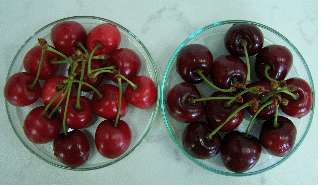Introduction
Materials and Methods
Plant material and LPE application
Pomological analysis
Sample preparation for phytochemical analysis
Analytical procedures
Total monomeric anthocyanins (TMA)
Determination of total antioxidant activity by (TEAC)
Determination of pH and total acidity
Statistical analysis
Results and Discussion
Introduction
Turkey is a leading country in sweet cherry production with total production of more than 400,000 tons and exports about 60,000 tons annually. Some favorable cir-cumstances of Turkey in sweet cherry market come from the following reasons; it is possible to grow sweet cherry both in early and late season in Turkey, due to its wide range of ecological and geographical conditions. Especially, labor cost for harvest is relatively cheaper in Turkey as compared to many other sweet cherry producing countries. Fruit cracking is not a major issue in Turkey due to the suitable weather conditions during the cherry growing season. The main advantage of significant export is the popular cultivar named as ‘0900 Ziraat’. This local cultivar is now known in world-wide to be sweet, dark skin colored, pink to red in flesh, crack resistant and very large in fruit size (Demirsoy and Demirsoy 2003). It is very firm with excellent flavor as well. ‘0900 Ziraat’ is known as mid- to late-season variety that ripens between ‘Bing’ and ‘Lapins’. These quality sweet cherries are exported in good price as a high value crop.
Lipids have been thought to play crucial roles in membrane structure and energy reserves. It is now evident that lipids and their metabolites play important role in other critical cellular functions particularly as mediators in cell activation, signal transduction, and cell proliferation (Cowan, 2006; Divecha and Irvine, 1995; Ryu et al., 1997). Lysophosphatidy-lethanolamine (LPE) is a natural product of membrane phospholipid metabolism and is formed from phosphatidy-lethanolamine (PE) by action of phospholipase A2 and remains in the lipid phase because LPE has a hydrophobic group such as fatty acid. It has been shown to control and manage quality of flowers, fruits, and other horticultural products. Studies have provided evidence that LPE can accelerate ripening of tomato and cranberry fruits while prolong shelf-life at the same time (Farag and Palta, 1993b; Özgen et al., 2004). LPE treatment has also been found to reduce senescence of leaves, fruits and cut-flowers (Cowan, 2009; Farag and Palta, 1993a; Hong et al., 2009a; Kaur and Palta, 1997). LPE retards polygalacturonase- mediated fruit softening (Hong et al., 2008), reduces activity of phospholipase D (PLD; EC 3.1.4.4) and membrane leakiness (Ryu et al., 1997; Hong et al., 2009b). Some other processes affected by LPE treatment include reduction of injury from stress and other xenobiotics (Özgen and Palta, 2003; Özgen et al., 2005), anthocyanin accumulation (Hong, 2008), and stimulus of ethylene synthesis (Hong et al., 2008). These results suggest a specific role of LPE both in ripening, anthocyanin accumulation and quality of fruits. In this study we investigated the use of LPE for accelerating ripening and enhancing phytochemical characteristics of high value crop sweet cherries.
Materials and Methods
Plant material and LPE application
Experiment was carried out in commercial sweet cherry orchard in Amasya, Turkey in 2011 and 2012 seasons. The experimental orchard was established with ‘0900 Ziraat’ and ‘Starks Gold’ was used as pollinizer. ‘0900 Ziraat’ was grown on Gisela-6 rootstocks and the trees were spaced at 5 × 4 m, trained to the vogel central leader system. The orchard was established in 2004. Trees were sprayed with 1,000 L/ha solutions by a turbo atomizer sprayer about 2 (the cherries on the canopy were in the blush stage) and 4 weeks before final harvest. Spray solution included 10 mg・L-1 of LPE (Doosan Coorperation, South Korea), commercially sold as “SignaFresh”.
Pomological analysis
Approximately 2 kg mature sweet cherries were randomly hand harvested from each experimental unit (10 trees each) and transferred to laboratory for analysis. 3 subsamples of 20 cherries were used for measurements of size (weight, width, length). Fruit samples were used to determine total soluble solid (TSS) contents by refractometery (Atago, Pal-1, Japan). The fruit color was measured using a Minolta portable chromameter (Minolta, Model CR-400, Japan) which provided CIE L*, a* and b* values. Chromameter describes color in three coordinates: L*, lightness, from 0 (black) to 100 (white); a*, from -60 (green) to 60 (red); and b*, from -60 (blue) to 60 (yellow). Chroma was calculated with the formula: (a*2 + b*2)1/2. Hue angle, in degrees, was calculated as h = tan-1 (b*/a*).
Fruit firmness was measured by a puncture test with a texture analyzer (Zwick/Roell, Materials Testing Machines/ BDO-FB 0.5 TS, UK) using 1.8-mm-diameter stainless steel round probe, at a speed of 50 mm・min-1 and with a 3 mm puncture distance. Firmness was expressed as maximum force needed for puncture, was measured at two locations on each fruit and was recorded as Newton (N).
Sample preparation for phytochemical analysis
Fruit slurry for phytochemical analysis was prepared as follows. Seeds of cherries were removed and homogenized in a standard food blender; for each replicate, excess fruits were used to minimize possible naturally-occurring fruit to fruit variation. Then, slurries were frozen immediately at -30°C until analyzed. For each fruit sample, 3 replicates were thawed at 25°C and RH 60%. All phytochemical analysis was completed within a month after harvest.
Analytical procedures
Determination of total phenolics (TP)
TP content was measured according to Singleton and Rossi (1965) procedure. An aliquot of fruit slurry was extracted with buffer containing acetone, water and acetic acid (70:29.5:0.5 v/v) for 2 hr in the dark. 3 parallel extracts were used. Then, extract, Folin-Ciocalteu’s reagent and water were incubated for 8 min, followed by adding 7% sodium carbonate. After 2 h, the absorbance was measured by an automated UV-VIS spectrophotometer (Model T60U, PG Instruments, UK) at 750 nm. Gallic acid was used as a standard. The results were expressed as μg gallic acid equivalent in g of fresh weight (GAE/g FW).
Total monomeric anthocyanins (TMA)
TMA were estimated by a pH differential method (Giusti and Wrolstad, 2005), using a UV-VIS spectrophotometer (Model T60U, PG Instruments, UK). Absorbance was measured at 533 nm and 700 nm in buffers at pH 1.0 and 4.5 using A = (A533 - A700) pH 1.0 - (A533 - A700) pH 4.5 with a molar extinction coefficient of 29,600. Results were expressed as μg of cyanidin-3-glucoside equivalents per g fresh weight (μg cy-3-glu/g FW).
Determination of total antioxidant activity by (TEAC)
Total antioxidant activity was estimated by TEAC (Trolox equivalent antioxidant capacity) assays. For TEAC assay, ABTS was dissolved in acetate buffer and prepared with potassium persulfate, as described by Özgen et al. (2006). The mixture was diluted in acidic medium of 20 mM sodium acetate buffer (pH 4.5) to an absorbance of 0.700 ± 0.01 at 734 nm for longer stability (Özgen et al., 2006). For the spectrophotometric assay, 2.97 mL of the ABTS (Azino-Bis- ethylbenzo-Thiazoline-Sulphonic acid) solution and 30 μL of fruit extract were mixed and incubated for 10 min and the absorbance was determined at 734 nm. Results were expressed as μmol of trolox equivalents per g fresh weight (μmol TE/g FW).
Determination of pH and total acidity
pH was determined with using a pH meter (Orion 3 star, Thermo Electron Co., Beverly, MA, USA). An aliquot of fruit slurry with a standard food blender was used for pH measurement. Total acidity was measured according to AOAC procedure (1975). 7.5mL of fruit slurry was poured into erlenmyer flask, and then, titrated with 0.01N NaOH to pH 7.0. Total acidity was expressed as percentage of acetic acid and calculated with following formula;
Total acidity (% acetic acid, w/v)
Statistical analysis
Experimental design was randomized block design with 3 replications for each treatment. Each experimental unit consisted of 63. Data were analyzed using SAS procedures and software (SAS, Cary, NC, USA). Means and standard deviations were obtained using TABULATE procedure. The Analysis of variance (ANOVA) tables were constructed all 15 variables tested in the study using GLM procedure. The mean separations were calculated for those significant variables by MEANS options of GLM procedure by Least Significance Difference (LSD) method at 5% significance level.
Results and Discussion
The results from two-year analysis indicated that among the 15 pomological and phytochemical characteristics and color measurements; the year effect was only significant for few variables as shown in Table 1 and 2. The year x treatment interaction was not significant for any of the variable tested indicated that the treatments were similarly affected by year factor. Thus, the data from 2011 and 2012 experimental years were combined in constructing ANOVA tables although the means were presented separately for two years.
Untreated LPE treated (10 mg・L-1 ) |
Fig. 1. Sweet cherry cv ‘0900 Ziraat’ treated with LPE (right) or untreated (left). |
Preharvest spray solution of LPE resulted in significant improvement on both pomological (fruit weight, size, color) and phytochemical (total phenolic content, antioxidant capacity) attributes of sweet cherry (Table 1 and 2; Figure 1). LPE treated fruits showed a 12% and 8% increase in 2011 and 2012 for fruit weight. Fruits treated with LPE had an average of 8.7 g in weight and 25 and 25 mm in width and length as compared to 7.2 g in weight, 23 and 23 mm in width and length for the untreated control when averaged over two experimental years. Although the mechanism of action by LPE was not exactly understood, earlier application of LPE may responsible for cell enlargement of sweet cherries. Earlier studies demonstrated that LPE acts as a signaling molecule and impacts either directly or indirectly membrane transport processes, ionic balance, homeostasis, and cell/organ turgor (Cowan, 2006). Fruit size is the main criteria for export quality of sweet cherries. Consequently, having an impact on fruit size directly reflects the value of products not only by the total yield but also by the higher quality standard.
Color characteristics of LPE treated and untreated fruits were compared in Table 2 and Figure 1. L* a* and b* values of sweet cherries treated with LPE were significantly lower than control fruits. These results indicated LPE treated fruits had darker red color than control fruits. Similar pattern was observed in chroma and hue angle values. These color values indicate LPE treated fruits accumulated higher anthocyanin than control fruits. It is also important to point out that we have not observed any product residue on dark red colored cherries treated with LPE.
LPE treatment had no significant effect on fruit firmness at harvest. However, in some of the earlier LPE trials in sweet cherries we have observed that LPE treated fruits were maintaining fruit firmness in cold storage (data not shown) as compared to control fruits. Similar results were observed in earlier LPE studies with tomatoes, cranberries and cut flowers (Farag and Palta, 1993a; Özgen et al., 2004; Kaur and Palta, 1997).
Sweet cherries had relatively low acidity; 0.71 and 0.74% was observed in treated and untreated control in 2011 and 0.68% and 0.70% in respectively. TSS content of LPE treated sweet cherries was determined relatively high with average of 15.7% as compared to 14.5% in 2011 and average of 15.4% as compared to 14.7% in 2012 resulting in 6% increase over the control.
Fruit phytochemical content and antioxidant capacity were increased significantly by LPE treatments. Total phenolic content of LPE treated fruits had 685 μg GAE/g FW compare to 540 μg GAE/g FW on untreated control in 2011 and 721 μg GAE/g FW compare to 598 μg GAE/g FW on untreated control in 2012. Fruits treated with LPE had 27% and 16% higher anthocyanin than control fruits in 2011 and 2012. Antioxidant capacity of fruits measured by TEAC assay displayed 13.3 and 11.9 μmol TE/g FW on LPE and untreated control fruits in 2011 and 11.7 and 10.9 μmol TE/g FW on LPE and untreated control fruits in 2012. Our results suggest that preharvest application of LPE may have the potential to accelerate anthocyanin accumulation, improve fruit quality and enhance phytochemical characteristics of sweet cherries. Our results were consistent with previous results where acceleration of color development and anthocyanin accumulation were observed in “Red Globe” and “Crimson” red colored table grapes (Hong, 2008) and cranberries (Özgen et al., 2004). Improved anthocyanin, color and TSS in fruits with LPE applications are also important for precocity of products.
Several researchers have previously reported that antho-cy-anins have remarkable antioxidant and free-radical scavenging activities (Stintzing et al., 2002; Wang et al., 1997). Additionally, multiple findings suggest that anthocyanin rich red fruits may provide possible health benefits such as reduced risk of stroke, coronary heart disease, certain types of cancers and aging (Zafra-Stone et al., 2007). Results of this study suggest that LPE may help to improve health benefits of sweet cherries by enhancing phytochemical profile of fruits.