Introduction
Materials and Methods
Plant Materials and Treatments
Plant Growth Parameters
Statistical Analysis
Results
The Effect of the Treatment with GA3 Alone was Insignificant
Growth Inhibition by DIN was Recoverable with GA3
Discussion
The Effect of the Treatment with GA3 Alone is Insignificant
Growth Inhibition by DIN is Recoverable with GA3
Conclusion
Introduction
Tomato (Solanum lycopersicum L.) is a widely used plant that produces important secondary metabolites, as well as lycopene, beta-carotene, flavonoids, and vitamin C (Gerszberg et al., 2015; Paduchuri et al., 2010). As the production and consumption of tomatoes increase, the value of the seedling production business for cultivating crops is also increasing (Oduol and Franzel, 2014). Uniform seedling emergence is essential for the efficient production of seedlings (Herrera et al., 2008). Plant growth regulators (PGRs) have not only been applied successfully to overcome potential production restriction factors but also to promote the quality of produce in many crops (Kaur et al., 2015). PGRs are naturally occurring hormones or synthetic analogs that significantly affect plants, even at low concentrations, and can regulate cell division, cell elongation, and gene expression (Basra, 2000; Nambara and Marion-Poll, 2005; Teale et al., 2006; Rostami et al., 2016). Various PGRs regulate the mechanism of action of phytohormones, such as auxin, abscisic acid (ABA), brassinosteroid, cytokinin, ethylene, gibberellin acid (GA), jasmonic acid, salicylic acid, and strigolactone (Rajala et al., 2002; Ault and Siqueira, 2008; Carey et al., 2008; Gopi et al., 2009; Anosheh et al., 2012; Choudhury et al., 2013; Banerjee and Roychoudhury, 2018; Rafeie et al., 2020; Raza et al., 2021).
Triazole compounds regulate plant growth by altering the balance of important phytohormones, including GA and ABA (Jaleel et al., 2007). Triazole-based PGRs inhibit catabolism by ABA-8'-hydroxylase, a cytochrome P450 monooxygenase in the ABA catabolism pathway (Mizutani and Todoroki, 2006). ABA accumulation by catabolism inhibition causes stomatal closure, which minimizes water loss through plant transpiration and increases drought stress tolerance (Leung and Giraudat, 1998; Zhang et al., 2006; Herrera-Medina et al., 2007; Veselov et al., 2016). GA inhibition by triazole-based PGR occurs by inhibiting the oxidation of ent-kaurene to ent-kaurenoic acid by inactivating cytochrome P450-dependent oxygenases along the GA biosynthesis pathway (Desta and Amare, 2021). A typical symptom of GA inhibition is a decrease in plant height (Fernandez et al., 2009; Tanimoto, 2012; Wang et al., 2017). Triazole compounds are generally known as plant multi-protective agrochemicals owing to their ability to induce tolerance to both intrinsic and extrinsic stresses in plants. Triazole-based PGRs have been used for various purposes, such as fungicides and PGRs, and for modulating seedling growth, environmental adaptability, and quality improvements in various horticultural plants (Fletcher et al., 2000; Gopi et al., 2007). Kim and Lee (1997) suggested that triazole-based PGRs show activity corresponding to that of auxin, such as increasing the rooting effect and inhibiting stem elongation owing to GA inhibition, also suggesting possible uses in the seedling industry. Triazole-based PGRs have been used to improve seedling quality by inhibiting the growth of tomato seedlings (Lee et al., 1997) and been used as well to prevent overgrowth due to transplanting density, temperature, humidity, and light conditions during the production of tomato seedlings (Zhang et al., 2003). However, the inhibitory effect of triazole-based PGRs on GA can reduce production because growth inhibition can act as a side effect depending on the concentration and frequency of the PGR treatment (Herrera-Medina et al., 2007). Seedling quality can vary depending on the species, treatment methods, and other factors during the seedling period (Malik et al., 2020; Qin and Leskovar, 2020). Therefore, plant and environmental characteristics should be scrutinized and optimized to minimize the damage caused by PGR (Venkatramesh and Croteau, 1989).
We hypothesized that an exogenous GA treatment after the application of a triazole-based PGR would resolve the plant growth inhibition issue. After applying triazole-based PGR, the optimum GA concentration and number of treatments were confirmed. The objective of this study was to investigate whether an exogenous GA treatment could mitigate the growth inhibition of tomato seedlings when used in conjunction with a diniconazole (DIN) treatment. The effects of the DIN treatment on the survival rate and growth of tomato seedlings were examined. After the DIN treatment, GA3 was sprayed onto the leaf surfaces to confirm the growth outcomes of the tomato seedlings.
Materials and Methods
Plant Materials and Treatments
A 50-cell plug tray (54 × 28 × 5 cm) with a cell capacity of 72 mL was filled with a 30:70 (v/v) pearlite-to-peat moss mix and five cherry and five ordinary tomatoes for greenhouses were sown (Table 1). Fifteen days after sowing, when three to four true leaves on the cherry and ordinary tomato plants had developed, Binnari (diniconazole, DIN; 50,000 mg·L-1, Dong-Bang Agro Corp., Korea) was diluted to 250 mg·L-1 and foliar-sprayed such that each plant received 0.3 mg. After 37 days of sowing, the plants were transplanted into rockwool (1000 × 150 × 75 mm, Grodan GT MASTER, Grodan, The Netherlands) and cultivated for 19 days in a Venlo-type greenhouse. The nutrient solution was used for tomato cultivation and was modified such that it improved the Dutch Greenhouse Crop Research Institute (PBG) nutrient solution. The composition of the modified nutrient solution was N 8.0 me·L-1, P 2.4 me·L-1, K 2.4 me·L-1, Ca 4.8 me·L-1, and Mg 1.6 me·L-1. A GA3 treatment was used to assess growth recovery after the DIN treatment. The plants were treated with Daeyugibberellin (GA3; 31,000 mg·L-1, Daeyu Co., Korea) at concentrations of 5 mg·L-1 and 10 mg·L-1 41 days after sowing (one-time treatment, OTT), and the same GA treatment was applied at concentrations of 5 mg·L-1 and 10 mg·L-1 44 days after sowing (two-time treatment, TTT). The diluted GA3 aqueous solution was applied to tomato leaves at 2.5 mL per plant. We compared the plant height, stem diameter, leaf length, fresh weight, and dry weight between the one-time treatment (OTT) and two-time treatment (TTT) groups. We also examined the effects of the GA3 treatment at concentrations of 5 mg·L-1 and 10 mg·L-1 on the plant growth outcomes.
Table 1.
Cherry and ordinary tomato cultivars used in the experiment
Plant Growth Parameters
The plant height, stem diameter, leaf length, fresh weight, and dry weight were measured 19 days after transplanting. The plant height was measured from the substrate surface to the apical meristem using tape. The stem diameter and leaf length were measured using a caliper (SD500-300PRO, Shin Con Co. Ltd., Korea) as a non-destructive method. The stem diameter of the main stem closest to the first inflorescence was measured. The leaf length was measured in the lower unfolded leaf of the first inflorescence. The fresh weight was measured using an electronic scale (MW-2N, CAS Co. Ltd., Korea). For the dry weight measurements, the samples were dried for seven days in a dryer (HB-501M, Hanbaek Scientific Technology Co. Ltd., Korea) at 80°C and then measured using the same electronic scale.
Statistical Analysis
The statistical analysis of the experimental data was conducted using SAS Statistical Software version 9.4 (SAS Institute Inc., Cary, NC, USA). The effects of the DIN treatment and GA3 treatments at different concentrations and frequency after the DIN treatment on the survival rate of cherry and ordinary tomato seedlings were evaluated using a three-repetition randomized block design. Duncan’s multiple range test (p < 0.05) was used to confirm statistically significant differences among all treatments.
Results
The Effect of the Treatment with GA3 Alone was Insignificant
There were no significant differences in the plant height, leaf length, stem diameter, fresh weight, or dry weight between the GA3 treatment (5 mg·L-1, 10 mg·L-1) and the control (GA3-untreated) in the cherry (TH, TD, CA) and ordinary (DO, DA, PI) tomatoes without a DIN treatment (Figs. 1 and 2).
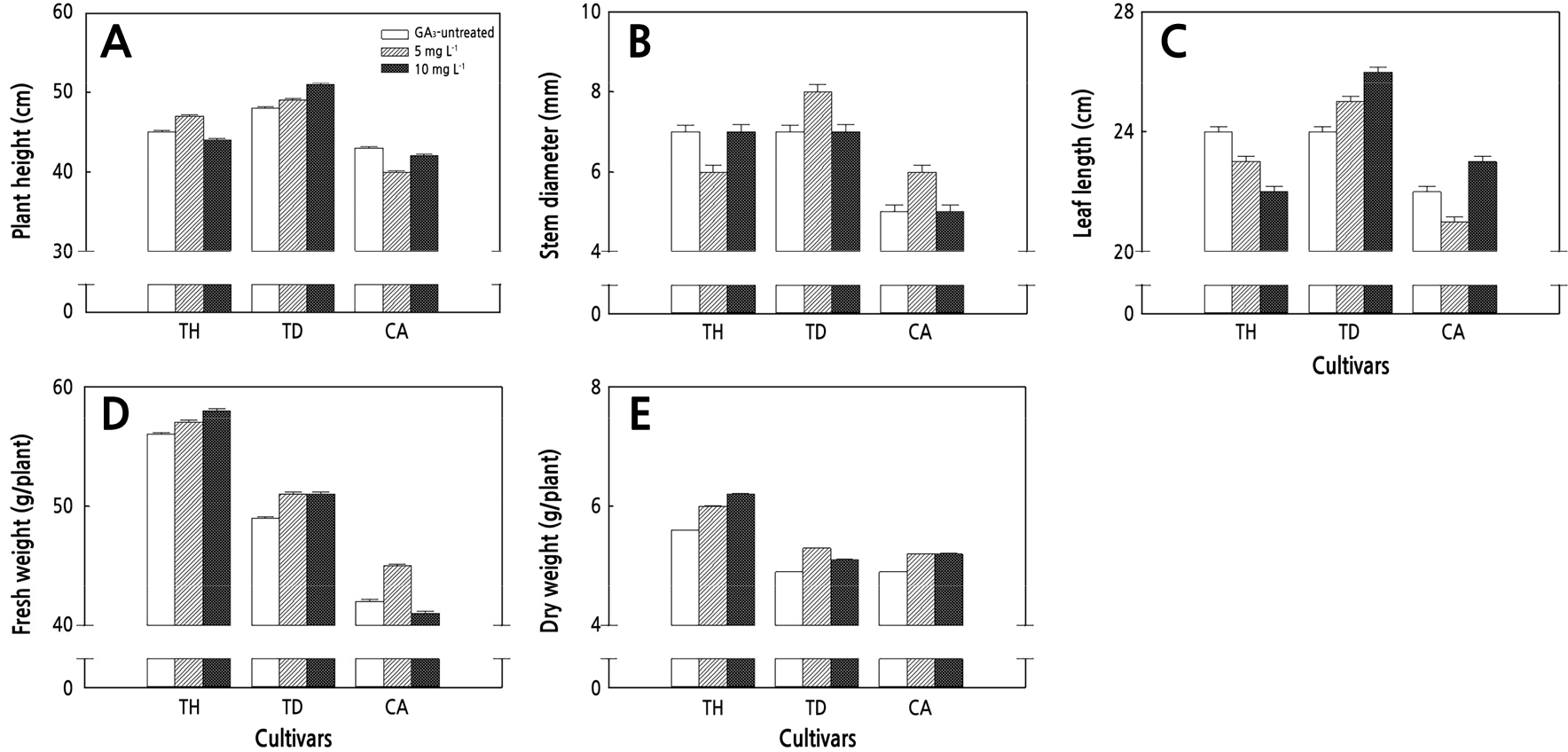
Fig. 1.
Negligible plant heights (A), stem diameters (B), leaf lengths (C), fresh weights (D), and dry weights (E) relative to different gibberellin acid (GA) treatment concentrations in diniconazole (DIN) non-treatment cherry tomato seedlings. The data show the means and the vertical bars show the stand errors (n = 20). Different letters show significant differences among the GA treatments (Duncan’s multiple range test, p < 0.05, n = 20). TH, TYholyday; TD, Tydaejanggeum; CA, Candyplus.
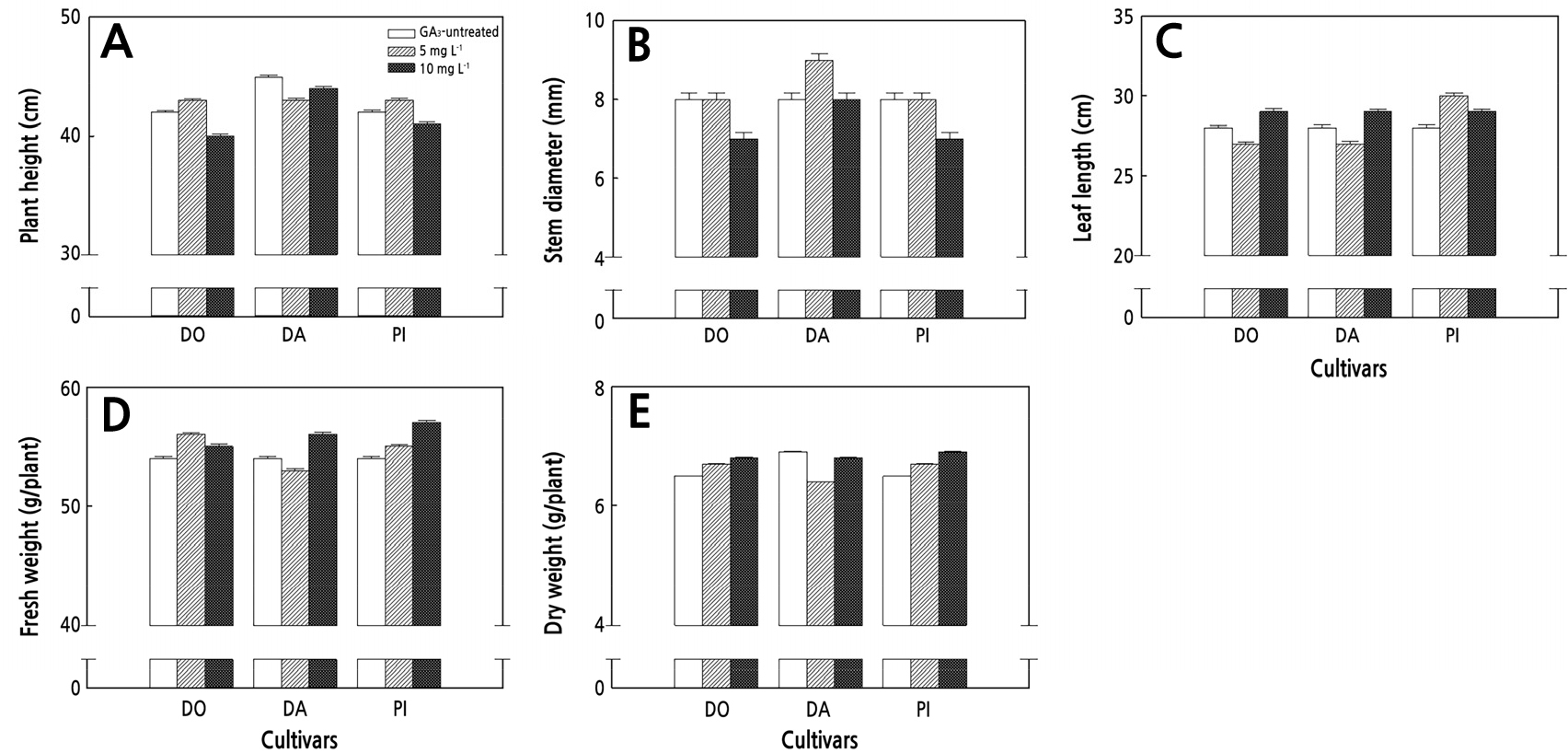
Fig. 2.
Negligible plant heights (A), stem diameters (B), leaf lengths (C), fresh weights (D), and dry weights (E) relative to different gibberellin acid (GA) treatment concentrations in diniconazole (DIN) non-treatment ordinary tomato seedlings. The data show the means and the vertical bars show the stand errors (n = 20). Different letters show significant differences among the GA treatments (Duncan’s multiple range test, p < 0.05, n = 20). DO, Dokia; DA, Daefnis; PI, Pillabi.
Growth Inhibition by DIN was Recoverable with GA3
After the DIN treatment, the leaves of GA3-treated tomato seedlings were wide and light green compared to the control (GA3-untreated) tomato seedlings regardless of the concentration (5 mg·L-1, 10 mg·L-1) and/or frequency of the treatments (Fig. 3). The plant heights of the cherry tomato cultivars (SW, OL, CA, TD, and TH) did not increase significantly with the GA3 treatment concentration but tended to increase with the number of treatments (Fig. 4A). Among these, the one-time treatment (OTT) of GA3 at 5 mg·L-1 and 10 mg·L-1 did not significantly increase the plant height in four of the ordinary tomato cultivars (PI, TO, DA, and HO), with DO being the exception, compared to the control. The two-time treatment (TTT) of GA3 at 5 mg·L-1 and 10 mg·L-1 tended to increase stem elongation in ordinary tomatoes (Fig. 5A). There were no significant differences in the stem diameters of the cherry and ordinary tomatoes between the control and treatment groups (Figs. 4B and 5B). The leaf length increased in the treatment group compared to the control in both cherry and ordinary tomatoes, but there were no significant differences according to the treatment concentration and/or number of treatments (Figs. 4C and 5C). In the GA3 5 mg·L-1 treatment, there was a significant difference in the fresh weight between the OTT and TTT cases in SW, CA, TD, and TH. In all cherry tomato cultivars, a TTT of 10 mg·L-1 tended to increase the fresh weight. There was no significant difference in the fresh weight between the 5 mg·L-1 and 10 mg·L-1 TTT cases in four cultivars (SW, CA, TD, and TH), but it increased at 10 mg·L-1 in the OL cultivar (Fig. 4D). The TTT of GA3 at 5 mg·L-1 increased the fresh weights of DO, PI, TO, and DA among the five ordinary tomato cultivars. The fresh weights of PI, HO, and DA increased after the GA3 10 mg·L-1 TTT but not after the OTT. There were no differences in the fresh weights of PI, HO, and DA between the TTT of GA 5 mg·L-1 and 10 mg·L-1, but for DO, the fresh weight increased only with the TTT of 10 mg·L-1 (Fig. 5D).
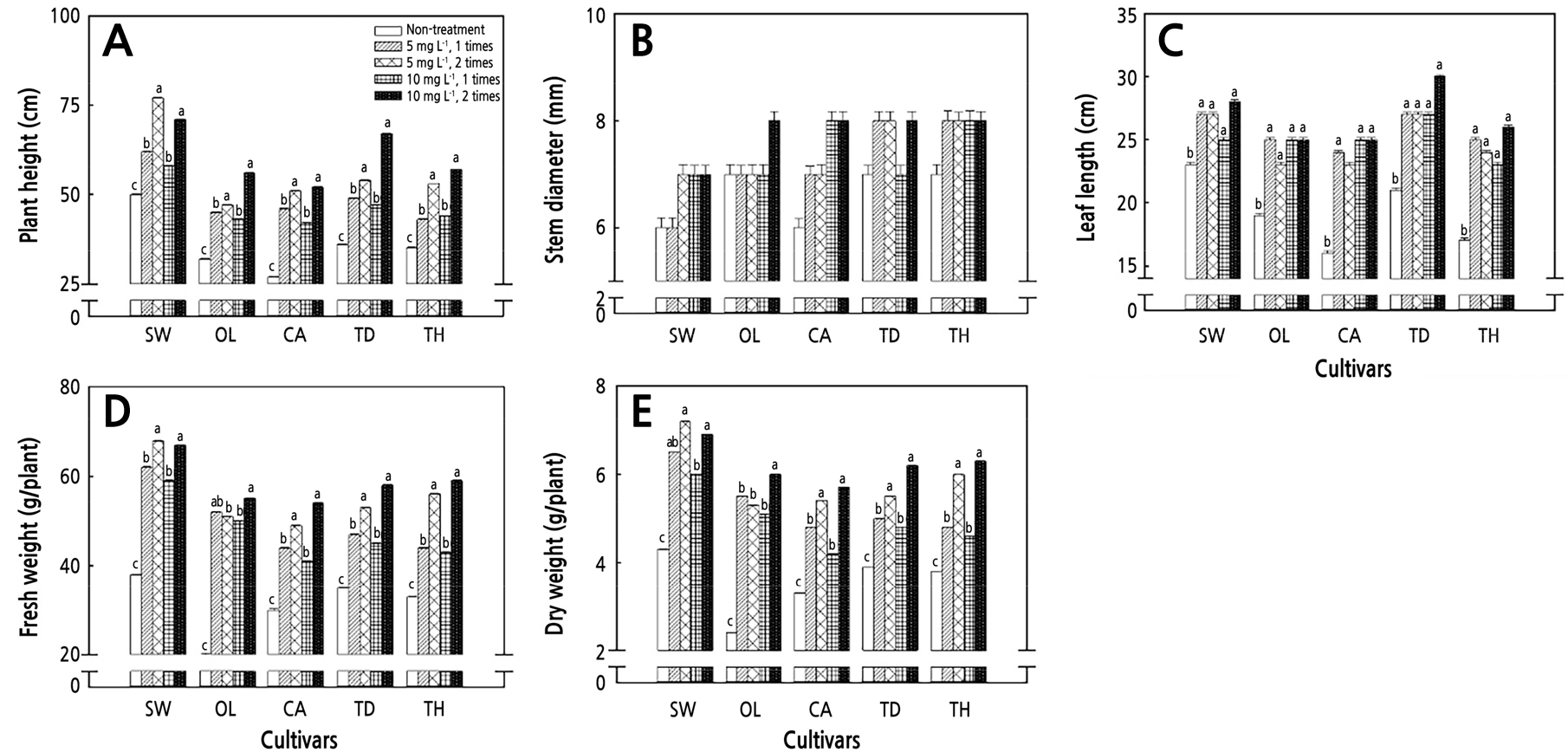
Fig. 4.
Alterations of the plant height (A), stem diameter (B), leaf length (C), fresh weight (D), and dry weight (E) relative to different gibberellin acid (GA) concentrations after a diniconazole (DIN) treatment of cherry tomato seedlings. Plant growth measurements were taken 19 days after transplantation. The data show the means and the vertical bars show the stand errors (n = 20). Different letters show significant differences among the GA treatments (Duncan’s multiple range test, p < 0.05, n = 20). SW, Sweetelle; OL, OlleTY; CA, Candyplus; TD, Tydaejanggeum; TH, TYholyday.
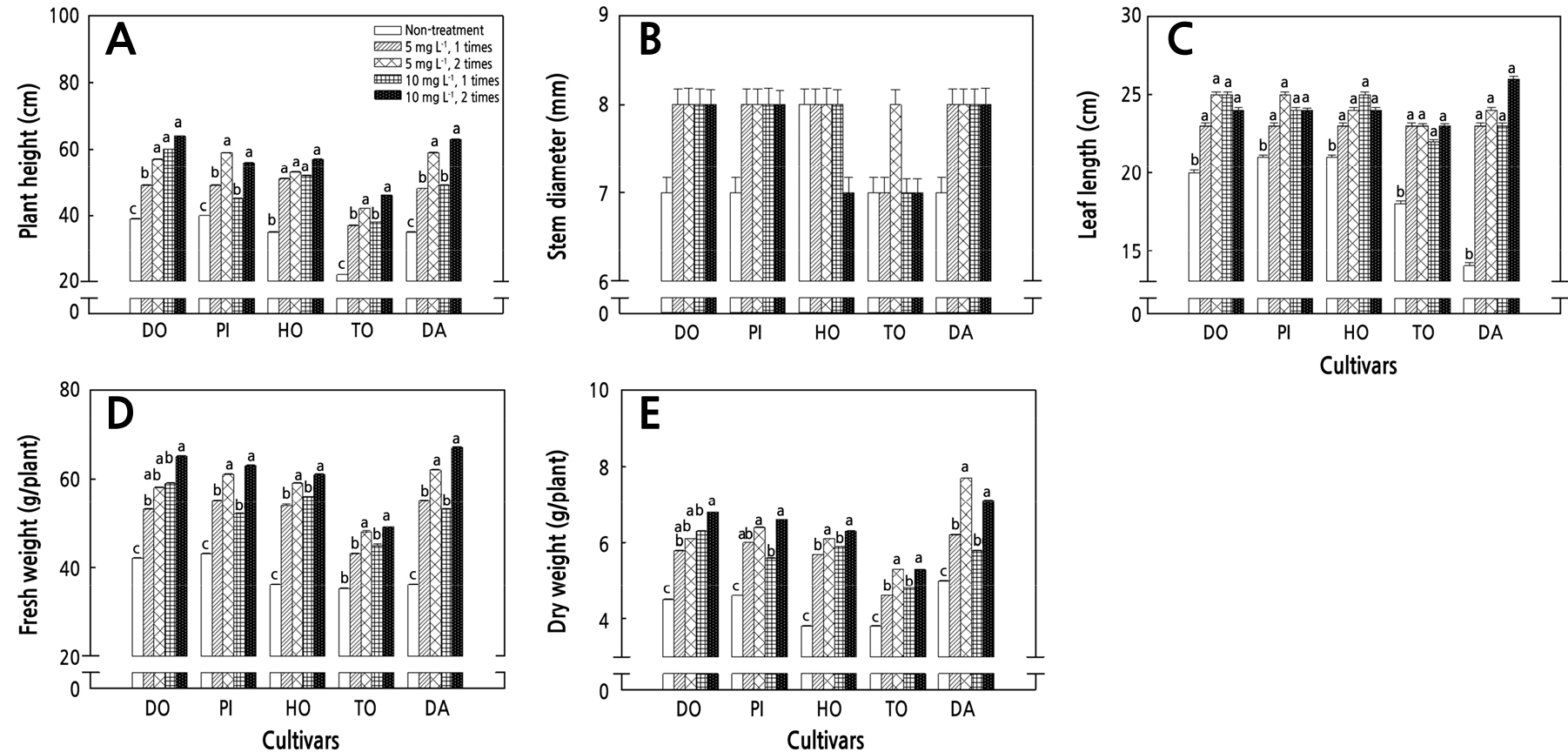
Fig. 5.
Alterations of the plant height (A), stem diameter (B), leaf length (C), fresh weight (D), and dry weight (E) relative to different gibberellin acid (GA) concentrations after a diniconazole (DIN) treatment of ordinary tomato seedlings. Plant growth measurements were taken 19 days after transplantation. The data show the means and the vertical bars show the stand errors (n = 20). Different letters show significant differences among the GA treatments (Duncan’s multiple range test, p < 0.05, n = 20). DO, Dokia; PI, Pillabi; HO, Hoyong; TO, TYonetop; DA, Daefnis.
Discussion
The Effect of the Treatment with GA3 Alone is Insignificant
Duckweed (Lemna gibba G3) treated with 0.1–100 mg·L-1 GA3 does not show an increase in the fresh weight (Cleland and Briggs, 1969). When common bean (Phaseolus vulgaris L.) was irrigated with a Hoagland solution having a GA3 concentration of 0.05 mM, there were no significant differences in the fresh and dry weights of the shoots (Saeidi-Sar et al., 2013). Contrary to earlier results, Qiu et al. (2019) reported that a GA3 (200 mg·L-1) treatment significantly increased the node length compared with control plants in sugarcane (Saccharum officinarum). In addition, a GA3 treatment at 50 mg·L-1 increased the plant height and decreased the stem diameter compared to control plants in wheat (Triticum aestivum L.) (Peng et al., 2014). It appears that the growth of each plant is the result of genetically different levels of sensitivity to phytohormones. In particular, GA sensitivity leads to a different response depending on the REPRESSOR OF GA (RGA)/GA-INSENSITIVE (GAI) gene among the GRAS family DELLA subsets (Pysh et al., 1999; Olszewski et al., 2002). RGA/GAI is a negative regulator of the GA response that regulates the flowering time, stem elongation, and root elongation (Dill and Sun, 2001; Fu and Harberd, 2003). It is likely necessary to confirm RGA/GAI gene expression outcomes in cherry and ordinary tomato plants upon a treatment with GA3. It is possible that the concentration of GA3 used in our experiment was insufficient to affect the growth outcomes.
Growth Inhibition by DIN is Recoverable with GA3
Davière and Achard (2013) reported that GA-deficient mutants exhibit dwarfism and delayed flowering and that an exogenous GA treatment of these plants can recover normal growth. These findings support the results of the present study. GA inhibits stem thickness growth and promotes stem elongation (Eriksson et al., 2000). When Hibiscus cannabinus was exogenously treated with GA3, the plant height increased compared to the control, but the stem diameter decreased (Muniandi et al., 2018). Wang et al. (2021) showed that an exogenous treatment with GA3 increased the height of Oryza sativa by 52%. These results are similar to those of our study with regard to plant height but not stem diameter (Figs. 4A, 4B, 5A, and 5B). Stem thickness growth in plants is associated with lignin accumulation, a major structure of the secondary cell wall (Ma, 2009). Peng et al. (2014) reported that lignin accumulation gradually increases after node formation, whereas a GA3 application decreases lignin accumulation. Because the tomato seedlings used in our experiment were young plants with fewer nodes, the absolute amount of lignin was also lower. Accordingly, it is presumed that there was no difference between the control and treatment groups. The leaf lengths of A. tubergenii ‘Zwanenburg’ increased when an increased concentration was used in the exogenous GA3 treatment compared to the control (Salachna et al., 2020). An exogenous application of GA3 stimulates mitosis in plants (Sauter and Kende, 1992). Therefore, an increase in the number of plant cells through mitosis may lead to the expansion of the leaves. Inoculation with the Pseudomonas putida H-2-3 strain, which increases GA4, significantly increased the shoot fresh and dry weight of soybean shoots (Glycine max. L. ‘Taekwang’) (Kang et al., 2014). The results shown in Figs. 4 and 5 can be explained by existing studies of the effects of exogenous active GA treatments on plant growth. Consequently, the inhibition of plant growth by DIN was reversible through an exogenous GA treatment.
Conclusion
The results of this study provide evidence that an exogenous GA treatment mitigates the negative effects of diniconazole (DIN). Our findings provide directions for further research to examine the concentration of an exogenous GA treatment required after a DIN treatment. However, little is known about recovery methods after accidents caused by a DIN treatment. We evaluated plant growth parameters related to the effects of an exogenous GA treatment after a DIN treatment at the tomato seedling stage, but we do not know if the trends we observed hold for other plant species. Further studies are needed to examine the long-term effects of a GA treatment after a DIN treatment at later stages of plant growth. These questions need to be addressed to provide the necessary guidance to growers engaged in seedling production.



