Introduction
Materials and Methods
Plants, Botrytis cinerea, and Preparation of the Slightly Acidic Electrolyzed Water
Botrytis cinerea Inoculation and Pulsing Treatment of Petals or Flowers
Evaluation of the Cut Flower Quality
Experimental Design and Data Analysis
Results and Discussion
Introduction
Cut rose flowers are among the world's top three flower crops and are in demand year-round regardless of the season (Kim et al., 2022; Park and Kim, 2022). The cultivation area of cut roses in Korea amounts to 225 ha, accounting for 19% of the cultivation area of all cut flowers in Korea, and cut rose sales were the highest among all cut flowers in 2021 (MAFRA, 2022). Botrytis cinerea is a significant determinant of the cut rose flower quality after harvest among several factors, including the harvest time, temperature, relative humidity (RH), ethylene level, postharvest dry weight, bacteria characteristics, and the B. cinerea factor (de Stigter and Broekhuysen, 1989; Ha et al., 2020a). B. cinerea ranks second, after Magnaporthe oryzae, among the top ten fungal pathogens affecting plants (Dean et al., 2012). The market shares of roses, gerbera, and lisianthus with gray mold in Dutch flower auctions account for 8–10%, 22–33%, and 50%, respectively, during the distribution of cut flowers (Vrind, 2005). Economic losses associated with export-quality roses due to gray mold disease in Colombia and Kenya amount to 1.6% and 10–20%, respectively (Salgado-Salazar et al., 2018). Gray mold disease is frequently caused by high relative humidity levels exceeding 93% coupled with temperatures of 15–20°C during postharvest storage and transportation (Williamson et al., 1995; Fanourakis et al., 2013). Chemical control using fungicides is relatively inexpensive, can be easily applied on site, and both prevents and controls mole infections (Usall et al., 2016). Postharvest treatments by spraying cut rose flowers with fungicides such as iprodione (C13H13Cl2N3O3) plus thiram (C6H12N2S4), tebuconazole (C16H22ClN3O) plus dichlofluanid (C9H11Cl2FN2O2S2) and polyoxin D (C17H23N5O14) reduced disease incidence significantly by 50–58% (Elad et al., 1993). A pulsing treatment with methyl jasmonate (C13H20O3) 400 µM provides protection against Botrytis rot by activating resistance mechanisms in cut roses without decreasing the flower quality (Meir et al., 1998). A combination of specific botryticide (Roval 250SC, Scala 400SC, Teldor 500SC, and V90) sprays and fan drying reduced Botrytis infections and causes. The treatments had no adverse effects on the quality of cut roses (Goss and Mazarura, 2013). However, research on fungicide resistance is motivated currently because the global trend appears to be shifting toward the reduced use of fungicides on cut flowers (Mari et al., 2014). The development of new practices has reduced the negative impacts of pesticides on the environment and on consumer health (Daugaard, 1999; Wang et al., 2019). Therefore, researchers have sought to explore eco-friendly fungicides with antimicrobial effects against gray mold disease in cut flowers that are not resistant to fungicides. Coating mixture treatments of chitosan (0.1%) with Aloe vera pulp (25%), oregano essential oil (1%), and thyme essential oil (0.1%) do not induce damage in roses; however, the tested combinations did not reduce gray mold during postharvest treatments either (Herrera-Romero et al., 2017). Nano silver, sodium hypochlorite (NaOCl), and salicylic acid (C7H6O3), which are relatively safe compared to fungicides, are also used, and a petal spray treatment with 20 mg·L-1 of nano silver was effective in inhibiting B. cinerea and improving the quality of cut rose flowers (Ha et al., 2020b). Bergmann and Dole (2018) suggest an eco-friendly alternative to fungicides; essential oils such as the leaves of Cinnamomum zeylanicum Blume and flower buds of Eugenia caryophyllata Thunb were sprayed on the petals to suppress gray mold. However, these applications did not consistently control B. cinerea in rose flowers (Ha et al., 2021).
Slightly acidic electrolyzed water is environmentally safe and has been recognized for its stability and high sterilization effects in various studies, including those by the U.S. Food and Drug Administration and Japan's Ministry of Health, Labor and Welfare (Goo and Koo, 2020). It has an effective chlorine concentration of 20–30 µL·L-1 and a pH in the range of 5.0–6.5. It exhibits a 100% sterilization effect on certain spores, including B. cinerea, Phytophthora capsici, and Colletotrichum acutatum (Yoon, 2006; Song et al., 2013). The vase life of the cut rose ‘Bubble Gum’ was effectively extended by a 5 min stem pulsing treatment with slightly acidic hypochlorous water (30 µL·L-1), which reduced bacteria in the stems (Kim et al., 2022). However, whether slightly acidic electrolyzed water as a petal pulsing solution on cut rose flowers inhibits gray mold has not been assessed. This study investigates the effects of a postharvest petal pulsing treatment with slightly acidic electrolyzed water on the vase life and quality of cut rose flowers through a reduction in B. cinerea levels.
Materials and Methods
Plants, Botrytis cinerea, and Preparation of the Slightly Acidic Electrolyzed Water
The cut rose flowers ‘Dominica’ and ‘Bubble Gum’ were cultivated via a hydroponics system and harvested at the commercial harvest phase (Kumar et al., 2008) in October and December of 2022, respectively, from a greenhouse in Gwangju, Korea. In each case, transportation took place within 30 minutes to a laboratory at Chonnam National University. In the laboratory, stems were recut to a shoot length of 40 cm, and all but three leaves were removed (Lee and Kim, 2020). Naturally occurring B. cinerea was used in this study, and it was isolated in a rose production greenhouse by the Division of Horticulture Research at Gyeonggi-do Agricultural Research & Extension Services. Its pathogenicity was confirmed by re-inoculation on rose petals followed by incubation on V8 (Campbell Soup Company, USA) agar (LPS solution, Korea) at 24°C under a blue light intensity level of 6.6 µmol·m-2·s -1 PAR and with a 14 h photoperiod for 14 days (Hwang et al., 2019). The spores were scratched using sterile water containing 0.1% Tween 20% (Samchun, Korea) and to create a 2 × 107 spores·mL-1 spore suspension. The B. cinerea conidia stock was prepared by adding 40% glycerol in an amount identical to that of the spore suspension to obtain a concentration of 1 × 107 spores·mL-1. Subsequently, 1mL of the B. cinerea conidia stock was mixed with 9 mL of PDB (potato dextrose broth, Becton, France) to obtain a solution of 1 × 106 spores·mL-1 or with 2.3 mL of PDB to obtain a solution of 3 × 105 spores·mL-1 (Fu et al., 2017; Hang et al., 2019). The stock solution of slightly acidic electrolyzed water was prepared by diluting HOCl (30 µL·L-1, pH 6.26, CNK Life Korea Inc., Korea) with distilled water. The stock was diluted to obtain solutions at the indicated concentrations, which were examined using a photometer (HI96771 Ultra High Range Chlorine Portable Photometer, ISM, Hanna Instruments Inc., USA) and a pH meter (Orion Star A211 pH Benchtop Meter, Thermo Scientific, USA).
Botrytis cinerea Inoculation and Pulsing Treatment of Petals or Flowers
To confirm the applicability of slightly acidic electrolyzed water to rose petals, three outer perianths per ‘Dominica’ stem were randomly selected in five repetitions. Petals were treated by spraying a spore suspension of 3 × 105 spores·mL-1 at 1.2 mL·flower-1, following by air-drying for one hour (Hwang et al., 2019; Lee and Kim, 2020). Petal treatments were designated as no treatment (NT, no petal pulsing treatment, and control) and HOCl treatments at concentrations of 0, 5, 10, 20, and 30 µL·L-1, into which the petals were dipped for three seconds and then allowed to dry for one hour. On day three after the treatment, gray mold incidence was measured in a controlled room at an air temperature of 21°C, RH ≥ 90, and 15.4 µmol·m-2·s-1 PPFD with a 12h photoperiod. Cut rose flowers were used in the experiments after confirming the applicability of the slightly acidic electrolyzed water to the petals. The ‘Bubble Gum’ cut roses were dried for one hour after inoculating them with gray mold (the same mold used in the petal experiment). The petal pulsing treatment of the cut rose flowers, for which the flower buds were dipped into the solution for three seconds and then dried for one hour, was done as HOCl treatments at concentrations of 0, 10, 20, and 30 µL·L-1 for three seconds (Lee and Kim, 2020). Each flower stem was placed in a vase containing 400 mL of distilled water in a 1 L glass bottle, and a completely randomized block design was used with nine replications in controlled chambers (21°C air temperature, ≥ 90% RH, and 15.4 µmol·m-2·s-1 PPFD with a 12h-photoperiod). After the treatment on day three, the disease incidence rate (%) and visual index of B. cinerea were measured (Lee et al., 2016).
Evaluation of the Cut Flower Quality
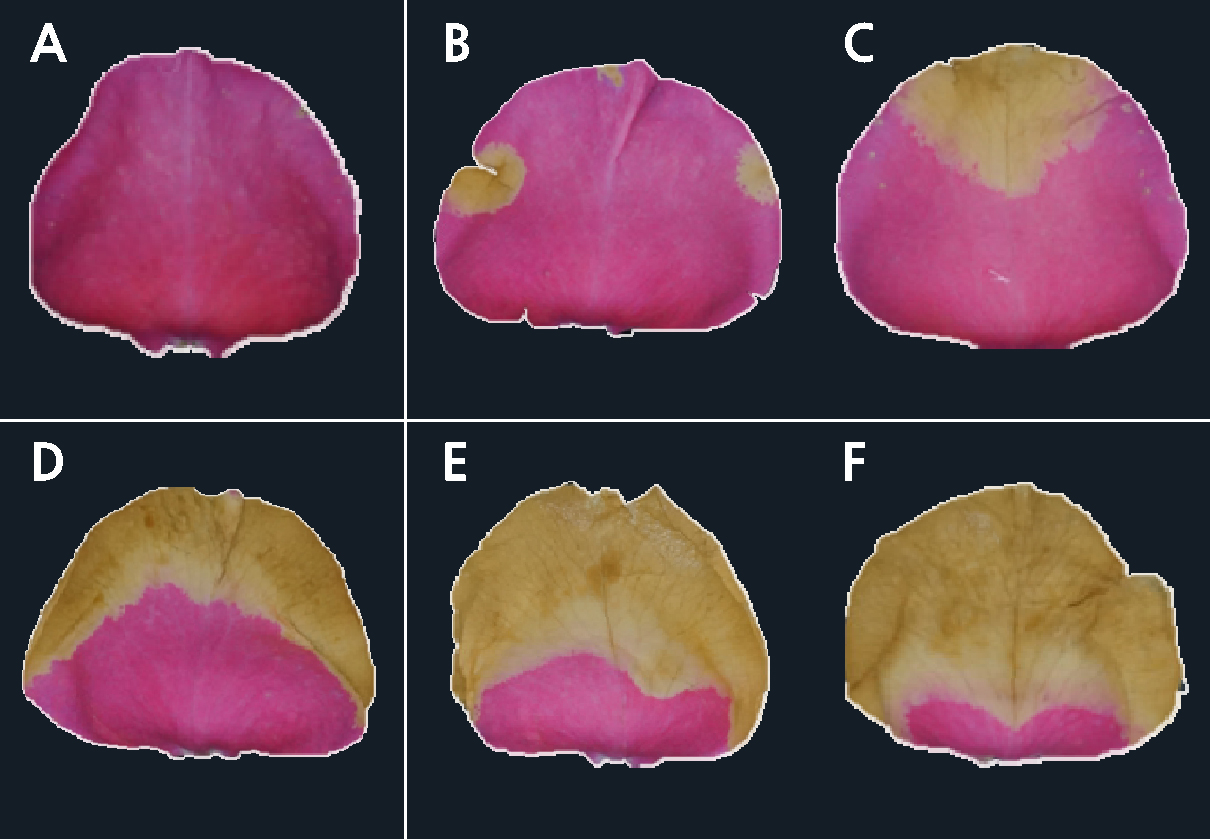
The disease incidence rate in the petals was measured in six stages by examining the rose petals blighted by B. cinerea infection. These were sage 0, no visible symptoms; stage 1, 15%; stage 2, 25%; stage 3, 50%; stage 4, 75%; and stage 5, 100% infection (Fig. 1). The incidence of gray mold on the cut flowers was calculated as the ratio of the number of diseased petals to the total number of petals, expressed as a percentage (Lee and Kim, 2020). The visual index of B. cinerea on cut roses was monitored in the following four stages: 0, no infection; 1, 1–25%; 2, 26–50%; and 3, 50–100% infection (Ha et al., 2020b). Vase life was recorded as the number of days from harvest (day 0) until the flowers began to exhibit vase life termination symptoms such as wilting petals or leaves with more than 50% of turgor loss and/or petal and leaf abscission or bending of the peduncle by a neck angle greater than 30° (Fanourakis et al., 2013). The flower color was measured according to the L* value (0, dark; 100, light), a* value (red-green; + red; –green), and b* value (yellow-blue; + yellow; –blue) using a color spectrometer (CR-400, Minolta, Japan). The ΔE* value represents a comparison of the flower color at the time of harvest with that at the time of vase life termination (McGuire, 1992). The rate of change of the flower size was expressed, in percentage, as the ratio of the size of the widest flower on day 6 after treatment to that on day 0 (Kim and Lee, 2022; Yeon et al., 2022).
To determine electrolyte leakage of the petals, an indicator of petal tissue damage, ten petals with diameters of 1 cm were placed in 20 mL deionized water and incubated with a shaking incubator (ThermoStable TM IS-20, DAIHAN, Korea) at 160 rpm for 18 hours at 24°C. After the initial conductivity was measured, the petals were incubated at 121°C for 20 minutes in an autoclave (MaXterile TM, DAIHAN, Korea) and then incubated while shaking at 160 rpm for 18 hours, with the final conductivity then measured (Lee et al., 2016) as follows: Electrolyte leakage (%) = [(Initial conductivity –deionized water conductivity) / (Final conductivity–deionized water conductivity)] × 100.
The chlorophyll contents of the leaves were measured using a soil plant analysis development (SPAD) chlorophyll meter (SPAD-502Plus MINOLTA, Japan). Chlorophyll fluorescence (Fv/Fm) was measured on the first edge of three-leaflet leaves using a portable chlorophyll fluorometer (MINI-PAM-II, WALZ, Germany) equipped with a leaf-chip holder for 20 minutes (WALZ, Germany) according to a method by An et al. (2022). The rate of change of the stomatal size was modeled by applying nail polish to the back of the first edge of the three-leaflet leaves, with subsequent observation at ×400 (Primostar 3, ZEISS, Germany) after attaching the sample to a slide glass (Microscope slides, SciLab, Korea; Lee et al., 2020). Measurements were taken of the stomatal pore and stomatal complex by measuring the size in dark and light conditions for 12 hours and calculating the pore size change rate via the following equation (Yi et al., 2018; Choi and Lee, 2020):
Change rate of stomatal size = (stomatal size under conditions for 12 h / stomatal size under cancer conditions for 12 h) × 100 (Yi et al., 2018; Choi and Lee, 2020).
Experimental Design and Data Analysis
A completely randomized block design was used. Analysis of variance and Duncan’s new multiple range tests (DMRT) as post-hoc analysis methods were conducted using SAS software (Statistical Analysis System, version 9.4, SAS Institute Inc., Cary, NC). Differences with p values lower than 0.05 were considered significant.
Results and Discussion
Lee et al. (2006) confirmed the importance of a postharvest fungicide treatment, as fungal infection is highly likely to occur after harvest if the petal treatment for inhibiting B. cinerea relies only on a pre-harvest treatment. We conducted tests to determine the proper pulsing treatment concentration of HOCl after harvest on the petals to inhibit gray mold. Compared to the no treatment (NT), the petal pulsing treatment with 0–30 µL·L-1 HOCl suppressed gray mold in ‘Dominica’ petals (Table 1). The treatment effect of the petals was highest at 30 µL·L-1, HOCl, resulting in a 65.6% reduction in B. cinerea compared to the NT outcome.
Table 1.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the incidence of Botrytis cinerea infection on day three after a petal pulsing treatment of ‘Dominica’ cut rose flowers
| Petal pulsing treatment (µL·L-1) | The disease incidence rate in petal (%) |
| NTz | 90 ay |
| HOCl 0 | 47 c |
| HOCl 5 | 70 b |
| HOCl 10 | 41 c |
| HOCl 20 | 36 c |
| HOCl 30 | 31 c |
In the ‘Bubble Gum’ cut rose flowers, the petal pulsing treatment at 30 µL·L-1 HOCl (27.8%) reduced B. cinerea by 71% compared to NT (94.6%) and reduced it by 68% compared to the 0 µL·L-1 HOCl (87.9%; Table 2). In rose ‘Dominica’ petals, the 0 µL·L-1 HOCl treatment reduced the disease incidence rate in petals compared to NT (Table 1). A tap water treatment suppresses the mycelial growth of B. cinerea in vitro, but the decay index of B. cinerea in vivo did not affect cut rose petals compared to NT (Lee et al., 2016). This study found that the treatment effect of 0 µL·L-1 HOCl differs depending on the cultivar and on the treated parts, such as petals or cut rose flowers (Tables 1 and 2). The petal pulsing treatment of distilled water or tap water without a sterilization effect had no treatment effect (Table 1). The vase life of cut rose flowers was lowest in the NT and highest in the flowers treated with 30 µL·L-1 HOCl. The disease incidence rate according to a visual index of 0 (no B. cinerea infection) was highest on flowers treated with 30 µL·L-1 HOCl, and the disease incidence rate of 3 (maximum B. cinerea infection) was highest in the NT (Fig. 2). The disease incidence rate decreased as the HOCl concentration was increased (Figs. 2 and 3). Slightly acidic electrolyzed water has high oxidation reduction potential (ORP-reaction) and dissolved chlorine (HOCl), which damages the microbial cell membrane and inactivates the defense mechanism of the microbe and oxidizes it (Schaik, 2009; Issa-Zacharia et al., 2011). Slightly acidic electrolyzed water had an antibacterial effect not only on fungi but also on certain bacterial species, such as Salmonella spp. and Yersinia enterocolitica in cabbage, Chinese celery, daikon sprouts, and lettuce (Issa-Zacharia et al., 2011; Yoon et al., 2021). The arrangement of water molecules is electrochemically altered, which allows better penetrability and interaction of the microbicidal ions. This feature is not found in conventional fungicides (Schaik, 2009). NaOCl, the most commonly used fungicide, produces disinfection byproducts and is unstable under conditions with dynamic changes in the pH, temperature, organic matter content, and light (Kim et al., 2009). Ahn (2015) found that slightly acidic electrolyzed water could replace NaOCl in treatments intended to reduce general bacteria and coliform infection levels in pickled radishes.
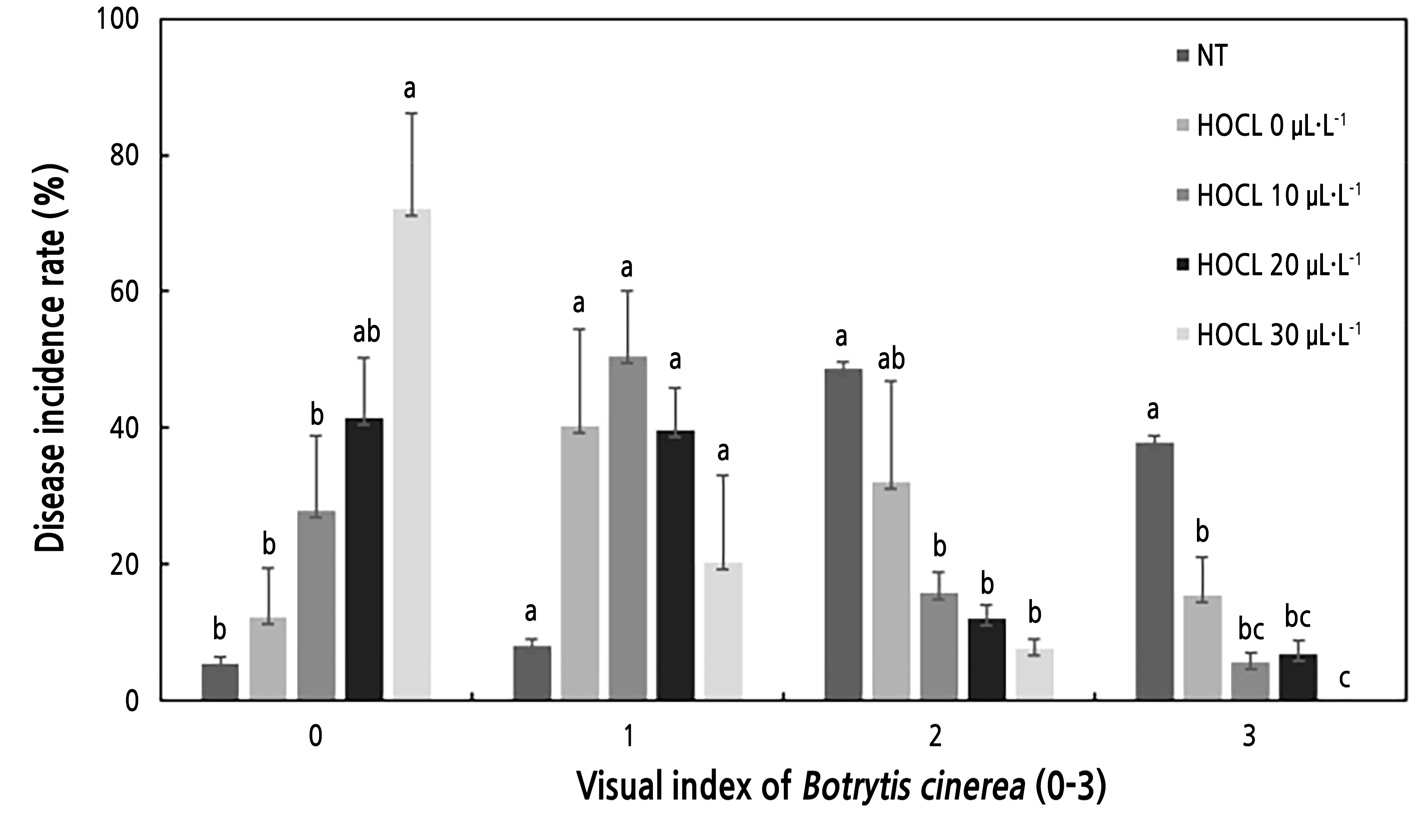
Fig. 2.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the disease incidence rate according to a visual index of B. cinerea on day three after the petal pulsing treatment of ‘Bubble Gum’ cut rose flowers. The visual index of B. cinerea indicated the following 0: no infection; 1: 1–25%; 2: 26–50%; and 3: 50–100% infection (Ha et al., 2020b). Error bars denote the standard errors (n = 3). Values with different letters are significantly different as analyzed by Duncan's multiple range test (p < 0.05). Flowers were treated by spraying a spore suspension of 3 × 105 spores·mL-1 at 1.2 mL·flower-1, followed by air-drying for one hour. NT means no petal pulsing treatment. Petal pulsing treatments were done as HOCl for three seconds with air-drying for one hour.

Fig. 3.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the visual index of B. cinerea on day three after a petal pulsing treatment of the cut rose flowers (A) and petals (B) of the ‘Bubble Gum’ type. The visual index of B. cinerea indicates the following: 0: no infection; 1: 1–25%; 2: 26–50%; and 3: 50–100% infection (Ha et al., 2020b). Flowers were treated by spraying a spore suspension of 3 × 105 spores·mL-1 at 1.2 mL·flower-1, followed by air-drying for one hour. NT means no petal pulsing treatment. Petal pulsing treatments were done as HOCl for three seconds with air-drying for one hour.
Table 2.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the vase life and incidence (%) of gray mold on day three after a petal pulsing treatment of ‘Bubble Gum’ cut rose flowers
| Petal pulsing treatment (µL·L-1) | Incidence (%) of gray mold | Vase life (days) |
| NTz | 94.6 a | 2.2 cy |
| HOCl 0 | 87.9 a | 3.0 b |
| HOCl 10 | 72.1 a | 2.6 c |
| HOCl 20 | 58.5 ab | 3.6 a |
| HOCl 30 | 27.8 b | 3.9 a |
A quality analysis of the cut flowers was conducted to assess whether the petal pulsing treatment with slightly acidic electrolyzed water induces petal damage. The petal color of roses is an important factor affecting the market value and quality of flowers (Roberts et al., 2003; Lim et al., 2011). The treatments did not cause any changes in the L* values, representing lightness (Table 3). Nor were there treatment-induced differences in the a* values, which represent the bluish-green to red-purple hue component, or in the b* values, which represent the yellow to blue hue components. The only difference noted was that between the NT and the day 0 treatments. NT exhibited the highest rate of color change as measured in ΔE* compared to the day 0 treatment, with the color being most stable in petals treated with HOCl. Discoloration of petals by gray mold is a significant factor causing a quality decline in Korean roses exported to Japan (Park and Park, 2006). The antifungal effects of the petal pulsing treatment with slightly acidic electrolyzed water did not cause any apparent damage to the petals, especially discoloration. Electrolyte leakage of the petals, which indicates petal tissue damage, was highest in the NT, and there was no petal tissue damage in petals treated with HOCl or distilled water (Table 4; Lee et al., 2016). Kim et al. (2009) suggest that hypochlorous acid water, including slightly acidic electrolyzed water, chlorine dioxide (ClO2), and ozonated (O3) water, is an alternative fungicide that can overcome the shortcomings of chlorine as a fungicide, with various advantages other than sterilization. As a result of simulating export distribution after a petal pulsing treatment with 5 µL·L-1 ClO2 for two seconds with a compound containing chlorine, gray mold was found to be suppressed without affecting the color, electrolyte leakage, or volatile compounds of the petals (Lee and Kim, 2020). The process of the petal pulsing treatment is identical to that of the petal dipping treatment. The petal pulsing treatment of cut roses with sulfur dioxide (SO2) and potassium permanganate (KMnO4) followed by dry transportation to cartons suppressed gray mold (van Jaarsveld, 2018). The petal pulsing treatment with 50°C hot water suppressed B. cinerea but induced various forms of petal damage, such as increased respiration, absorption, and reduced vase life at higher temperatures and longer treatment times (Lee et al., 2016). Therefore, a petal pulsing treatment that suppresses disease without affecting the quality of the petals is needed.
Table 3.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the flower color on day three after a petal pulsing treatment of ‘Bubble Gum’ cut rose flowers
| Petal pulsing treatment (µL·L-1) | Hunter valuez | ΔE*y | ||
| L* | a* | b* | ||
| day 0x | 40.95 aw | 50.63 a | 13.45 b | 0.00 c |
| NTv | 46.92 a | 27.72 b | 21.33 a | 23.34 a |
| HOCl 0 | 43.60 a | 46.60 a | 11.77 bc | 10.81 b |
| HOCl 10 | 40.77 a | 46.38 a | 12.36 bc | 7.52 bc |
| HOCl 20 | 43.40 a | 50.80 a | 11.41 bc | 3.83 bc |
| HOCl 30 | 39.38 a | 45.04 a | 9.73 bc | 4.54 bc |
The test room was maintained under the following conditions: air temperature 21°C, RH ≥ 90, and 15.4 µmol·m-2·s-1 PPFD with a 12h photoperiod.
Flowers were treated by spraying a spore suspension of 3 × 105 spores·mL-1 at 1.2 mL·flower-1, followed by air-drying for one hour.
The flowering rate of cut roses at the end of their vase life is one of the critical measures of the quality of cut flowers (Lee et al., 2005). In this study, the flowering rate was lowest in the NT, and there were few differences among the treatments (Table 4). There were no significant differences among the treatments onchlorophyll content and Fv/Fm of the leaves, indicating no damage to the quality of the leaves. In healthy plants, the Fv/Fm ratio is 0.80–0.83, decreasing if the photosynthetic reaction center in photosystem II is damaged or exposed to stress (Jung, 2002). In all treatments, the Fv/Fm was 0.81–0.83, indicating that the photosynthetic reaction center of the leaf was not damaged or not compromised due to the treatments (Table 4). The stomatal size change rate of the leaf is related to stomata opening and closing (KSFS, 2021). The smaller it is, the higher the moisture loss rate through the stomata, which shortens the vase life of cut roses (Fanourakis et al., 2013). The stomatal size change rate of the complex increased upon a treatment with 30 µL·L-1 HOCl by 45.4 and 47.9% compared to the NT or 0 µL·L-1 HOCl, respectively, but there were no significant differences among the treatments (Table 4). In conclusion, a postharvest petal pulsing treatment of cut rose flowers with 30 µL·L-1 HOCl reduced B. cinerea. It did not cause any apparent damage to the petals, such as discoloration or tissue damage, and did not affect the flowering rate and leaves, as measured by the chlorophyll content, Fv/Fm, and stomatal size change rate. Overall, these results suggest that immediate petal pulsing with slightly acidic electrolyzed water with 30 µL·L-1 HOCl is feasible for inhibiting gray mold on cut rose flowers.
Table 4.
Antimicrobial effect of slightly acidic hypochlorous water (HOCl, pH 6.26) on the rate of change of the flower size, electrolyte leakage of the petals, and the chlorophyll contents, chlorophyll fluorescence (Fv/Fm), and rate of change of the stomatal size of the leaves on day three after a petal pulsing treatment of ‘Bubble Gum’ cut rose flowers
|
Petal pulsing treatment (µL·L-1) |
Change rate of flower size |
Electrolyte leakage (%)z |
Chlorophyll contents | Fv/Fm | Change rate of stomatal size (%)y | |
| pore | complex | |||||
| NTx | 99.79 bw | 31 a | 40.5 a | 0.82 a | 134.51 a | 128.62 a |
| HOCl 0 | 111.41 ab | 25 b | 42.3 a | 0.83 a | 160.87 a | 126.48 a |
| HOCl 10 | 105.60 ab | 28 ab | 42.1 a | 0.83 a | 105.67 a | 128.03 a |
| HOCl 20 | 113.66 a | 25 b | 40.5 a | 0.81 a | 116.10 a | 111.28 a |
| HOCl 30 | 107.03 ab | 24 b | 42.8 a | 0.83 a | 125.59 a | 187.06 a |
The test room was maintained under the following conditions: air temperature 21°C, RH ≥ 90, and 15.4 µmol·m-2·s-1 PPFD with a 12h photoperiod.
Flowers were treated by spraying a spore suspension of 3 × 105 spores·mL-1 at 1.2 mL·flower-1, followed by air-drying for one hour.
yThe stomatal size change rate is calculated via (stomatal area on light / stomatal area on dark) × 100.
Petal pulsing treatments were done by dipping the petals for three seconds, followed by air-drying for one hour.
wMean separation within columns according to Duncan's multiple range test at p < 0.05. There were ten replications for the assessments of the change rate of the flower size, chlorophyll contents, and Fv/Fm via a non-destructive analysis and three for the assessments of the electrolyte leakage and rate of change of the stomatal size via a destructive analysis.