Introduction
Materials and Methods
Plant Materials and ClO2 with Sucrose Treatment
Evaluation of Vase Life
Antimicrobial Effect
Experimental Design and Data Analysis
Results and Discussion
The Effect of Processing Methods
Applicability Study for Export
Introduction
The cultivation area of roses in Korea is 292 ha, with 744 tons worth $2,250,000 exported in 2016 (Korea Agro-Fisheries and Food Trade Corporation, 2017). Korea exports 98% of its roses to Japan.
The process of exporting roses can generally be divided into two stages: the cultivation stage at the farmhouse and the sorting stage (Rosepia, Deokjin-gu, Jeonju-si, Jeonbuk, Korea). The cultivation stage involves harvesting, storage, and transportation of the cut roses to a cooperative sorting facility. The sorting stage involves length sorting, recutting, and packing (Rosepia, 2018). Cultivated roses are first harvested in the morning and then generally stored in a preservative solution such as Al2O3 (pH 4.5) in Paju-si or 1% Chrysal RVB clear intensive (Chrysal International B.V., The Netherlands) in a rose greenhouse in Jeonju-si. After storage, they are transported wet within 1 hour to a cooperative sorting facility, where they are sprayed with a fungicide upon arrival. The selection, cutting, and packing steps are performed within 1 hour. Plastic packaging export containers are put into a preservative solution (In et al., 2016; Lee et al., 2018). Because cut roses are influenced by water balance after harvest, when it was exported, wet transportation rather than dry transportation is used (Lee et al., 2016; Shi et al., 2019). In the US, a process of grinding, bunching, rehydration, pulsing, packing, and cooling is followed after harvest. In addition, wet transportation involves application of disinfectants such as cyclic acid, 8-hydroxyquinoline citrate, and aluminum sulfate (pH 3.5) (Reid, 2009). It takes 1 to 2 days to transport the cut roses from Korea to Japan. The main factor in determining the vase life of cut roses after harvest is bacteria and especially Botrytis cinerea (Hoogerwerf et al., 1988). To extend vase life, a preservative is used to inhibit bacterial accumulation in the vase solution and stem conduits, and the cut roses are sprayed with a disinfectant to inhibit Botrytis cinerea. Bacteria counts as high as 4.7 × 105 colony-forming units CFU/L have been detected in the holding solution (HS) in reservoirs of sorting places,despite the use of a floral preservative (Lee et al., 2018). Because disinfectants that remain in the waste process are harmful to humans and considered pollutants, environmentally friendly compounds are required (Florack et al., 1996; Williamson et al., 1995).
Chlorine dioxide (ClO2) remains a dissolved gas in water (Macnish et al., 2008). It is decomposed easily by light (Junli et al., 1997) and is commonly used as an alternative disinfectant in drinking water because it produces low concentrations of regulated trihalomethanes (THMs), which are harmful to human health (Rouge et al., 2018). ClO2 has therefore been listed by the World Health Organization (WHO) as a safe disinfectant (Fang et al., 2016). The antimicrobial effects of ClO2 on cut flowers have been studied extensively. The 2 - 8 µL·L-1 ClO2 in the vase solution was found to be consumed in 2 to 4 days, but the sterilization effect was sustained for 6 days or longer after treatment, and a pulsing solution (PS) treatment of 50 - 250 µL·L-1 ClO2 for 1 min prolonged the vase life of cut roses (Lee and Kim, 2014). An HS treatment of 5 - 10 µL·L-1 ClO2 also prolonged the vase life of cut flowers (Macnish et al., 2008). The sterilization effect of 2 - 8 µL·L-1 ClO2 with 2% sucrose was sustained for 6 days or longer after treatment and extended the vase life of roses (Lee and Kim, 2018).
Ichimura et al. (1999) proposed that treatment with disinfectants and sucrose is more effective at extending vase life than disinfectants alone, and a high positive correlation was obtained between vase life and total soluble carbohydrate concentrations in petals. A sucrose HS treatment shortened the vase life of roses and resulted in more bacterial accumulation in the preservative solution than did tap water (TW) and distilled water (DW) (Lee and Kim, 2018).
The purpose of this study was to investigate the possibility of using a ClO2 solution as a preservative for the export of roses from Korea and to identify the treatment effects of ClO2 and sucrose, separately and combined, in an HS and PS, separately and combined.
Material and Methods
Plant Materials and ClO2 with Sucrose Treatment
Cut roses (Rosa hybrida ‘Beast’) were harvested in the commercial harvest phase from a greenhouse in Paju, Korea. Cut roses with a stem diameter between 7 and 9 mm were used. Stems were transported within 1 hour to a post-harvest laboratory at the University of Seoul. ClO2 (CA-200, PurgoFarm Inc., Korea) was used with DW. Post-harvest treatments were divided into HS, PS, and PS + HS treatments. The HS treatments used tap water (TW), distilled water (DW), 2% sucrose, 4 µL·L-1 ClO2, 4 µL·L-1 ClO2 with 2% sucrose (Lee and Kim, 2014; Lee and Kim, 2018), and 200 µL·L-1 8-HQS with 2% sucrose (Inchimura et al., 1999). The PS treatment was carried out in 50 µL·L-1 ClO2 for 10 seconds at the end of the stem (Lee and Kim, 2014). PS + HS treatment involved pulsing with 50 µL·L-1 ClO2 for 10 seconds and then the flowers were placed in 4 µL·L-1 ClO2 with 2% sucrose until the end of vase life.
To test the effect of ClO2 on cut rose cultivars, the stems were recut to 40 cm in length, and all but three leaves were removed. The pulsing treatment was performed on 10 replicates per 400 mL of DW. To test applicability for export, the stems were recut to 50 cm in length, and all but 7 or 8 leaves were removed. When cut roses are exported to Japan, it typically takes 1 - 2 days to travel from the greenhouse to the cooperative sorting and packing facility in Korea and then 3 - 4 days to reach the auction house in Japan (Rosepia, 2018). Therefore, the cut roses were divided into PS and HS treatments of 2 days and 4 days in export containers (20 × 20 × 10 cm), using 30 stems for each treatment. After 6 days, each rose was recut to a 40 cm stem length with three leaves, and 10 replicates were placed in 400 mL of DW.
Evaluation of Vase Life
Vase life was recorded as the duration (days) after harvest (day 0) until the flowers began to exhibit vase life termination symptoms, such as wilting petals or leaves (50% turgor loss), a petal and leaf abscission (dropping one petal and leaf), or bending of the peduncle (bent neck) by a neck angle greater than 30° (Fanourakis et al., 2013). The relative fresh weight (RFW, %) and vase solution uptake (mL·d-1·g-1 FW) were calculated every 2 days using Jowkar’s (2015) formula.
Antimicrobial Effect
The antimicrobial effect was confirmed by the ClO2 concentration and bacteria in the vase solution using HI 93738 ISM ClO2 analysis equipment (Hanna Instruments Inc., USA) and chlorine dioxide Insta-test (LaMotte Instruments Inc., USA). To find the total number of aerobic bacteria in the vase solutions, 1 mL of the samples was taken from each vase and serially diluted 101 - 106-fold in sterile DW. Aliquots (0.1 mL) of the diluted samples were pipetted onto agar (NA, DifcoTM). Bacteria on the agar were incubated for 3 days at 28°C, and bacterial count was recorded as CFU/L (van Doorn et al., 1989).
Experimental Design and Data Analysis
The vase life experiment followed a completely randomized block design with 10 replicates. Analysis of variance was used to analyze the data, and Duncan’s new multiple range test (DMRT, p = 0.05) was performed as post hoc analysis. Statistical analysis was conducted using SAS software (Statistical Analysis System, version 9.2, SAS Institute Inc., Cary, NC, USA).
Results and Discussion
The Effect of Processing Methods
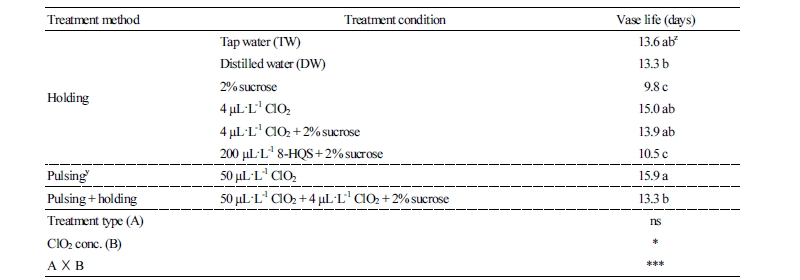
This experiment was carried out to investigate the treatment effect on ‘Beast’ of pulsing and holding solutions, alone or in combination with ClO2 and sucrose, and compared with 8-HQS + sucrose, which has been used in previous studies (Inchimura et al., 1999). As a result, an HS of 4 µL·L-1 ClO2 and 4 µL·L-1 ClO2 + 2% sucrose and a PS of 50 ClO2 µL·L-1 were extended on vase life by 0.3 to 2.6 days compared with controls (TW and DW), but the effect was not statistically significant (Table 1). It was found that the vase life of cut roses depends on early sterilization. Bacteria in the reservoir of the greenhouse and sorting facilities were detected at a level of 2.8 to 4.7 × 105 CFU/L despite the use of preservatives (Lee et al., 2018). In this experiment, ClO2 in one stem was treated in clean DW (Fig. 2A), and bacteria were not detected in the vase solution on day 0. Therefore, the extended vase life effect of ClO2 was statistically not significant (Table 1). Macnish et al. (2008) found that the vase life extension from adding 10 µL·L-1 ClO2 in a vase solution containing 1011 CFU/Lbacteria, which usually caused vascular occlusion and wilting in Rosa (van Doorn et al., 1995), was higher than that of 100 CFU/Lbacteria. Clean DW treatments prolonged the vase life of cut roses (Macnish et al., 2008). Therefore, it is expected that an HS of ClO2 and ClO2 + sucrose containing bacteria may prolong the vase life of cut roses. A PS + HS and HS of sucrose did not prolong vase life (Table 1). Including carbohydrates such as sucrose and glucose in the vase solution delayed the senescence of cut flowers (Eason et al., 1997). A PS treatment (for 6 to 12 h) of sucrose or 10 to 20% sucrose also extended the vase life of cut flowers (Finger, 2001; Alkal et al., 2005). Therefore, when processing using sucrose alone to prolong vase life, a short pulsing treatment would be better than an HS treatment for an extended time.
In the experiment with ClO2, an HS of ClO2 and ClO2 + sucrose did not statistically extended vase life, and vase life with a PS was numerically longer than an HS and PS + HS. An HS of 2% sucrose and 200 µL·L-1 8-HQS + 2% sucrose shortened vase life contrary to previous studies that found such an HS extended vase life (Inchimura et al., 1999).
Eason (2002) found that an HS of 20 g sucrose + 0.84 g sodium hypochlorite shortened the vase life of cut flowers and caused early browning of leaves, an effect that was not seen with sucrose alone. In this experiment, it was assumed that treatment with 8-HQS is toxic to cut roses and produces browning of stems, as reported by Eason (2002) and Knee (2000). We found that the treatment solution type was not significant, but a factor analysis showed ClO2 concentration was significant.
Initial weight was maintained for 6 days after treatment with an HS of 8-HQS + sucrose and sucrose; for 8 days in TW, DW, a PS of 50 µL·L-1 ClO2, and a PS of 50 µL·L-1 ClO2 + HS of 4 µL·L-1 ClO2 + sucrose; and for 10 days in an HS of 4 µL·L-1 ClO2 and ClO2 with sucrose (Fig. 1A). Weight was relatively high with PS + HS, HS of ClO2 + sucrose, and ClO2 6 days after treatment, at which point vase life was terminated, but with an HS of sucrose and 8-HQS + sucrose, it was low and was higher in combined treatment with sucrose than in ClO2 treatment. The sucrose with bactericide maintained a high relative weight (Ichimura et al., 1999). After 6 days of treatment, the vase solution uptake was higher in a PS and an HS of ClO2 than in HS of sucrose and 8-HQS (Fig. 1B). These results were similar in that the HS of disinfectant with sucrose resulted in higher fresh weight and lower water uptake than that of DW and 8-HQS (Ichimura et al., 1999).
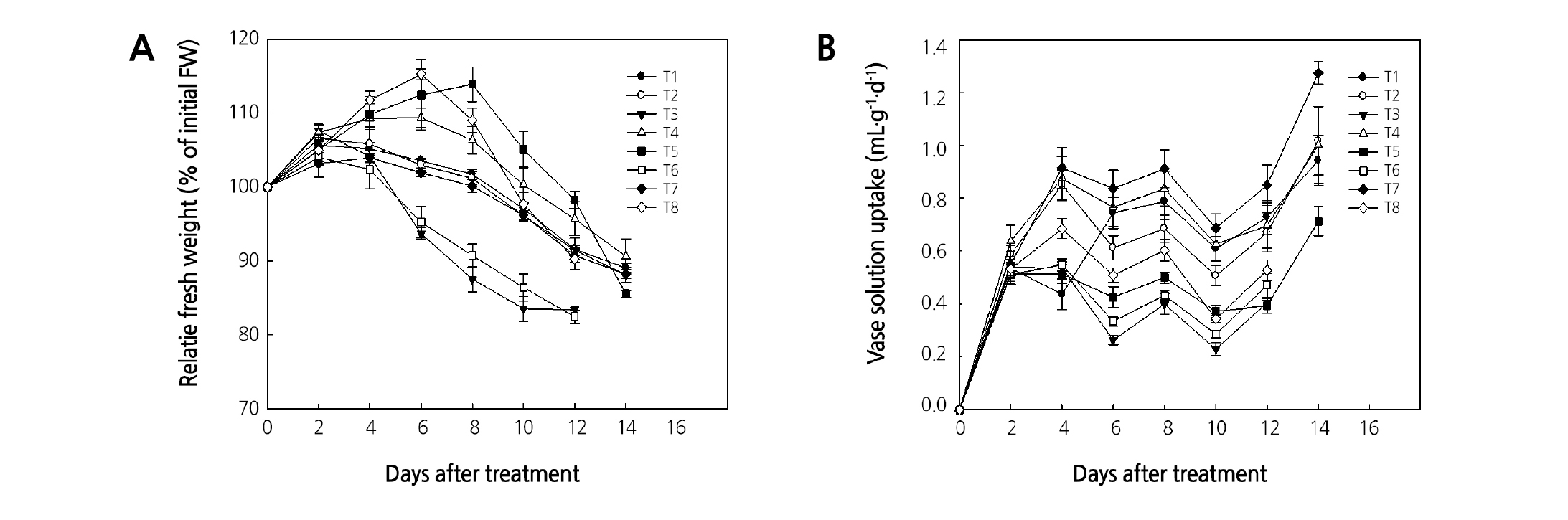
Fig. 1. Effects of chlorine dioxide at different treatment conditions on the relative fresh weight (A) and the vase solution uptake (B) of cut rose ‘Beast’ flowers. T1-T6: holding solution treatment, T7-T8: pulsing treatment. In detail, T1: tap water (TW), T2: distilled water (DW), T3: 2% sucrose in DW, T4: 4 µL·L-1 ClO2 in DW, T5: 4 µL·L-1 ClO2 + 2% sucrose in DW, T6: 200 µL·L-1 8-HQS + 2% sucrose in DW, T7: pulsed in 50 µL·L-1 ClO2 for 10 seconds and then replaced in DW, and T8: T7 with T5. The test room conditions were controlled with air temperature at 24.4°C, relative humidity at 23.6%, and light intensity at 5.8 µmol·m-2·s-1 PAR with a 12-hour photoperiod. Vertical bars indicate the standard error (n = 10).
The results of HS without the stems indicated that bacteria were continuously detected in an HS of TW, DW, and sucrose by 1.4 × 104, 3.8 × 104, and 1.4 × 105 CFU/L,respectively, 6 days after treatment, but not an HS of ClO2, ClO2 + sucrose, and 8-HQS + sucrose (Fig. 2A). Bacteria in an HS with the stems were detected in TW, DW, and sucrose by 8.0 × 104, 1.4 × 105, and 6.8 × 105 CFU/L, respectively, 6 days after treatment, but the sterilization effect of ClO2, ClO2 + sucrose, and 8-HQS + sucrose continued until 8 days after treatment (Fig. 2B). ClO2 can penetrate the cell membrane of bacteria and destroy them in water (Junli et al., 1997). Bacteria were not detected in all ClO2 treatments on the day after treatment and, it was higher with a stem than without (Fig. 2). The 2 to 4 µL·L-1 ClO2 in the vase solution was exhausted without a stem and on days 2 - 4 and 2, respectively (Lee and Kim, 2014). The bactericidal effect and consumption of ClO2 in the vase solution was different because of the presence of stems.
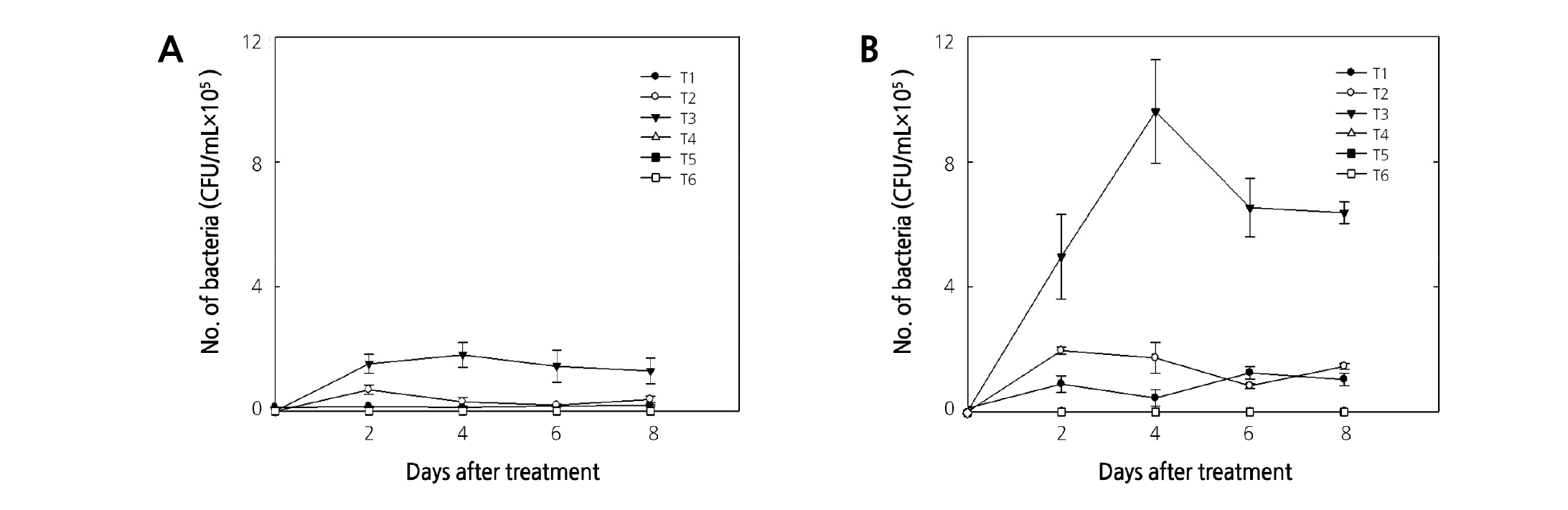
Fig. 2. Changes in the number of bacteria in the holding solution without stems (A) or with stems (B) of cut rose ‘Beast’ flowers at different treatment conditions. T1: tap water (TW), T2: distilled water (DW), T3: 2% sucrose in DW, T4: 4 µL·L-1 ClO2 in DW, T5: 4 µL·L-1 ClO2 + 2% sucrose in DW, T6: 200 µL·L-1 8-HQS + 2% sucrose in DW. Test room conditions were controlled with air temperature at 24.4°C, relative humidity at 23.6%, and light intensity at 5.8 µmol·m-2·s-1 PAR with a 12-hour photoperiod. Vertical bars indicate the standard error (n = 10).
We have previously shown that an HS of ClO2 and ClO2 + sucrose inhibited bacteria in the vase solution for 6 days after treatment, although the ClO2 concentration in the vase solution was exhausted by day 1 - 2 (Lee and Kim, 2018). This study confirmed that the bactericidal effect of ClO2 and ClO2 + sucrose continued until 8 days after treatment.
Applicability Study for Export
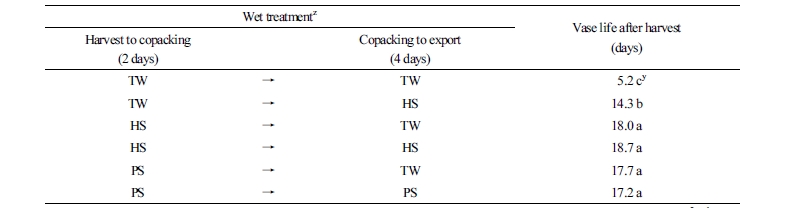
To apply ClO2 treatment during the export process for cut roses, an HS of 4 µL·L-1 ClO2 + 2% sucrose or a PS of 50 µL·L-1 ClO2 for 10 seconds on cut rose ‘Beast’ flowers was applied to export containers. The vase life of roses in HS + HS, HS + TW, PS + TW, PS + PS, and TW + HS was 18.7, 18.0, 17.7, 17.2, and 14.3 days, respectively (Table 2). Treatment with ClO2 extended vase life by 9.1 - 12.8 days, compared with TW + TW (5.2 days), regardless of the treatment method.
Relative fresh weight was lowest in the TW + HS treatment after 2 days; roses had a relatively short vase life among the ClO2 treatments and did not maintain initial relative fresh weight (Fig. 3). Among ClO2 treatments, HS + HS was the highest and maintained initial relative fresh weight, and TW + HS was the lowest 6 days after treatment.
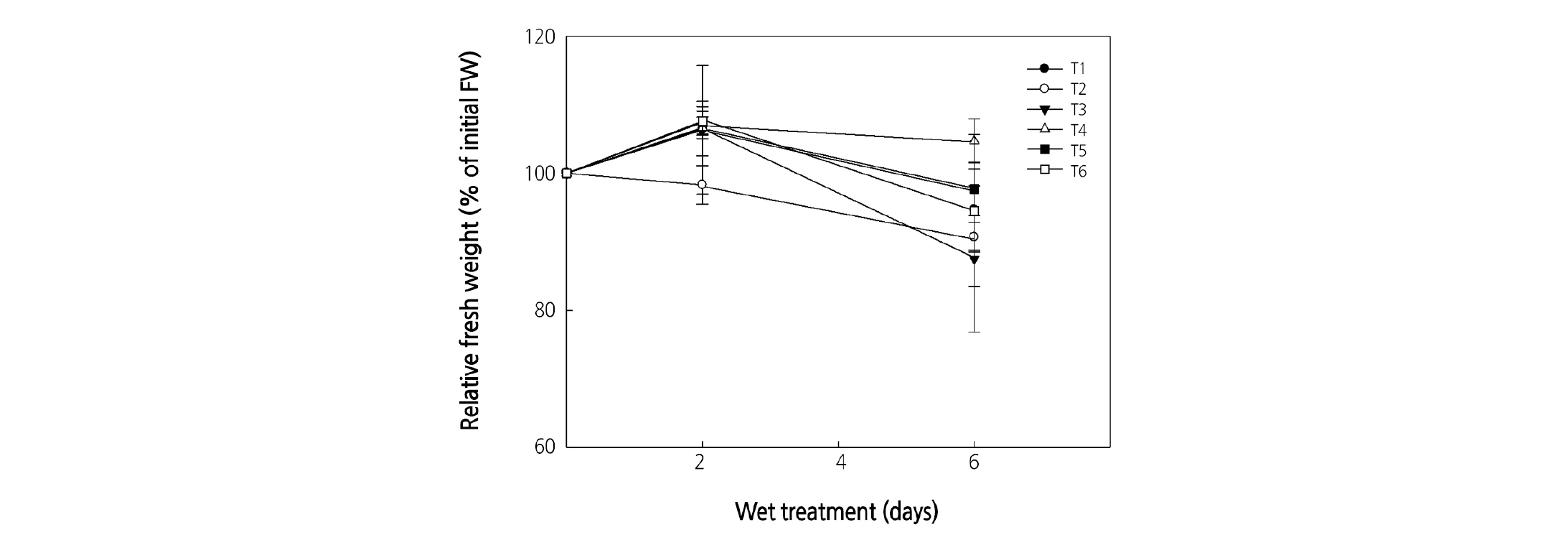
Fig. 3. Effect of chlorine dioxide as the wet treatment on the relative fresh weight of cut rose ‘Beast’ flowers during export transportation (6 days). Export of cut roses was separated by two phases: the first phase involved farming collection and transportation to the packing house, and the second phase was shipping for export. Each phase was treated by a different wet condition. T1: tap water (TW) + TW, T2: TW + holding solution of 4 µL·L-1 ClO2 + 2% sucrose in DW (HS), T3: HS + TW, T4: HS + HS, T5: pulsed in ClO2 50 µL·L-1 by 10 seconds and then replaced in DW (PS) + TW, T6: PS + PS. Test room conditions were controlled with air temperature at 19.6°C, relative humidity at 23.4%, and light intensity at 16.2 µmol·m-2·s-1 PAR with a 12-hour photoperiod. Vertical bars indicate the standard error (n = 30).
Alkal et al. (2005) found that a double pulsing treatment of disinfectant for 1 hour, followed by sucrose for 12 hours, extended vase life and increased water uptake and relative fresh weight. It was found that it was important to use the fungicide and sucrose treatment at the proper time, with initial sterilization in the vase solution.
In conclusion, to apply ClO2 in the export processing of cut roses, an HS of 4 µL·L-1 ClO2 + 2% sucrose and a PS of 50 µL·L-1 ClO2 for 10 seconds were effective for the 2 days after harvest, and HS + HS maintained both the sterilization effect in the vase solution and high relative fresh weight.