeds of the 41 DH lines and ‘Taebaek’ cultivar were planted in 32 hole trays filled with autoclaved artificial soil. Plants were grown in a room at 25.0 ± 1.0°C under a 16/8 h (light/dark) photoperiod. After 4 weeks, leaves were harvested from healthy and uniform seedlings selected from each DH line and kept frozen. Lyophilized tissues were analyzed by high performance liquid chromatography (HPLC). Data were obtained from five seedlings and each seedling was analyzed with three technical replicates.
Analysis of Glucoraphenin and Glucoraphasatin by HPLC
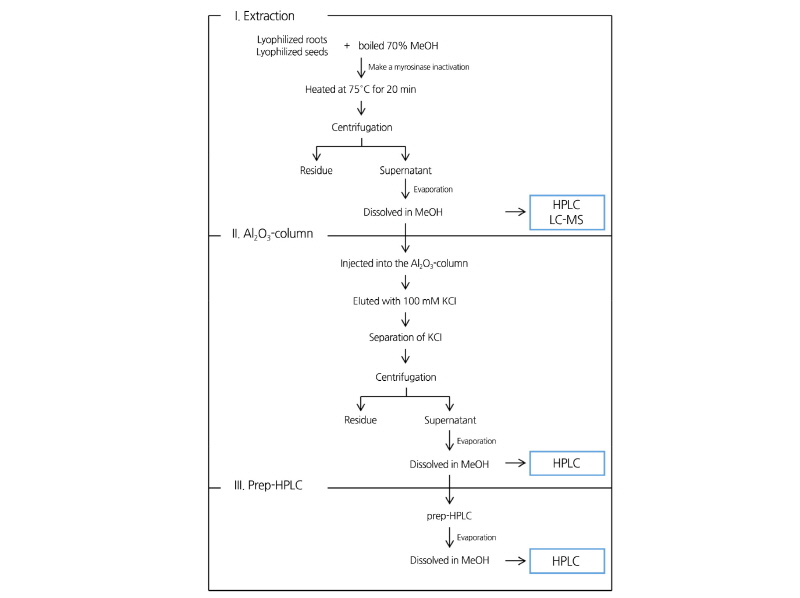
Intact GRE and GRH were extracted as described by Ito and Horie (2008) with a few modifications (Fig. 1). Lyophilized samples were ground in a mortar with 70% methanol. The suspension was incubated at 75.0°C for 20 min to inactivate the hydrolytic enzyme. The samples were centrifuged at 800 × g for 10 min and the supernatants were evaporated to dryness and dissolved in methanol. The methanol extract was filtered by a 0.45 µm polyvinylidene fluoride syringe filter (Acrodisc LC 13 mm syringe filter, Pall, NY, USA). The filtrate was, then analyzed using a Gilson HPLC system with the UV detector set at 229 nm and a C-18 reverse phase column (Zorbax Eclipase XDB-C18, 4.6 × 250 mm i.d., 5 µm particle sizes; Macherey- nagel, Dűren, Germany). The mobile phase solvent system was 65% acetonitrile containing 2.5 mM tetrakis (decyl) ammonium bromide for ion-pairing reagents.
To identify GRE and GRH, HPLC-(ESI) mass spectrometry (MS)/MS analysis was performed using an Accela HPLC system coupled with a mass detector (ATQ velos; Thermo Scientific, Waltham, MA, USA) and a Zorbax Eclipse XDB-C18 column (4.6 × 250 mm, 5 µm; Agilent Technologies, Santa Clara, CA, USA). The capillary and source heater temperatures were 275 and 250°C, respectively. The flow rate was 0.2 mL·min-1 and the mass was scanned from 150 to 2,000 m/z.
Purification of Glucoraphenin and Glucoraphasatin by Al2O3-column and prep-HPLC
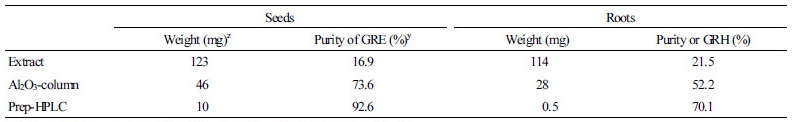
To quantitatively analyze the GRE and GRH content in the DH plants, GRE and GRH were purified from ‘Taebaek’ seeds and roots, respectively, using ion-exchange column and prep-HPLC as outlined in Fig. 1.
The methanol extract of the GSLs were purified using the column chromatography method described by Kuang et al. (2010) with a few modifications. Acidic alumina (100-200 mesh, about 100 g) was washed with deionized water and the slurry was packed into the 500 × 10 mm glass column. Elution was performed using a Spectra/Chrom MR-1 pump (Spectrum Chromatography, Houston, TX, USA) to deliver a constant flow rate of 2.5 mL·min-1. The separation procedure was monitored with a UV-1800 spectrophotometer (SHIMADZU, Kyoto, Japan) at 229 nm. After the extracts were injected into the acidic alumina column, the column was washed exhaustively with deionized water, and then, eluted with 100 mM KCl. The fractions were directly frozen, and the lyophilized powder containing GSLs was dissolved with 100% methanol to remove the KCl. The solution was stored at 4°C to allow the KCl to precipitate. After centrifugation at 800 × g for 10 min, the methanol phase was collected and dried under nitrogen gas and re-dissolved in 100% methanol.
The methanol extract was filtered using a 0.45 µm polyvinylidene fluoride syringe filter (Acrodisc LC 13 mm syringe filter, Pall, NY, USA) before the fractionation using prep-HPLC. The extracts were isolated using a dual Jaigel LC-9104 system with a UV detector set at 229 nm and a JAI GS-310 F column (20 × 300 mm i.d., 3 µm pore size, 40,000 MW exclusion, Japan Analytical Industry Co., Ltd., Tokyo, Japan). 85% methanol was used as an elution solution at a flow rate of 3 mL·min-1.
Statistical Analysis
SigmaPlot 12.0 software (Systat Software Inc., San Jose, CA, USA) used for statistical analysis. All data are presented as the average of the replicates followed by the standard error. The level of significant difference was calculated based on the Student’s t-test. Significance of the correlation coefficient was at the 5% or 1% level.
Results and Discussion
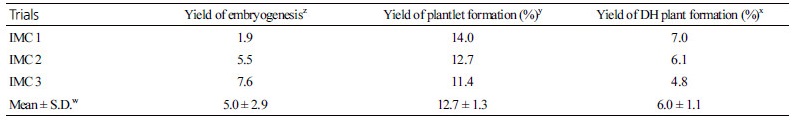
Using the isolated microspores from the radish ‘Taebaek’, globular embryos were obtained very quickly and they developed into green cotyledonary tissues of about 5 mm in length in 2 weeks (Fig. 2A). The embryo yield was 5.0 ± 2.9 per petri dish (Table 2). The cotyledonary embryos in the liquid medium were transferred to MS medium in order to induce hypocotyl and root elongation (Fig 2B). Once a single shoot differentiated from the calli, it become dominant and developed into a complete plantlet (Fig. 2C). The yield of regeneration from the microspore-derived embryos was 12.7% (Table 2). The ploidy level testing showed that the percentage of haploid, DH, tetraploid and uncertain mixed nuclei were 11.6, 45.0, 21.7, and 21.7%, respectively (Supplementary data 1 and 2). Successful induction of embryogenesis from IMC has been reported in several Brassica crops. For example, the induction rate of embryogenesis in broccoli was 30.1 embryos per petri dish and the frequency of plant regeneration was from 65 to 79% depending on the culture conditions (Zeng et al., 2017). However, radish is one of the most difficult species to regenerate by tissue culture (Baillie et al., 1992; Lionneton et al., 2001; Barro et al., 2003; Gu et al., 2003). In the present study, a substantial yield in embryogenesis was obtained with the ‘Taebaek’ cultivar, but little regeneration was achieved with other cultivars including ‘Chungwoon’ (Data not shown). The efficiency of DH production by IMC from ‘Taebaek’ radish plants merits further research, such as comparative study on regulation of GSL biosynthesis in radish. Bud pollination was conducted to maintain the DH microspore-derived radish plants. The DH plants had normal flowers and anthers containing pollen (Fig. 1F), but the haploid and polyploid plants had abnormalities such as flowers without anthers (Fig. 2D and 2E). A total of 41 lines of DH radish plants were obtained from IMC and bud pollination (Fig. 2G, 2H, and 2I).

Fig. 2. Embryogenesis processes from microspore-derived embryos to regenerated plants by isolated microspore culture. Microspore-derived embryos (A), young seedlings with root differentiation and elongation (B), a regenerated young plant transplanted into the soil (C), abnormal flowers (C,D), a normal flower containing pollen (F),flower buds and stigmas for bud pollination (G), formation of the silique 50 d after bud pollination (H), and mature seeds (I).
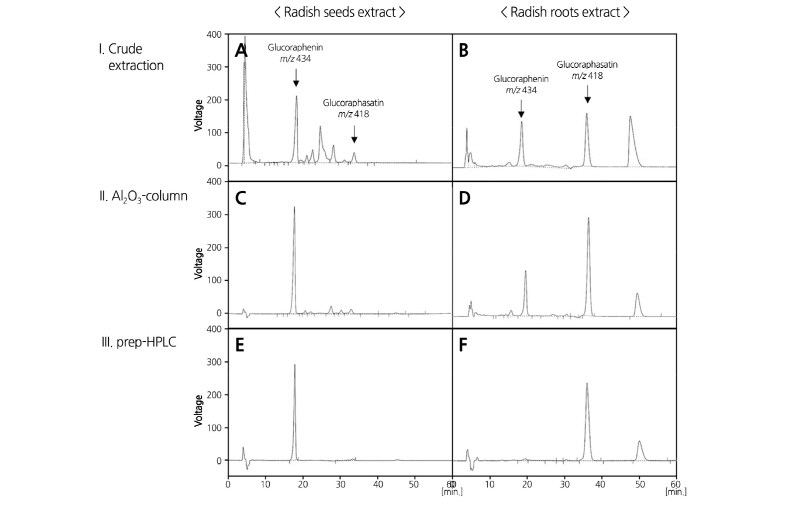
To quantitatively analyze the content of GSLs contents in the DH plants, intact GRE and GRH were extracted and purified by Al2O3-column and prep-HPLC because purified GSLs were not commercially available (Fig. 3). Previous studies showed that radish seeds primarily contain GRE, whereas roots accumulate high levels of GRH (Barillari et al., 2005; Han et al., 2015). Therefore, radish seeds and roots were used for extraction of GRE and GRH, respectively. The HPLC chromatogram of the crude extract of radish seeds and roots is shown in Fig. 3A and 3B, respectively. Two major peaks were detected in both extracts at a retention time of 18 and 34 min. MS analysis was performed to identify the compounds in the two peaks. The molecular mass of the compounds at the 18 and 34 min retention times were 434 and 418 m/z, respectively (Supplementary data 3), which theoretically coincide with GRE and GRH, respectively. The MS data of GRE and GRH indicated the distinguishing fragment peaks with glucose fragments at 257, 259, and 195 m/z. Moreover, peaks at 388, 240, and 176 m/z in GRH and at 419, 354, 301, 291, and 241 m/z in GRE were identified (Clarke, 2010). The ion-exchange column packed on acidic oxide (Al2O3-column) can purify GSLs from crude extracts. In comparison with the sample prior to passage through the Al2O3-column, the purity of GRH was improved from 21.5 to 52.2% and that of GRE up to 73.6% (Fig. 3C and 3D and Table 3). Samples purified by Al2O3-column chromatography were subjected to isolation using prep-HPLC. The purity of the extracted GRE and GRH from radish plants was up to 96.2 and 71.0%, respectively (Fig. 3E and 3F and Table 3), which was high enough to use as a standard. Standard calibration curves were made from concentrations of 0.0625 to 1.0 mg in weight of purified GRE and GRH in accordance with each purity.
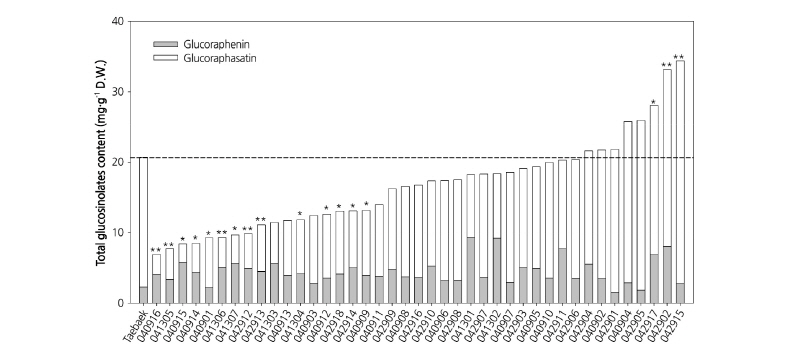
The major aliphatic GSLs in radish, GRE and GRH, can be determined in leaf tissues of DH lines. Kakizaki et al. (2017) suggested that GSLs in radish plant were synthesized in the leaf tissue and accumulated in root tissue by long-distance transport. Therefore, since leaf tissue is the source of active biosynthesis of GSLs, we used young leaf tissue for extracting GSLs from each genotype of the DH lines. The variation of GSL content in the DH lines and the ‘Taebaek’ cultivar is shown in Fig. 4. The results have indicated a variety of segregation of GSLs, and these results are similar to previous studies (Jo et al., 2016). The sum of GRE and GRH contents were highly variable among the DH lines, ranging from 6.9 to 34.4 mg·g-1 dry weight (D.W.). The GRH contents were reduced by as much as 14.1% or increased by up to 171.2% compared with the donor plants (18.4 mg·g-1 D.W.). There was a 6-fold difference in the amount of GRE between the line with the highest GRH content (‘041301’, 9.3 mg·g-1 D.W.) and the line with the lowest GRH content (‘042901’, 1.5 mg·g-1 D.W.). Among the 41 DH lines tested, 14 lines contained significantly reduced contents of GRE and GRH compared with the donor plants (p = 0.95), whereas three lines contained increased levels of GRE and GRH compared with the donor plants. Using quantitative analysis of GRH and GRE contents in a radish breeding program will facilitate the development of new cultivars with improved nutritional value. Additionally, the radish DH lines generated in this study with varying GRE and GRH concentration will be helpful for studying the regulation of the biosynthesis of major aliphatic GSLs in response to various biotic and abiotic stimuli and for elucidating the role of glucoraphasatin synthase 1, responsible for the formation of GRH from GRE.