Introduction
Materials and Methods
Collection of Samples and Stereomicroscopy
Culture Isolation and Light Microscopy
DNA Extraction and PCR Amplification
RT-PCR Amplification
Results and Discussion
Symptomatic Comparison between ABLS and ABD Leaves
Cultural Isolation and Microscopic Observation
PCR and RT-PCR Analysis
Introduction
According to the Korean Society of Plant Pathology, approximately 41 apple diseases have been reported in Korea (Kim et al., 2009). Among these, 32 are caused by fungal plant pathogens. The major apple leaf diseases are apple blotch caused by Marssonina coronaria, alternaria leaf spot caused by Alternaria mali, gray mold caused by Botrytis cinerea, rust caused by Gymnosporangium yamadae, powdery mildew caused by Podosphaeria leucotricha, silver leaf caused by Stereum ourpureum, and leaf spot caused by Leptosphaeria sp. These diseases damage apples leaves and affect fruit quality and yield. However, among these diseases the mosteconomically important in Korean orchards are apple blotch and alternaria leaf spot (Lee et al., 2007). Apple blotch diseases caused by M. coronaria have been reported in many countries including Korea (Lee and Shin, 2000; Sharma, 2000; Tamietti and Matta, 2003; Lee et al., 2011). Furthermore, Korean isolates were very similar to Japanese and Chinese isolates, where the disease has caused a significant production problem over the past few years (Lee et al., 2011). According to previous reports, the leaves are the main photosynthetic organs in plants, and as such, are closely associated with photosynthetic function. Apple leaves produce sucrose and starch as the primary products of photosynthesis, which affect fruit quality and plant vigor. The allocation and distribution of carbon resources from leaves plays a key role in how these resources are used in sink tissues like fruit, where they can affect downstream traits like fruit quality (Gianni et al., 2006). Therefore, many farmers use fungicides to manage and control leaf diseases. Important research has focused on the intensive management of these diseases through the development of a fungicidal spraying program (Lee et al., 2009).
During 2010-2014, apple blotch-like symptoms (ABLS) were observed on ‘Fuji’ apple leaves in several apple orchards located in Gyeongbuk Province, Korea. ABLS are characterized by the progressive yellowing of leaves, the appearance of brown and green spots, and defoliation of leaves, which are very similar to those observed in apple blotch diseased (ABD) leaves as a result of M. coronaria infection. Therefore, farmers misdiagnosed this disease and almost all cultivation areas were sprayed with fungicide several times from May to early June to control ABLS. However, fungicides were unable to control the ABLS. Furthermore, it was difficult to differentiate between these two diseases by eye. The only observable distinction of apple ABLS was its lack of dissemination ability in orchards, unlike ABD. However, there have been no previous reports on ABLS in Korea. Therefore, this study was conducted to investigate the cause of ABLS and to enable its distinction from ABD by eye, by microscopic and molecular methods.
Materials and Methods
Collection of Samples and Stereomicroscopy
Abnormal yellow leaves having brown and green spots were collected from 30 apple orchards located in Cheongsong, Gunwi, Yeongcheon in Gyeongbuk Province during 2010-2014. Over 70 samples were collected from each orchard, and were all visibly similar and difficult to differentiate from ABD leaves by eye. To observe the external leaf surface, approximately 30 samples of both ABLS and ABD leaves were examined under stereomicroscope (Dimis-M, Korea).
Culture Isolation and Light Microscopy
For culture isolation of ABLS and ABD, we cut the infected lesion, including brown spots, out of leaves into small pieces (3-5 mm) and surface sterilized in 70% ethanol and 1% (v/v) sodium hypochloride for 30 s, each followed by three rinses with sterilized distilled water (SDW). Dried pieces were placed on PDA plates and incubated at 25°C in the dark for 3 days. To examine the internal leaf structure, samples were fixed (Karnovsky’s fixative) and dehydrated in an ethanol series. Next, samples were infiltrated (propylene oxide) and polymerized in Spurr’s resin at 70°C for 8 h. After polymerization, 3 µm semi-thin sections were cut using an ultra-microtome (MT-7000, RMC), stained with methylene blue, and examined under a light microscope (BX-50, Olympus, Japan).
DNA Extraction and PCR Amplification
To perform PCR, total DNA was extracted from infected leaves using the cetyltrimethyl ammonium bromide (CTAB) method (Namba et al., 1993). The PCR primer set, Mar- M-for (5′-GCT TAT TCT TGG CAA GCC-3′)/Mar-M-rev (5′-CAC AGT CTT GGA GCA TCG-3′) was disigned from partial of putative sequences of the metallopeptidase gene with a predicted PCR product of 250 bp. Amplification reactions were performed using a thermal cycler 9700 (Applied Biosystems, Foster City, CA, USA) with the following program: an initial denaturation stage of 3 min at 94°C, followed by 30 cycles of denaturation for 30 s at 94°C, annealing for 30 s at 60°C, extension for 30 s at 72°C, and a final 3 min extension at 72°C. Amplified PCR products were electrophoresed on a 1% agarose gel using a 100-bp Plus DNA ladder as a marker, stained with ethidium bromide, and visualized on a UV transilluminator.
RT-PCR Amplification
Total RNA was extracted from abnormal yellow leaves with brown spots using an RNeasy mini kit (Qiagen, USA), according to the manufacturer’s protocol. RT-PCR was then conducted to detect apple chlorotic leaf spot virus (ACLSV), apple stem pitting virus (ASPV), apple stem grooving virus (ASGV), apple mosaic virus (ApMV) and apple skin viroid (ASSVd) using specific primers pairs, and conditions were adjusted as previously reported (Menzel et al., 2002; Shim et al., 2006; Hassan et al., 2006; Lee et al., 2001). RT-PCR was conducted using an Applied Biosystems 2720 Thermal Cycler (Applied Biosystems, USA) with AccuPower® RT-PCR Premix (Solgent, Daejeon, Korea). The RT-PCR products were then electrophoresed on a 1.5% agarose gel, stained with ethidium bromide, and visualized under UV light.
Results and Discussion
Symptomatic Comparison between ABLS and ABD Leaves
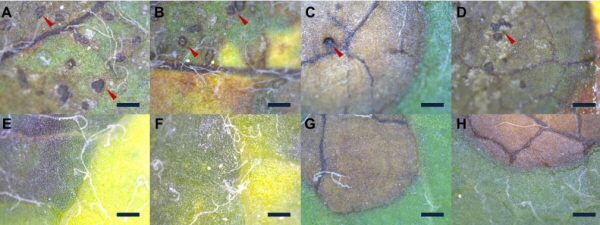
ABLS were observed on several farms located in Gyeongbuk Province during 2011-2014. Brown and dark brown spots appeared on infected leaves and led to their discoloration from green to yellow, and caused defoliation in early June (Fig. 1A-H). These symptoms are very similar to those of ABD, which also causes small black or brown spots to develop on the surface of leaves, causing discoloration and defoliation of apple leaves (Fig. 1I-L). The only difference was the formation of acervuli on ABD leaves. These acervuli were black and greenish in color and were observed only on ABD leaves. From the above observation, it was concluded that ABD and ABLS were very similar diseases, although it is difficult to differentiate between the two by eye. However, the presence of acervuli on ABD leaves enables us to differentiate between these two similar diseases by morphology alone.
Cultural Isolation and Microscopic Observation
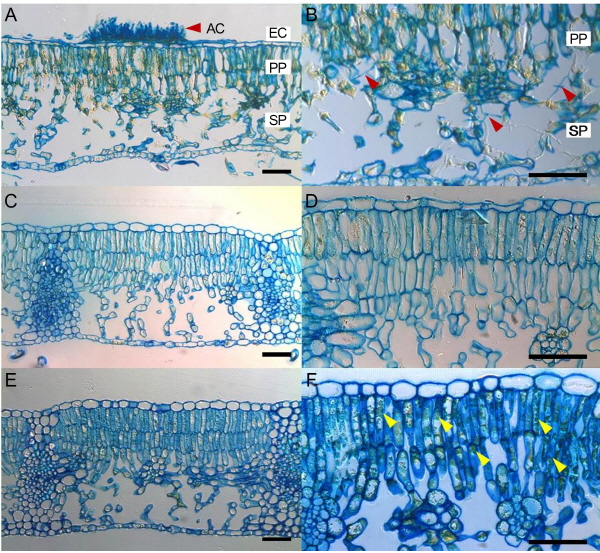
In the culture isolation method, ABLS and ABD leaves generated very different results and no pathogen was isolated from ABLS leaves cultured on PDA media (data not shown). However, in the case of ABD leaves, M. coronaria grew well on PDA media and was successfully isolated (data not shown). The microscopic study of abnormal ABLS and ABD leaves revealed very different morphologies caused by the two diseases. Typical apple blotch symptoms were observed on ABD leaves, which displayed brown spots that were actually acervuli on the external surface of leaves as previously reported (Lee et al., 2011; Back and Jung, 2014). However, the ABLS leaves also developed brown spots, although no acervuli were found on the surface of infected leaves (Fig. 2E-H). A similar disease as similar symptoms,having irregular, brown, necrotic areas between veins with no acervuli was reported (Sutton and Sanhueza, 1998; Table 1). According to Sutton and Sanhueza (1998), the disease has been given many common names, including Golden Delicious leaf blotch, necrotic leaf spot, physiological leaf spot, and necrotic leaf blotch. In our study, similar leaf spots were observed for the first time in ‘Fuji’ apple orchards in Korea. The microscopic observation of the internal leaf structure showed contrasting results after comparing cross sectional images of ABLS and ABD leaves. Large numbers of mycelia-like structures were observed in ABD leaves, which is evidence of M. coronaria infection (Fig. 3A-B). However, in ABLS leaves, mycelia-like structures were absent and a dense green structure was observed on the palisade parenchyma (Fig. 3C-F). Similar to the culture isolation method, cross-sectional analysis also showed that M. coronaria was not the causal agent of ABLS, because from microscopic observation it was confirmed that no acervuli or hypha exist on the surface of ABLS-infected leaves (Fig. 2E-H). Similarly, no mycelium was observed on the inner surface of ABLS leaves (Fig. 3C-F).
|
Fig. 4. PCR amplification of M. coronaria: lanes 1-3, leaves with apple blotch-like symptoms; lanes 4-6, apple blotch diseased leaves; M, 100 bp DNA ladder. |
PCR and RT-PCR Analysis
PCR and RT-PCR methods were used for the first time to observe ABLS infection and to compare it with diseases with known causal agents, such as M. coronaria and viruses. Our data revealed a 250-bp DNA fragment that was amplified from ABD leaves, whilst this fragment was not amplified from ABLS leaves (Fig. 4). Although, ABLS leaves have different characteristics from virus-infected leaves, due to the ambiguous characteristics of ABLS, we used RT-PCR to allow virus detection. However, similar results were observed using the RT-PCR detection method and no virus was detected in ABLS leaves (data not shown). Therefore, our results are similar with previous reports that this disorder is not caused by fungus or other known pathogen, and thus the abiotic cause of this disease remains unknown (Ellis, 2008; Table 1). Moreover, previous reports have suggested that abiotic causes such as air pollution and nutrient deficiencies do not appear to be involved (Jonkers, 1973; Kenderand andSpierings, 1975). Evidence suggests that the disease may be associated with a rapid synthesis of gibberellins that is triggered by a combination of environ-mental factors, including low light intensity, cool temperatures, and high soil moisture (Sutton and Clayton, 1974; Jonkers, 1973).
The present results confirm that ABLS is very different from ABD, and it may represent aphysiological disorder in plants. ABLS resulted in symptoms similar to previously reported leaf spot diseases caused by different pathogens (Yoder and Biggs, 1998; Rosenberger, 2001; Sutton and Sanhueza, 1998). The present results are also parallel with previous reports that leaf blotch begins as a pale green area in the leaf that turns to brown (Jonkers, 1973; Table. 1). According to this report, these blotches vary in size from 5 mm to several centimeters in diameter, and one to several blotches can occur on the same leaf (Table 1). The ABLS occurred in hot dry weather from May to June, which differs from the period in which the occurrence of other leaf spot diseases has been reported (Table 1). ABLS does not cause fruit drop or fruit blemishes. However, it may have adverse effects on fruit size because, from the present study, it was confirmed that ABLS infects the main photosynthetic organ of affected plants and causes defoliation in a very short time, which may disturb photo-synthetic function.
According to previous reports apple that apple leaves produce starch as a photosynthetic product, which has great influence on fruit quality and plant vigor (Gianni et al., 2006). In our study, ABLS caused defoliation and previous studies have shown that this disease occurs in distinct periods or in waves and can result in up to 70% defoliation (Jonkers, 1973; Babadoost, 1984). Furthermore, ABLS is similar to leaf spot disease caused by physiological disorders or abiotic factors that also caused defoliation of leaves in a short period of time (Table 1). No studies have reported the effects of this disorder on fruit size, because it is not known how to maintain the affected and unaffected trees in the field to allow for parallel comparisons to be made. Although it appears that ABLS is not caused by a fungus or bacterium and the causal agent of this disorder is unknown. Sometime a positive correlation of ABLS with fertilizer overuse was found in different orchards. Therefore, the possible factors of ABLS in Korean orchards are the overuse of fertilizer, hot and hazy weather following a humid rainy period. Different varieties respond differently to fertilizer application and nutrient level. Fertilizers play a key role in orchard management and relation to the growth rate of leaves; sometime high magnesium and potassium level causes defoliation in apple orchards. Therefore, the formers should follow proper fertilizer program to manage the crop nutrition that may be helpful to control ‘ABLS’ in future.