Introduction
Materials and Methods
Plant Materials
Growth Survey of Licorice
Analysis of the Glycyrrhizin Contents of F1 Lines
RAPD Analysis of F1 Lines
Results
Phenotypic Variation among F1 Lines
Glycyrrhizin Content of F1 Lines
RAPD Analysis of F1 Lines
Discussion
Introduction
Licorice is a perennial herb belonging to the Leguminosae family. Approximately 18 licorice species are recognized worldwide; these species generally thrive in Asia, Europe, Australia, and the U.S. Among naturally growing species, Glycyrrhiza glabra (European licorice), G. echinata (Russian licorice), and G. uralensis (Chinese licorice) are mainly cultivated as medicinal herbs in Europe, Russia, and the Far East, respectively.
Licorice is utilized in Korea, China, and Japan as a traditional medicinal herb; in Korea, licorice is specifically used in the preparation of herbal medicines. The traditional Chinese pharmaceutics book,Botanical List, describes the medicinal benefits of licorice, including drug detoxification, harmonization of all medicinal ingredients, strengthening the digestive tract, and preventing food poisoning when used in its roasted form. However, this book cautions against the sole use of licorice because it can also cause whole-body swelling. Recent reports have described the beneficial effects of licorice, including its antibiotic, anti-inflammatory, anti-allergic, anti-cancer, and blood pressurereducing effects (Chung et al., 2001; Bai et al., 2003; Mae et al., 2003; Kim et al., 2006; He et al., 2006), as well as side effects such as edema, hypertension, and hypokalemia-induced myasthenia gravis (Cho et al., 2001).
G. uralensis, G. glabra, and G. inflata are recognized in China as medicinal plants, whereas in Japan, G. uralensis and G. glabra are considered to be beneficial herbs (Kondo et al., 2007). Korea also utilizes these three licorice species for medicinal purposes, although G. uralensis is the preferred herb. The main active ingredient (AI) of licorice plants is glycyrrhizin (Hayashi et al., 2000). The Korean Pharmacopoeia, Chinese Pharmacopoeia, and Japanese Pharmacopoeia have established a 2.5% threshold for the use of glycyrrhizin in medicinal remedies; however, the World Health Organization (WHO) recommends a glycyrrhizin threshold level of 4.0% (Kojoma et al., 2011). One problem associated with cultivated licorice is that its glycyrrhizin content is generally lower (< 2.5%) than that in its naturally growing counterparts, which could be as high as < 8.0% in mature plants (Yamamoto and Tani, 2002; Zhu et al., 2009).
The Annals of the Choson Dynasty and Dongeuibogam describe how Chinese licorice was introduced to Korea during the Choson Era under Sejong’s reign and planted in Sanglimwon; licorice was subsequently cultivated in the remaining provinces. The Dongeuibogam Annals reported that most Chinese licorice plants did not thrive well in most Korean soils, although those cultivated in Hamgyeong-Province were of good quality. European licorice was subsequently introduced in the 1970s; however, attempts to utilize it for pharmaceutical purposes failed. Research efforts on the domestic cultivation of this herb began with the import of Chinese licorice seeds in 1997, resulting in its successful cultivation and medicinal application in the 2000s.
From 2009 to 2011, the average wholesale price of licorice amounted to approximately 21,074 KRW/kg, with an average annual import value of approximately 3,700,000 USD. From the perspective of production, the principal regions of licorice cultivation during this time were Jecheon (Province Chungbook), as well as Sangjoo and Youngjoo (Province Kyungbook), with a total cultivation area of 43 ha. Annual licorice production amounted to only 262 tons due to its low yield per unit area and the limited cultivation area; this accounted for only a tiny portion of the domestic demand for licorice of 6,000 tons (MIFAFF, 2011). Moreover, the glycyrrhizin content of licorice cultivated in Korea has generally been lower than the reference level set by the Korean Pharmacopoeia; a major portion of the domestic licorice crop has been used as a natural sweetener.
Inherent heterosis, also known as hybrid vigor, is a genetic mechanism that is mainly responsible for the agriculturally superior genetic traits observed in F1 hybrids. Increases in crop production and AI content are the most common F1 traits controlled by heterosis (Burt et al., 2011; Kosev et al., 2012). Several F1 hybrids showing high levels of heterosis include major crops such as bean, rice, and corn (Hoecker et al., 2008; Jelena et al., 2011; Song et al., 2011). However, no research on the cultivation of F1 hybrids possessing phytopharmaceutical features has been performed to date. Recent attempts have been made to cultivate heterotic hybrids of Platycodon grandiflorus (Shi et al., 2011)
Medicinal plants have become highly valued as potential herbal medicines in many countries. Thus, various molecular markers are development such as RAPD, ITS/NTS, SCAR (Kim et al., 2015; Moon et al., 2016). They have been used for authenticate and distinguish plants from their relatives.
In this study, to overcome the problems associated with domestically cultivated licorice, such as low productivity and low glycyrrhizin content, we generated interspecific hybrids of European licorice and Chinese licorice, taking into consideration the effects of heterosis. We compared and analyzed various traits of the hybrids, including AI content, and performed RAPD analysis on the 32 F1 licorice lines.
Materials and Methods
Plant Materials
European licorice (G. glabra L.; female parent, collected in China) and Chinese licorice (G. uralensis Fisch; male parent, collected in China) were planted in a greenhouse and artificially crossed in May 2007 in a field (National Institute of Horticultural and Herbal Science, RDA, Suwon, Korea). In September 2007, germinated seeds obtained from the cross were retrieved and sown in the greenhouse. In June 2008, stolons were separated from the F1 licorice seedlings and cultivated, resulting in the production of 32 clonal lines of interspecific hybrids. In May 2013, stolons were separated from the 32 F1 lines and transplanted in the field (National Institute of Horticultural and Herbal Science, RDA, Eumseong, Korea). In April 2014, the roots of the F1 lines were collected for phenotypic analysis (Fig. 1). Lines Korea #1 and #2 are G. uralensis lines collected at Yeongcheon (30 samples) and Jecheon (20 samples), Korea. China #1 and #2 are G. uralensis lines purchased from the Korea Medicine Herbal Association (five samples) and Kyungdong market, Korea (12 samples). Neimenggu is a G. uralensis line collected in Neimenggu (10 samples), China (Table 2).
Growth Survey of Licorice
Approximately 10 roots from each F1 line were collected, and the root diameters and fresh weights were measured. The roots were then dried at 50°C for 10 days, after which the dry weights were measured (Table 1). The dried roots were also used to measure glycyrrhizin contents.
Table 1. Root characteristics and glycyrrhizin contents of F1 lines and their parents.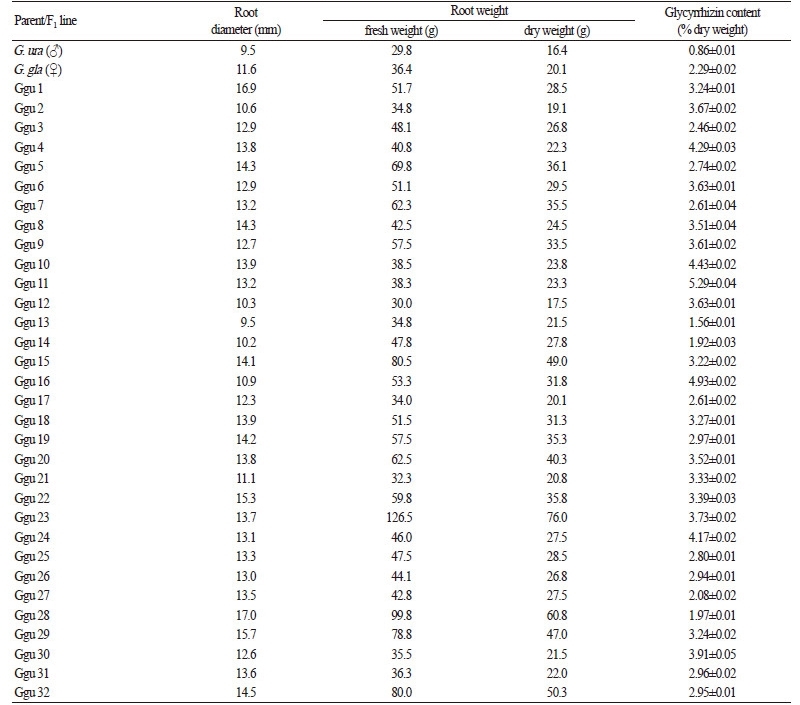 |
*Data are presented are the mean ± SD of more than three repliactes. |
Analysis of the Glycyrrhizin Contents of F1 Lines
Vacuum concentration was conducted on 10 g root samples from each F1 line that had been dissolved in 200 mL of methanol. Each root sample was extracted three times. Approximately 10 mg of each concentrate was diluted in 1 ml of methanol and used for high performance liquid chromatography (HPLC) analysis. As a reference, 1 mg of glycyrrhizin (Sigma-Aldrich) was dissolved in 1 mL of methanol and analyzed by HPLC using an Agilent 1100 Series HPLC System (Agilent Tech., USA) and a Zorbax SB-C18 HPLC column (4.6 × 250 mm, 5 μm, Agilent Tech., USA). The injection volume of the sample was 10 μL, and the flow rate was set at 1 mL/min. An ultraviolet (UV) detector with a fixed wavelength of 254 nm was used. The mobile phase was A: 100% acetonitrile and B: 0.5% H3PO4 with H2O, with a gradient of A 30–100%. The following time course was performed: 0–30 min, 30–100% A; 30–37 min, 100% A; and 38–42 min, 30% A.
RAPD Analysis of F1 Lines
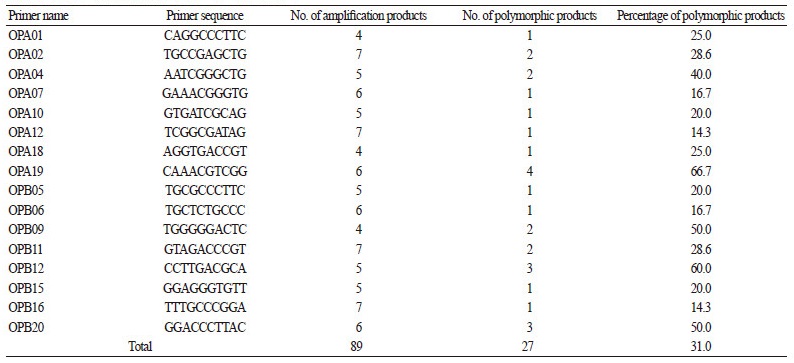
Forty decamer primers (OPA1-20 and OPB1-20, Operon Technologies, USA) were screened by RAPD analysis to detect genetic variation. Eighteen primers were selected based on the production of reproducible banding patterns and used throughout the study: OPA01, OPA02, OPA04, OPA07, OPA10, OPA12, OPA18, OPA19, OPB05, OPB06, OPB09, OPB11, OBP12, OPB15, OPB16, and OPB20 (Table. 3). PCR of genomic DNA from the F1 lines and their parents was conducted using the RAPD primers under the following conditions: 94°C or 5 min, 25 cycles of 94°C for 30 s, 32-34°C for 30 s, 72°C for 1 min, and a final extension at 72°C for 5 min. Amplification was conducted in a 20 μl reaction mixture containing 10 μl of Prime Taq Premix (Genet Bio, Korea), 2 μl of each primer set (10 pmol/μl), and 500 ng of template DNA. The DNA amplification products were separated on 1.5% agarose gels, after which the fragments were visualized under 300 nm UV light (after ethidium bromide staining) and photographed. Dendrograms were constructed by the unweighted pair-group method with arithmetic average (UPGMA) cluster analysis (Sneath and Sokal, 1973) using MEGA software ver. 4.1 (http:// www.megasoftware.net).
Forty decamer primers (OPA1-20 and OPB1-20, Operon Technologies, USA) were screened by RAPD analysis to detect genetic variation. Eighteen primers were selected based on the production of reproducible banding patterns and used throughout the study: OPA01, OPA02, OPA04, OPA07, OPA10, OPA12, OPA18, OPA19, OPB05 , OPB06, OPB09, OPB11, OBP12, OPB15, OPB16, and OPB20 (Table. 3). PCR of genomic DNA from the F1 lines and their parents was conduct edusing the RAPD primers under the following conditions: 94°C or 5 min, 25 cycles of 94°C for 30 s, 32–34°C for 30 s, 72°C for 1 min, and a final extension at 72°C for 5 min. Amplification was conducted in a 20 μl reaction mixture containing 10 μl of Prime Taq Premix (Genet Bio, Korea), 2 μl of each primer set (10 pmol/μl), and 500 ng of template DNA. The DNA amplification products were separated on 1.5% agarose gels, after which the fragments were visualized under 300 nm UV light (after ethidium bromide staining) and photographed. Dendrograms were constructed by the unweighted pair-group method with arithmetic average (UPGMA) cluster analysis (Sneath and Sokal, 1973) using MEGA software ver. 4.1 (http:// www.megasoftware.net).
Results
Phenotypic Variation among F1 Lines
Leaves of the licorice F1 lines showed characteristics of both the European licorice and Chinese licorice parental plants; some leaflets resembled the leaflets of either parental plant at a level of 50%, whereas others had entirely different shapes from those of both parental plants (Fig. 2). In terms of roots, Chinese licorice cultivated by sowing generally had taproots, whereas the main roots of the F1 lines had various shapes because the plants were cultivated from planted stolons. A comparison of cross-sections of roots of the F1 progeny versus Chinese licorice parental plants revealed that the roots of the F1 plants possessed a stronger yellow hue (Fig. 2).
The dry weights of 30 of the 32 F1 lines (except for Ggu 2 and Ggu 12) were higher than those of the European and Chinese licorice parental plants. The root diameters of the 32 F1 lines were larger than those of the parental Chinese licorice line; however F1 hybrid lines Ggu2, Ggu12, Ggu13, Ggu14, Ggu16, and Ggu21 had smaller root diameters than European licorice (Table 1). Comparisons of fresh weight versus dry weight showed that the drying rates ranged from 39 to 46%.
Glycyrrhizin Content of F1 Lines
For 25 (78.1%) of the 32 F1 lines, the glycyrrhizin content was >2.5%; that of four lines (Ggu 8, Ggu 15, Ggu 30, and Ggu 31) even exceeded the 4.0% glycyrrhizin level established by the WHO. The average glycyrrhizin content of the F1 lines was 3.2%, which is higher than those of the parental plants. Four hybrid lines (Ggu3, Ggu 13, Ggu14, and Ggu28) failed to reach the glycyrrhizin threshold level of 2.5% set by the Korean Pharmacopoeia (Table 1).
To compare the glycyrrhizin content among the F1 lines, we also analyzed the glycyrrhizin content of domestically cultivated and imported licorice (Table 2). Licorice plants cultivated in China and Inner Mongolia (Neimenggu) had higher glycyrrhizin contents than domestically cultivated plants, and they generally satisfied the reference level of 2.5% set by the Korean Pharmacopoeia. One Chinese sample showed a glycyrrhizin content exceeding the 4.0% recommended threshold level established by the WHO.
Table 2. Glycyrrhizin contents of G. uralensis collected from a medicinal herb market in Korea. |
*Data are presented as the mean ± SD of more than three replictaes. |
RAPD Analysis of F1 Lines
Eighteen primers produced polymorphic bands via PCR: the percentage of polymorphic products was 25.0% for primer OPA01, 28.6% for OPA02, 40.0% for OPA04, 16.7% for OPA07, 20.0% for OPA10, 14.3% for OPA12, 25.0% for OPA18, 66.7% for OPA19, 20.0% for OPB05, 16.7% for OPB06, 50.0% for OPB09, 28.6% for OPB11, 60.0% for OPB12, 20.0% for OPB15, 14.3% for OPB16, and 50.0% for OPB20 (Table 3). Polymorphic bands that were clearly resolved on the gels were scored as 1 or 0 for present or absent, respectively, while monomorphic, ambiguous, and faint bands were regarded as missing data and were therefore ignored. The 16 selected primers generated a total of 89 bands, among which 27 specific bands were used to construct the dendrogram: 15 bands were amplified in GU, 17 were amplified in GG, and 27 were amplified in the Fl lines. Cluster analysis via UPGMA suggested that the F1 lines share high genetic similarity (Fig. 3).
Discussion
Licorice has traditionally been used as a medicinal remedy; however, due to its low productivity in Korea, most of the domestic demand for this crop relies on imports from China. Furthermore, domestically cultivated licorice has a low glycyrrhizin content and is therefore ineffective for medicinal use. To improve this situation, we generated an interspecific hybrid by crossing European licorice with Chinese licorice using traditional breeding methods and cultivated the resulting F1 lines.
The phenotypic characteristics of the hybrid lines included leaves of variable shapes resembling both parental plants, although the leaves were generally larger than those of the parents. A similar study of hybrids of European and Chinese licorice also revealed variations in leaf form (Hayashi et al., 2003a, b). In the current study, we compared the leaves of the F1 lines with those of natural hybrids; the leaves of F1 hybrids possessed unique characteristics, sharing features of both European and Chinese licorice plants. Although the hull surfaces of European and Chinese licorice are smooth, the hybrid leaves were covered with rough hairs. The identical phenotypes of hulls from natural hybrids and the F1 lines cultivated in the present study indicate that the lines were all interspecific hybrids (Suppl. Fig. 1). The success rate of obtaining seeds from interspecific breeding was shown to be <75% (Ashurmetov, 1996). Thus, the results of the current study suggest that European licorice and Chinese licorice can be naturally interbred.
Chinese licorice plants possess creeping stems, whereas those of European licorice are straight. Because most of the F1 progeny had straight stems, this phenotypic trait must have been derived from the female parent, European licorice. The yellow hue of licorice roots is attributable to the presence of flavonoids such as liquiritin and isoliquiritine (Yamamura et al., 1992). Our examination of root cross-section revealed that the F1 lines had a stronger yellow hue than the parental Chinese licorice, suggesting that the progeny contained higher flavonoid levels than any of the parental plants.
The glycyrrhizin content of Chinese and European licorice cultivated under similar conditions was 2.99 and 2.56%, respectively (data not shown), indicating that since Chinese licorice is not highly different from European licorice in terms of AI, it has responded favorably to local cultivation efforts. In general, the glycyrrhizin content of domestically produced Chinese licorice is lower than that of licorice produced in China and Inner Mongolia. This may be attributable to the fact that Chinese licorice does not grow naturally in Korea and is therefore considered to be a cultivated plant. The glycyrrhizin content of naturally growing licorice plants is 4.43 ± 1.32%, whereas that of cultivated licorice is 1.51 ± 0.49%; the glycyrrhizin content of cultivated licorice is thus below the reference level (Wei et al., 2012). The low glycyrrhizin content of cultivated licorice may be attributed to two main mechanisms. First, cultivated licorice is less exposed to stressful environmental conditions than wild licorice, and thus, it generates less glycyrrhizin, a secondary metabolic product. Second, most domestically cultivated licorice plants reach developmental maturity within one year, which may be temporally insufficient for the accumulation of glycyrrhizin in various parts of the plant.
The glycyrrhizin content of licorice produced in Korea was <2.5%, which is lower than the reference level set by the Korean Pharmacopeia; on the other hand, the glycyrrhizin contents of Chinese and Inner Mongolian licorice plants were above the reference level (Table 2). The glycyrrhizin contents of some Chinese cultivated licorice plants were below the reference level, which may be attributed to two scenarios: first, the imported licorice was cultivated, not naturally grown; and second, even for naturally growing licorice, the plants were generally less than one year old and thus, the amount of glycyrrhizin in the plants may have been lower than expected (Wei et al., 2012). In fact, an earlier study showed that both cultivated and naturally growing licorice plants from Inner Mongolia have glycyrrhizin levels of <2.5% (Zhu et al., 2009).
The F1 lines in our study showed variations in glycyrrhizin levels, ranging from 1.56% (Ggu13) to 5.29% (Ggu11), suggesting that heterosis affected not only the phenotypic characteristics of the F1 plants, but also the levels of the secondary metabolic product, glycyrrhizin. In addition, the hybridization experiments led to changes in other plant features such as root shape and productivity, which may be useful in the selection of varieties for further propagation. For example, the hybrid line Ggu11 showed the highest glycyrrhizin content (5.29%), although its root diameter and dry weight were the lowest among the F1 lines, thus indicating low productivity.
Thirty-two F1 lines were generated from interspecific hybridization of European licorice and Chinese licorice. We plan to perform productivity testing (regional yield trials) using hybrid lines with glycyrrhizin contents of <4.0%; the lines selected from this assay will be further cultivated in Korean soil. In addition, the remaining 23 lines whose glycyrrhizin content is >2.5% will be reused as breeding resources.
RAPD and UPGMA clustering analysis showed that the F1 lines are closely related, perhaps because the F1 lines were produced through artificial breeding from GU and GG. We compared the genetic distances of the F1 lines, which were divided into four groups. However, there were no significant trends between the groups, such as similar root diameters, root weights, or glycyrrhizin contents.
Further studies using markers such as RFLP, SSR, and SNP markers would help confirm the RAPD data, allowing us to develop markers for F1 hybrid lines.