Introduction
Materials and Methods
Seed Collection and General Procedures
Effects of Temperature and Light on Seed Germination
Effects of GA3 on Seed Germination
Morphological Observation of Embryo Growth and Radicle Emergence
Statistical Analyses
Results
Morphology of Embryo and Seed
Effects of Temperature and Light on Seed Germination
Effects of GA3 on Seed Germination
Discussion
Introduction
Seed dormancy is considered to be one of the characteristics of seeds that determine the timing of germination and seedling establishment (Vandelook and Van Assche, 2008). Dormancy also plays a critical role in the plant life cycle because seeds represent the crucial link for species to persist across different locations and over time (Harper, 1977). A seed that does not germinate within 30 days under favorable physical conditions (temperature, light, etc.) is considered to be dormant (Baskin and Baskin, 1998). Dormancy-breaking and germination requirements are often species-specific and related to differences in the species’ habitat preferences (Vandelook et al., 2008). In many plant species from temperate climates, seeds are dormant at the time of dispersal from the mother plant, and specific temperature requirements must be reached before they overcome dormancy (Baskin and Baskin, 1998).
An exquisite and experimentally useful classification system for seed dormancy has recently been proposed by Baskin and Baskin (2004). According to Baskin and Baskin (2004), seed dormancy can be categorized into five classes [physio-logical (PD), morphological (MD), morphophysiological (MPD), physical (PY), and combinational dormancy (PY + PD)]. In temperate regions, many perennial herbs disperse seeds with underdeveloped embryos that must elongate to a critical length prior to radicle emergence (Baskin and Baskin, 1998). Seeds with an underdeveloped embryo have either MD or MPD (Baskin and Baskin, 1998, 2004). Seeds have MD if embryo growth and germination of seeds are completed within 30 days under suitable incubation conditions without any dormancy-breaking pretreatment. However, if the seeds require more than 30 days and a dormancy-breaking treat-ment, such as warm and/or cold stratification, to germinate, they are classified as MPD (Baskin and Baskin, 1998; Nikolaeva, 1977). Nine levels of MPD have been proposed related to the requirements for breaking seed dormancy, the temperature requirements for embryo elongation and response to gibberellic acid (GA3) (Baskin and Baskin, 1998, 2004; Baskin et al., 2008).
The genus Heloniopsis (Melanthiaceae) includes nine species; eight of them are distributed throughout Asia and the other is found in North America (Tanaka, 1998). The Asian species are mainly distributed in Japan, Taiwan, Korea, and southern Sakhalin (Fuse et al., 2004). Heloniopsis are typically ever-green perennial herbs that grow in moist, well-drained soils rich in organic matter under mesic temperate deciduous forests (Ahn and Lee, 1997; Cha et al., 2002; Kawano et al., 2007). These species have evergreen rosette leaves that turn purple in the winter and rose-purple, hermaphroditic flowers that bloom in the early spring with great potential for ornamental purposes (Kawano et al., 2007). In addition, the whole plant extract from Heloniopsis orientalis has been shown to have potent cytotoxic and antiproliferative activities as an antitumor agent (Min et al., 2002; Panda et al., 2012). Two new species of Heloniopsis endemic to Korea, H. tubiflora and H. koreana, have recently been described based on morphological traits and molecular phylogeny (Fuse et al., 2004). These two species are restricted to altitudes above 1,300 m in the Korean Peninsula (Fuse et al., 2004) and have recently been evaluated as sensitive species due to climate change in which the flowering date of H. koreana was 22 days faster than in the previous year (Kim et al., 2011). Miller-Rushing and Primack (2008) reported that the flowering time of early flowering species change more rapidly in response to climatological factors than do late flowering species. To our knowledge, efforts towards understanding the ecological requirements of these species, including seed dormancy and germination, have been very limited. Understanding germination requirements is important for propagation and conservation practices.
Very little is known about seed dormancy and germination in the Melanthiaceae. According to Copete et al. (2011), seed dormancy has only been identified in five species. Embryos in seeds of Melanthiaceae have been observed to be small in relation to the size of the endosperm (Baskin and Baskin, 1988; Baskin et al., 2001; Kondo et al., 2011). In Trillium camschatcense, seeds had underdeveloped embryos at dispersal and took more than 1 year before radicle emergence occurred in the field conditions, and were therefore classified as having MPD (Kondo et al., 2011). It has also been observed that seeds of several species such as T. flexipes, T. sessile (Baskin and Baskin, 1988), and Chamaelirium luteum (Baskin et al., 2001) had MPD. Takahashi (1984) reported that 66.7% of the seeds of Heloniopsis orientalis collected from central Japan had germinated within 30 days of incubation at 20°C with 1,500 lux light. Copete et al. (2011) inferred that the seeds of H. orientalis have MD in response to the available infor-mation on germination and on characteristics of seeds in Melanthiaceae. However, none of these authors reported detailed observations on the morphology of embryo growth in Heloniopsis.
To the best of our knowledge, no detailed studies have been conducted to determine whether seeds of Heloniopsis species (H. koreana and H. tubiflora) have an underdeveloped embryo, and if so, what the temperature requirements are for embryo growth and germination. Therefore, the objectives of this study were to determine whether seeds have MD or MPD, and to determine the level of MPD if they have MPD. Specifically, we investigated the effects of the typical triggers of vegetation gaps (temperature and light) on embryo growth and germination. In addition, we determined the effects of GA3 on dormancy-break and germination. This is the first report on embryo growth morphology in Heloniopsis species.
Materials and Methods
Seed Collection and General Procedures
Mature fruits (captures) from H. koreana (Fig. 1A) were collected on 16 May and 20 May in 2012 and on 25 May and 6 June in 2013 from plants growing in the eco-garden within the Hantaek Botanical Garden (37°09´N, 127°40´E), Yongin-si, Korea. Mature fruits of H. tubiflora (Fig. 1B) were collected from plants growing in Hyangjeokbong (35°51´N, 127°44´E), Mt Deogyu, Muju-gun, Korea on 18 June 2012. Fruits (Figs. 2A and 2B) were allowed to dry in laboratory conditions (20-25°C, 8-11 µmol・m-2・s-1 light) for 2 weeks, then packed in sealed plastic bags and stored dry at 4°C until the beginning of the experiment in July.
Three replicates of 30 seeds each were used for all laboratory experiments. The seeds were placed on two sheets of filter paper (Whatman No. 2, GE Healthcare Co., Ltd., Buckinghamshire, UK) in 90 × 15 mm Petri dishes and moistened with distilled water. All dishes were wrapped with parafilm to restrict water loss during incubation. At all temperature regimes, a 12-h light/dark photoperiod was provided by a cool white fluorescent lamps that provided a photon flux density of approximately 8-11 µmol・m-2・s-1 in the incubators (DS-13MCLP, Dasol Scientific Co., Ltd., Hwaseong, Korea). Radicle emergence was monitored weekly to calculate percent germination. Seeds were considered “germinated” when radicles emerged at least 1 mm.
Effects of Temperature and Light on Seed Germination
Seeds of H. koreana and H. tubiflora collected on 16 May 2012 and 18 June 2012 were used for temperature and light experiments, respectively. Seeds were incubated at 12 h/12 h alternating temperature regimes of 15/6°C, 20/10°C, 25/15°C, and 30/20°C and at a constant temperature of 5°C. Seeds in another three Petri dishes were wrapped with two layers of aluminum foil and incubated at 25/15°C to create a dark treatment. Radicle emergence was recorded weekly for 8 weeks and results were expressed as mean percent germination.
Effects of GA3 on Seed Germination
Seeds of H. koreana collected on 20 May 2012 and 6 June 2013 and seeds of H. tubiflora collected on 18 June 2012 were used for the GA experiment. The seeds collected in 2012 were soaked in 0, 10, 100, or 1,000 mg・L-1 GA3 for 24 h at room temperature (20-25°C) and then incubated at 15/6°C and 25/15°C in a 12-h light/dark photoperiod. In H. koreana, seeds collected on 6 June 2013 were placed on two sheets of filter paper (Whatman No. 2, GE Healthcare Co., Ltd., Buckinghamshire, UK) in 90 × 15 mm Petri dishes and moistened with solutions of 0, 10, 100, or 1,000 mg・L-1 GA3 before incubation. Radicle emergence of all treatments was recorded weekly and percent germination was calculated after 4 weeks of incubation.
Morphological Observation of Embryo Growth and Radicle Emergence
During incubation, freshly imbibed seeds were cut into thin sections using a razor blade, and the length of seeds and embryos was measured under a dissecting microscope fitted with an ocular micrometer (KSZ-1B, Samwon Scientific Co., Ltd., Seoul, Korea). To determine embryo elongation, 10 seeds incubated at 25/15°C were randomly selected over a 4-week period, and the length of embryos (before radicle emergence) was measured. Embryo elongation was expressed as a ratio of embryo length to seed length (E:S ratio) (Vandelook et al., 2007). After measuring the length of seeds and embryos using the dissecting microscope, the sections of the seeds were viewed at 60 to 120 × magnification and photographed with a Miview USB digital microscope (MV 1302U, CosView Technologies Co., Ltd., Shenzhen, China).
Statistical Analyses
This experiment was conducted with a completely rando-mized block design with three replications. Final percentages of germination in each experiment were analyzed statistically using the GLM procedure of SAS program (SAS Institute Inc., Cary, NC, USA). Means were compared using Tukey’s studentized rage (HSD) test at the 5% level to compare treatment differences within the germination data.
Results
Morphology of Embryo and Seed
Seeds of H. koreana and H. tubiflora had caudal appendages at each end at the time of dispersal. Mean seed lengths of H. koreana and H. tubiflora with caudal appendage were 5.7 ± 0.11 mm and 4.8 ± 0.30 mm, respectively (Figs. 2C and 2D). However, mean lengths of the endosperm in seeds without caudal appendages were 1.3 ± 0.04 mm and 1.4 ± 0.07 mm, respectively (Figs. 2E and 2F).
In H. koreana, freshly matured seeds had small embryos (Fig. 2E). The mean length of the embryos was 0.12 ± 0.005 mm. Thus, the ratio of embryo to true seed length (E:S ratio) was 0.09. Embryo length before radicle emergence increased to 0.85 ± 0.095 mm (Fig. 2G). Therefore, both embryo length and E:S ratio increased to more than 5 times their original values in newly matured seeds.
In H. tubiflora, freshly matured seeds had small embryos (Fig. 2F). The mean length of the embryos was 0.16 ± 0.003 mm. Thus, the E:S ratio was 0.11. Embryo length before radicle emergence increased to 0.63 ± 0.010 mm (Fig. 2H). Therefore, both embryo length and E:S ratio increased to more than 4 times their values in freshly matured seeds.
The mass of seeds increased during incubation regardless of species, indicating an uptake of water. Water moved from the outside of the seed coats to the inside of the endosperm through caudal appendages in both species (data not shown).
Effects of Temperature and Light on Seed Germination
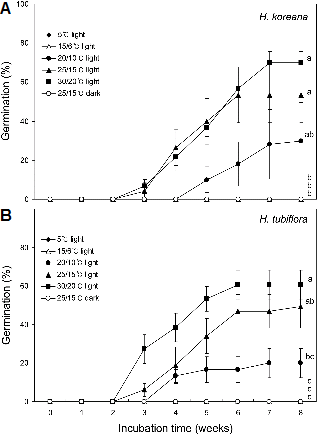
Fresh seeds of H. koreana incubated at 25/15°C and 30/20°C under 12-h alternating light conditions germinated to 26% and 22%, respectively, in 4 weeks (Fig. 3A). Extending the incubation time to 8 weeks increased seed germination to 70% at 30/20°C, 53% at 25/15°C, and to 30% at 20/10°C. However, there was no significant difference in the final percent germination between the 25/15°C and 30/20°C incubation groups. The optimal temperatures for seed germination were found to be 25/15°C and 30/20°C in H. koreana. No seed germination was recorded at 5°C and 15/6°C under alternating light conditions or at 25/15°C under constant dark conditions (Fig. 3A).
Fresh seeds of H. tubiflora that were incubated at 20/ 10°C, 25/15°C, and at 30/20°C under 12-h alternating light conditions resulted in 13%, 18%, and 38% germination, respectively, over 4 weeks. Extending the incubation time to 8 weeks increased seed germination to 60% at 30/20°C, 49% at 25/15°C, and 20% at 20/10°C, but there was no significant difference in percent germination between 25/ 15°C and 30/20°C temperature conditions (Fig. 3B). The optimal temperatures for seed germination were found to be 25/15°C and 30/20°C in H. tubiflora. No seed germination was recorded at 5°C and 15/6°C under 12-h alternating light conditions or at 25/15°C under constant dark conditions (Fig. 3B). It should be noted that seeds of these two Heloniopsis species turned green at the time of radicle emergence (Figs. 2I and 2J). Seedling development after radicle emergence was also observed (Figs. 2K and 2L).
Effects of GA3 on Seed Germination
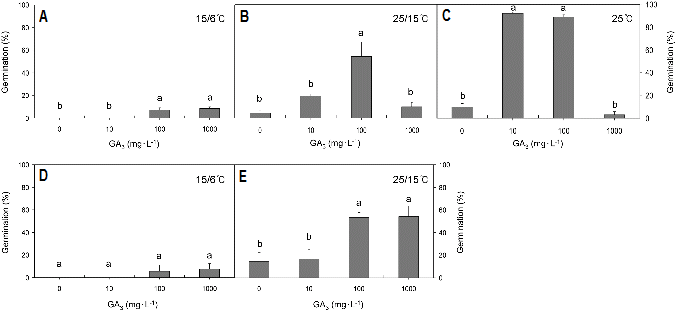
Germination responses to various concentrations of GA3 and to various incubation temperature regimes varied significantly in both species (p < 0.05). In seeds of H. koreana, the addition of GA3 at 15/6°C, 25/15°C, or 25°C significantly improved the percent germination after 4 weeks of incubation (Fig. 4). However, an increasing effect on percent germination was significantly different depending on the incubation temperatures (p < 0.05). In the seeds incubated at 15/6°C, the percent germination increased from 0 to 8.6% as the concentration of GA3 increased from 0 to 1,000 mg・L-1 (Fig. 4A), whereas in the seeds incubated at 25/15°C, the percent germination increased from 4.2 to 54.4% with increasing GA3 concentration from 0 to 100 mg・L-1. There was no statistical difference between 0 and 1,000 mg・L-1 GA3 (Fig. 4B). At a constant temperature of 25°C, percent germinations were 92.0% and 88.9% for 10 and 100 mg・L-1 GA3, respectively (Fig. 4C).
In seeds of H. tubiflora incubated at 15/6°C, percent germination increased from 0 to 7.6% as the concentration of GA3 increased from 0 to 1,000 mg・L-1 (Fig. 4D). In the seeds incubated at 25/15°C, percent germination increased from 14.5 to 54.2% as the concentration of GA3 increased from 0 to 1,000 mg・L-1. No significant difference was observed between 100 and 1,000 mg・L-1 GA3 (Fig. 4E).
Discussion
In temperate regions, many herbaceous plant species have small, underdeveloped embryos in seeds at the time of dispersal (Baskin and Baskin, 1988). Mature seeds from H. koreana and H. tubiflora had small embryos, and the length of embryos increased more than 300% before radicles emerged. According to Baskin and Baskin (1998), rudimentary or small, linear embryos must elongate before germination occurs; thus, the seeds generally are referred to as having underdeveloped embryos. Previous studies showed that seeds of Melanthiaceae had small, underdeveloped embryos (Baskin and Baskin, 1988; Baskin et al., 2001; Kondo et al., 2011; Martin, 1946). Since considerable embryo elongation occurred in seeds of the two Heloniopsis species before radicles emerged, we have confirmed that the seeds have underdeveloped embryos.
Underdeveloped embryos in seeds with MD are not physiologically dormant, and the seeds will typically germi-nate within 30 days. However, the embryos with MPD are dormant at the time of dispersal and need more than 30 days for germination (Baskin and Baskin, 1998, 2004; Nikolaeva, 1977). The fact that embryos in freshly mature seeds of the two Heloniopsis species are small and under-developed implies that the seeds are morphologically or morphophysiologically dormant at dispersal. In this study, approximately 30% of the H. koreana seeds and appro-ximately 40% of the H. tubiflora seeds germinated at favorable conditions in 4 weeks. Therefore, approximately 30% of H. koreana and 40% of H. tubiflora have MD, and approximately 70% and 60% of them, respectively, have MPD. The seeds of the two species studied showed a large variation in time to germination, independent of incubation temperatures. Seeds in the population that continued to germinate after 30 days were probably in various states of MPD, and thus needed different lengths of time at favorable temperatures and light to germinate (Adams et al., 2005; Baskin and Baskin, 2004). It is suggested that such a delay mechanism within the seed population can be an ecologically advantageous strategy for unpredictable environmental conditions (Alves- Da-Silva et al., 2011; Doussi and Thanos, 2002).
Embryo growth in the seeds of the two species was promoted under relatively high temperatures (25/15°C and 30/10°C). Based on temperatures at the time of embryo elongation, seeds with MPD have been divided into two categories: simple and complex (Baskin and Baskin, 1998, 2004). Embryo growth occurs at relatively warm tempera-tures (≥ 15°C) in simple MPD; whereas, in complex MPD, embryo growth occurs at low temperatures (0-10°C) (Baskin and Baskin, 1998, 2004). Percent germination of the two Heloniopsis species was higher at relatively warm temperature regimes (25/15°C and 30/20°C) than at low temperature regimes (5°C and 15/6°C) (Fig. 3), indicating that the seeds of the two species have simple MPD.
Each type of MPD (simple or complex) can be subdivided into non-deep, intermediate, and deep MPD depending on the physiological states of seeds (Baskin and Baskin, 1998, 2004; Nikolaeva 1977). The dormancy of seeds with non-deep dormancy can be broken by warm or cold stratification, and GA can be used to overcome the dormancy (Baskin and Baskin, 1998). In this study, warm stratification increased percent germination, and GA3 overcame dormancy in seeds of H. koreana and H. tubiflora. Furthermore, embryos of these two species grew better in warm (rather than cold) stratification. Therefore, we have confirmed that seeds of these two species have non-deep simple MPD. These results are similar to those reported by Walck et al. (1999) on seeds of Thalictrum mirabile, by Hidayati et al. (2000) on seeds of Lonicera morrowii, and by Chen et al. (2013) on seeds of Nageia nagi.
The kind of dormancy observed in H. koreana and H. tubiflora seeds is well-represented by the formula presented by Nikolaeva (2001) for non-deep simple MPD, C1bB-C1b (Baskin and Baskin, 2008). These seeds need a period of warm (C1b) temperatures to break physiological dormancy (PD) and for the growth of an underdeveloped embryo (B). After MPD (C1bB) is broken, the seeds, now with a fully developed embryo, germinate at warm temperature regimes (-C1b).
Many species in the order Liliales have MD and/or MPD in seeds at the time of dispersal (Copete et al., 2011). In particular, seeds of Melanthiaceae have been reported to express non-deep simple, deep simple epicotyl, and deep simple double MPD in Chamaelirium luteum (Baskin and Baskin, 2001), Trillium flexipes (Baskin and Baskin, 1988), and Trillium camschatcense (Kondo et al., 2011), respectively. Takahashi (1984) reported that seeds of Heloniopsis orientalis were little dormant, and germinated to 10%, 66.7%, and 4.7% at incubation temperatures of 15°C, 20°C, and 25°C, respectively, in a 30-day period. Furthermore, extending the incubation time to 60 days increased percent germination to 99.3% at 15°C, 97.3% at 20°C, and 25.3% at 25°C. There were, however, differences in the optimal temperatures for germination because H. koreana and H. tubiflora seeds germinated in higher percentages at 30/20°C (25°C on average) under 12/12 h alternating light conditions. Inter-specific variations in stratification temperatures for dormancy- break and germination were reported in Lonicera fragrantissima and L. morrowii (Hidayati et al., 2000), Corylopsis coreana and C. sinensis (Roh et al., 2008), and in Muscari spp. (Doussi and Thanos, 2002).
Copete et al. (2011) classified the seeds of H. orientalis as having MD, due to the available information and germination data published by Takahashi (1984). However, in seeds of H. orientalis incubated at 20°C, there was a further increase (to 30%) in germination percentage between 30 and 60 days (Takahashi, 1984). According to Baskin and Baskin (1998), most seeds with MD germinate within 30 days without any dormancy-breaking pretreatment. Hidayati et al. (2000) reported that approximately 50% of Lonicera maackii and L. morrowii have MD and approximately 50% have MPD, due to studies in which fresh seeds of L. maackii and L. morrowii germinated to 48-52% in the light after 4 weeks at 15/6-25/15°C. Thus, they concluded that the seeds have non-deep simple MPD which has about 50% MD. Therefore, further studies should be conducted to determine whether Heloniopsis species have MD or MPD.
Light was a critical signal for seed germination in H. koreana and H. tubiflora seeds since no seeds germinated under 25/15°C in constant dark conditions. The light requirement for germination and seedling emergence in these two species indicates that the initial establishment of these species may favor open habitats. It has also been suggested that smaller seeded taxa might germinate only on the soil surface or at shallow depths (Koutsovoulou et al., 2014). Once small-sized, photoblastic seeds are buried, they are expected to be contained in soil seed banks (Milberg et al., 2000; Rodrigues and Silveira, 2013). In this study, we did not observe germination or seedling emergence for more than 5 months in seeds buried for a field test (data not shown). These characteristics suggest that the seeds of H. koreana and H. tubiflora have the potential to form a soil seed bank.
We conclude that the seeds of the two Heloniopsis species possess MD with some portion of MPD. Warm stratification and GA3 can effectively break seed dormancy of the two species. Light was essential for germination. Therefore, a practical production plan can be created for seed propagation and conservation strategy when this information is coupled with investigation dealing with the effects of storage method, cold stratification and growing media on viability, dormancy, germination, and seedling growth of the two Heloniopsis seeds.