Introduction
Materials and Methods
Research Materials and Storage Conditions
Fresh Weight Loss Rate and Gas Concentration Inside the Film Package
Chromaticity Change and Chlorophyll Content
Quality Change
Statistical Analysis
Results and Discussion
Fresh Weight Loss Rate and Changes in Gas Concentration in Film Package
Changes in Chromaticity and Chlorophyll Content
Quality Change
Conclusion
Introduction
Increasing interest in health foods has led to increased consumption of asparagus spears (Asparagus officinalis L.) (Seong et al., 2012). Accordingly, the domestic cultivation area of asparagus spears has expanded to an estimated 55.4 ha in Korea as of 2016 (Rural Development Administration, 2016). Currently, asparagus spears are packed using perforated film for distribution and sale in Korea. The high respiration and transpiration rate of asparagus spears means that this method of packing results in large fresh weight losses during storage and distribution (Yoon et al., 2016).
Although they are a non-climacteric crop (Zhang et al., 2012), as a young, vigorously growing organism (Ryall and Lipton, 1972), asparagus spears are notoriously difficult to store for long periods because of their very high respiration and decomposition rates (Kader, 2002). Therefore, many studies have sought to identify more effective methods of asparagus storage, including controlled atmosphere (CA) (Lee, 1981), modified atmosphere (MA) (Baxter and Waters, 1991; Gariepy et al., 1991), and high hydrostatic pressure storage (Yi et al., 2016). Of these, MA storage has a low processing cost and is easy to apply to distribution sites, thus is considered the most effective storage method for maintaining good asparagus spear quality. However, the high respiration rate of asparagus potentially leads to an off-odor caused by low oxygen content inside the packaging (Gariepy et al., 1991; Siomos et al., 2000). Baxter and Waters (1991) also found that off-odor and asparagus spear yellowing occurred at low oxygen levels (less than 2%) during MA storage, leading to a shorter shelf life compared to the conventional perforated packaging.
Some researchers have developed technologies to enhance the oxygen transmission rate (OTR) of film treatments (Blakistone, 1998; Zeng et al., 2002), with a view to improving the MA storage of crops with high respiration levels. A technique reported by Sohn et al. (2008) adjusts the OTR of a non-perforated film from 5,000 to up to 50,000 cc/m2·day·atm by irradiating with a 785 nm laser at 10-15 s intervals. Use of OTR film treatments created by this technology has also been used to enhance the quality of crops including apples (Choi et al., 2014), vegetable sprouts (Choi et al., 2013), and glassworts (Baek et al. 2013) under MA storage.
This study was conducted to assess the quality of asparagus spears under different OTR film treatments, and thus determine which OTR films are most suitable for the MA storage of asparagus spears.
Materials and Methods
Research Materials and Storage Conditions
Asparagus spears (Asparagus officinalis L., variety “Wellcome”) were cultivated in Yanggu-gun, Gangwon-do, Korea. The asparagus spears selected were about 1.4 ± 0.1 cm in diameter. OTR films (Dae Ryung Precision Packaging Industry Co., Ltd., Korea) with oxygen transmission rates of 10,000, 20,000, 40,000, 70,000, and 100,000 cc/m2·day·atm were used for MA storage, and conventional polypropylene (PP) film, perforated by drilling with 0.5 cm diameter holes every 25 cm, was used as the control. All experimental groups were stored at 4°C with a relative humidity of 85%.
Fresh Weight Loss Rate and Gas Concentration Inside the Film Package
Weight reduction was recorded as a percentage of weight lost. Carbon dioxide and oxygen concentrations within packages were measured using an infrared sensor (Checkmate, PBI, Denmark), and gas chromatography (GC-2010, Shimadzu, Japan) was used to measure ethylene concentration (Park et al., 2000).
Chromaticity Change and Chlorophyll Content
To determine the chromaticity, the hue angle value of asparagus tips and stems were measured with a color-difference meter (CR-400, Minolta, Japan) to indicate the degree of yellowing (Chang and Kim. 2015). To calculate total chlorophyll content, 1.0 g of tips or stems were dissolved in 10 mL methanol, before leaving at 4°C for 48 h to extract chlorophyll. A UV-VIS spectrophotometer (UV mini model 1240, Shimadzu, Japan) was then used to measure the absorbance at 642.5 and 660 nm. The following formula was applied to determine total chlorophyll (AOAC, 1997; Baek et al., 2014):
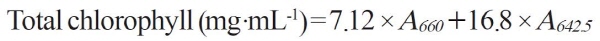
Quality Change
Five skilled panelists evaluated visual quality and off-odor using the method of Choi et al. (2015), Visual quality was assessed on a scale of 1 - 5, where 5 represented the highest visual quality, and 1 represented complete disposal. A grade of 3 or more was considered saleable. Off- odor was assessed on a scale of 0 - 5, where 0 represented very little off- odor, and 5 represented a strong off-odor.
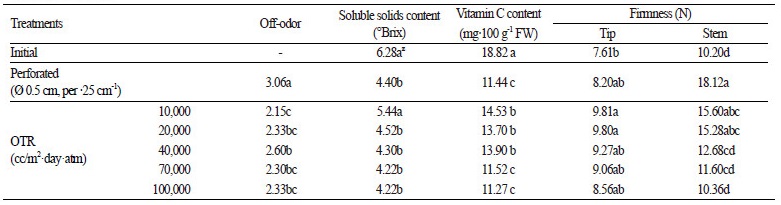
The soluble solids content of each sample was measured with a refractometer (PAL-1, Atago, Japan) and indicated as ºBrix. Vitamin C content was measured using a ZORBAX Eclipse XDB- C18 analytical column (4.6 × 2.5 cm, 5 μm, Agilent, USA) and a tunable absorbance detector (Waters 486, Waters, USA) using high performance liquid chromatography (Waters, USA) (Kim et al., 2011). A rheometer (Compac-100, Japan) using a No. 5 (Ø 3.0 mm) probe at a speed of 1.0 mm·s-1 was used to measure the firmness of tips (the topmost 5 cm of asparagus spears) and stems (the lowermost 8 cm).
Statistical Analysis
Graphs of statistical analysis were created using Microsoft Excel 2013. SPSS Statistics 21.0 software was used to perform Duncan’s multiple range tests.
Results and Discussion
Fresh Weight Loss Rate and Changes in Gas Concentration in Film Package
In the perforated film treatment, the fresh weight loss of asparagus spears was about 7.5%, and less than 1% in all MA storage treatments. However, there was no difference in the OTR of film treatments between MA storage treatments (Fig. 1A). Since the maximum allowable fresh weight loss of asparagus spears during storage is 8% (Kays and Paull, 2004), no deterioration of quality resulting from Fresh weight loss was observed in any storage treatments in this experiment. The rate of fresh weight loss was much lower in the MA storage treatments than in the perforated film treatment; this was caused by relatively high humidity in the MA packaging that inhibited transpiration (Lee, 1996).
Gas concentration was only measured in the MA storage treatments. The ethylene concentration increased rapidly and proportionately to the film transmission rate on the first day of storage. This was presumably caused by endogenous ethylene synthesis stimulated by the physical stress applied to the delicately structured asparagus spears during early storage processes such as packaging (Kato et al., 2000). Thereafter, ethylene concentration gradually decreased with increasing storage time. This may have been caused by the decreased production of ethylene in response to increased carbon dioxide (2 - 8%) and reduced oxygen (12 - 20%) in the packaging (Figs. 1C and 1D). After 20 days of storage, the ethylene concentration in all treatments was about 2.0 μL∙L-1 (Fig. 1B).
Lee et al. (1995) suggested that ethylene biosynthesis is inhibited under CA and MA storage conditions, and that carbon dioxide is a competitive inhibitor of ethylene. During storage, the carbon dioxide concentration in the packaging was also proportionate to the film permeability level: the highest concentration of carbon dioxide was 7 - 9% in the 10,000 cc OTR film treatment, and 5 - 7% in the 20,000 cc OTR film treatment (Fig. 1C). The oxygen concentration in the packaging decreased on the first day of storage. However, even in the 10,000 cc OTR film treatment, which had the lowest oxygen concentration, was maintained above 12% (Fig. 1D).
A carbon dioxide concentration of 5 - 12%, and an oxygen concentration of 5% or above is suitable for both CA and MA storage of asparagus in most cases (Kader, 2002). Conventional experiments on the MA storage of asparagus spears have reported a deterioration in quality at oxygen concentrations less than 3% because of anaerobic respiration (Baxter and Waters, 1991; Gariepy et al., 1991). In this experiment, laser-controlled oxygen permeability enabled us to maintain the oxygen concentration at 12% or more during storage. In addition, the carbon dioxide and oxygen levels observed in the 10,000 and 20,000 cc OTR film treatments satisfied the proper MA composition ranges of asparagus spears. When asparagus spears are stored for a long time, the atmospheric composition of the packages has a positive effect in terms of inhibiting yellowing and off-odors, and in maintaining visual quality (Fig. 2, 3, Table 1).
Fig. 1.
Changes in fresh weight loss rate (A), ethylene (B), carbon dioxide (C), and oxygen concentrations (D) of asparagus spears packaged with different oxygen transmission rate (OTR) film treatments (from 10,000 to 100,000 cc/m2·day·atm) for MA storage for 25 days, and the perforated film treatment (Ø 0.5 cm, per 25 cm-1 ) for 20 days at 4°C. Vertical bars represent ± SE (n = 5).
Changes in Chromaticity and Chlorophyll Content
US quality standards place importance on the color change of green asparagus spears (Kader, 2002), and stem yellowing during long-term storage is a major factor in visual quality deterioration (Yoon et al., 2016). Therefore, in this experiment, hue angle values of asparagus spear tips and stems were obtained to determine the degree of yellowing during storage. On the last day of storage, the highest hue angle value of the tips was obtained in the 10,000 cc OTR film treatments. There was no statistically significant difference between the values obtained for the 40,000, 70,000, or 100,000 cc OTR or perforated film treatments. In stems, the highest hue angle value was recorded in the 10,000 cc OTR film treatment, and the lowest value in the perforated film treatment (Fig. 2A). Furthermore, the total chlorophyll content was similar to that of the hue angle value: total chlorophyll was highest in tips in the 10,000 and 20,000 cc OTR film treatments, and in stems, it was highest in the 10,000 cc OTR film treatment, and lowest in the perforated film treatment (Fig. 2B). Consistent with conventional reports, the total chlorophyll content of asparagus spear tips was higher than that of the stems (Baek et al., 2014), many horticultural products are subject to color change and yellowing by aging and ethylene (Kader, 2002). In the case of asparagus (Wang et al., 1971) and broccoli (Cho et al., 2009), decreased chlorophyll is associated with aging and ethylene activity. A study by Lee (2015) found that chlorophyll reduction was more progressive in asparagus spears processed with 10 μL∙L-1 ethylene. However, in the present experiment, the highest hue angle and total chlorophyll content values were in the asparagus spears packed with 10,000 cc OTR film, which had the highest ethylene concentration in early storage. This was most likely because treatment with this film satisfied the conditions for MA (i.e., a carbon dioxide concentration of 5 - 12% and an oxygen concentration of 5% or more). Additionally, the concentration of ethylene, which is the cause of yellowing, was 2.0 μL∙L-1 after 20 days of storage, similar to other treatments.
Fig. 2.
Changes in hue angle (A) and total chlorophyll (B) of asparagus spears packaged with different oxygen transmission rate (OTR) film treatments (from 10,000 to 100,000 cc/m2·day·atm) for MA storage for 25 days, and the perforated film treatment (Ø 0.5 cm, per 25 cm-1 ) for 20 days at 4°C. Vertical bars represent ± SE (n = 5).
In general, horticultural products inhibit the biochemical activity of 1.0 μL∙L-1 ethylene at highly concentrated carbon dioxide levels of 10%; this implies that carbon dioxide can accumulate in the intercellular space and inhibits ethylene creation (Yang, 1985). Kader (2002) also reported that ethylene production was inhibited under appropriate CA conditions, and the chloroplast enzyme activity of broccoli was inhibited under high carbon dioxide conditions, thereby delaying yellowing (Cho et al., 2009).
Quality Change
Visual quality of asparagus spears examined during the storage period (25 days) were maintained at a saleable level of 3 points or above only in the 10,000 cc OTR film treatment, and lost commercial value after 16 days in the perforated film treatment (Fig. 3). In the perforated film treatment, a high fresh weight loss rate (6%) and degree of yellowing were identified as causes of the deterioration in quality. On the other hand, asparagus packaged in 10,000 cc OTR film with appropriate CA and MA conditions displayed reduced yellowing and a shelf-life extension of 10 days.
The off-odor was assessed on the final day of storage. An off-odor caused by soft rot (data not shown) in the tips was most apparent in the perforated film treatment after 15 days of storage. According to Cho et al. (2009), soft rot and decomposition was also the cause of an off-odor in broccoli. Storage under MA conditions often suffers from increased generation of off-odors in the later stages of storage (Kader, 2002). In this experiment, the least off-odor was generated in the 10,000 cc OTR film treatment, which had the lowest oxygen permeability. Generally, acetaldehyde and ethanol-the chemicals responsible for off-odor-are produced by anaerobic respiration when oxygen concentrations are less than 2.5% (Park et al., 2015). However, in this experiment, because the oxygen concentration in the 10,000 cc OTR film treatment was maintained at 12% or higher, there was no anaerobic respiration (Kays and Paull, 2004), and textural quality was unaffected.
Fig. 3.
Change in visual quality of asparagus spears packaged with different oxygen transmission rate (OTR) film treatments (from 10,000 to 100,000 cc/m2·day·atm) for MA storage for 25 days, and the perforated film treatment (Ø 0.5 cm, per 25 cm-1 ) for 20 days at 4°C. Vertical bars represent ± SE (n = 5).
In all treatments, the soluble solids content was lower than the initial recorded value. The highest soluble solids content was observed in the 10,000 cc OTR film treatment-we assume this was caused by reduced respiration under proper CA and MA conditions. There were no significant differences between other film treatments.
The Vitamin C contents of asparagus spears under all treatments were reduced after storage compared to the initial recorded value. The highest level of Vitamin C was found in the 10,000 cc OTR film treatment, followed by the 40,000 and 20,000 cc OTR film treatments. In a study by Soliva-Fortuny et al. (2002), Vitamin C degradation was higher when highly oxygenpermeable packing materials were used during the MA storage of kohlrabi (Park et al., 2014). Similarly, in this experiment with asparagus spears, the highly oxygen-permeable 70,000 and 100,000 cc OTR film treatments, and the treatment with perforated film, demonstrated the greatest decrease in Vitamin C. The highest content of Vitamin C was found in the 10,000 cc OTR film treatment, which had the lowest oxygen concentration.
In all treatments, the firmness of both asparagus tips and stems increased after storage compared to pre-storage, but in tips, these results were not statistically significant. Stem firmness was highest in the perforated film treatment, increasing by 77% compared to pre-storage, followed by the 10,000 and 20,000 cc OTR film treatments. These results were similar to the prestorage firmness of asparagus stored with 70,000 and 100,000 cc OTR film treatments (Table 1). Studies of other horticultural products showed that firmness also decreased because of increased polygalacturonase (PG) activity caused by ethylene gas (Kays and Paull, 2004). In cherimoya, high carbon dioxide conditions inhibited PG protein synthesis and delayed softening (Del Cura et al., 1996).
Asparagus spear firmness is also increased by tissue lignification during storage. Pretreatment with ethylene at 1.0 μL∙L-1 for 24 h before storage enhanced the lignification of tissues by activating phenylalanine ammonia-lyase and peroxidase (Liu and Jiang 2006). In this experiment, packages with higher concentrations of ethylene had an increased tendency to firmness (Table 1). However, the firmest asparagus stems were found in the perforated film treatment, which had no exposure to ethylene because of the fresh weight reduction (7.5%). Firmness may be increased by fibrosis caused by moisture loss, even in mung bean sprouts (Cho et al., 2005).
Conclusion
In asparagus spears, fresh weight loss, yellowing, soluble solids content, Vitamin C content, and off-odor generation are suppressed by storage at a low temperature of 4°C, when packaged with 10,000 cc OTR film, and when proper CA or MA conditions are maintained. As a result, the storage life can be extended by about 10 days compared to the conventional perforated film treatment.