Introduction
Materials and Methods
Experimental Orchards and Application of CPPU and Urea
Physiological Measurements
Phytohormonal Analysis
Anatomical Analysis
Statistical Analyses
Results and Discussion
Physiological Changes of Persimmon Fruits in Response to CPPU and Urea
Effects of CPPU and Urea on Phytohormones
Anatomical Changes in Persimmon Fruits in Response to CPPU and Urea
Introduction
The Asian persimmon (Diospyros kaki Thunb.) originated in Eastern Asia, and many other cultivars have since been developed in China, Japan, and Korea (Cho and Cho, 1965; Wang et al., 1997). Asian persimmons are cultivated worldwide, including in Brazil, Israel, Italy, Spain, Azerbaijan, Uzbekistan, New Zealand, and Australia (FAOSTAT, 2015), but new cultivars have only been developed in Japan and Korea between 1959 and 2012 (Sato and Yamada, 2016). The cultivar ‘Fuyu’ stands out as highly adaptable with good productivity. It bears intermediate to large fruits (150–350 g) with a yellow-reddish coloration and a sweet flavor (Martins and pereira, 1989).
Calyx-end cracking (i.e. calyx separation) is a type of cracking specific to the Asian persimmon (Kitagawa and Glucina, 1984; Yamada and Sato, 2002). Calyx-end cracking is an important problem, which ultimately has a negative impact on fruit marketability. Splits reduce fruit quality owing to poor appearance, decreased shelf life, and fungal infection. Calyx-end fruit cracking can occur during rapid fruit growth, development, and ripening, and also during the postharvest period, mostly as a result of persimmon fruit spoilage.
Several factors promote calyx-end fruit cracking, including the intake of too much water at the end of the season during fruit development, seed numbers per fruit, and tree vigor. Some studies have also argued that genes related to cell wall development might be involved in fruit cracking (Wakasa et al., 2003; Kasai et al., 2008). The magnitude of cracking is highly influenced by the environment (Yamada et al., 1988), while the degree of cracking fluctuates from year to year because of changes in environmental conditions (Yamada et al., 1987).
The time of application of plant growth regulators such as gibberellic acid (GA3) influence cracking. For example, application of GA3 at flowering increases cracking in citrus fruits, but decreases cracking if applied shortly after flowering (Agusti et al., 2002). GA3 also influences fruit cracking when used in conjunction with other growth regulators such as auxin and cytokinin. For example, spraying a mixture of GA3 and 2,4-D reduces cracking in citrus, as does a combination of GA3 and naphthaleneacetic acid (NAA) (Monselise et al., 1985; Almela et al., 1994). In ‘pink Lady’ apples, application of cytokinin 6-benzyladenine in combination with gibberellins A4+A7 reduces calyx-end cracking (Stern et al., 2013; Ginzberg et al., 2014). Spraying with other chemicals including boron, aluminum, and calcium nitrate also decreases fruit cracking (powers and Bollen, 1947; Bullock, 1952; Glenn and poovaiah, 1989) in many commercially important fruits (e.g., cherry, apple, citrus, plum, apricot, banana, avocado, and pomegranate).
In general, ‘Fuyu’ persimmons with large calyxes do not exhibit calyx-end fruit cracking. previous reports have indicated that spraying with CppU (N-(2-chloro-4-pyridyl)-N′-phenylurea), a synthetic cytokinin, increases the calyx size of the ‘Taishu’ persimmon (Niikawa and Suzuki, 2013), while urea is most commonly used as a fertilizer to increase fruit size. Most recently it has also been observed that urea application effectively maintained the year-round production of pineapple by interrupting natural flowering during winter seasons (Lin et al., 2014). Therefore, we evaluated the physiological, phytohormonal, and anatomical characteristics of persimmons sprayed with CppU or urea.
Materials and Methods
Experimental Orchards and Application of CppU and Urea
Experiments were conducted in 2015 and 2016 on 25-year-old ‘Fuyu’ persimmon trees (Diospyros kaki) in a persimmon orchard in Jinju, Republic of Korea. Trees were grown normally using traditional cultural techniques in Korea (i.e., trees were annually pruned in winter, irrigated regularly in summer, and fertilized). Ten biological replicates of persimmon trees were treated with 10 mg∙L−1 N-(2-chloro-4-pyridyl)-N′-phenylurea (CppU) before flowering, and 1% urea before and after the flowering stages. Controls were not treated. CppU (Fulmet; Kyowa Hakko Kogyo Co., Ltd., Tokyo, Japan) and urea (Dongbu Hannong Chemical Co., Daejeon, Korea) treatments were prepared in aqueous solutions and applied directly to shoots using a handheld sprayer. persimmon calyxes were collected and transported in a low-vent plastic container (HpL826M, Lock&Lock, China) to the Department of Horticulture at Gyeongsang National University for further experimental analysis.
physiological Measurements
The number of persimmon fruits exhibiting calyx-end fruit cracking was counted, and fruit weights were measured using a weighing balance (MW-1, CAS, Korea). The length, width, and height of fruits and calyxes were measured using Vernier callipers (Mitutoyo CD-20Cp, 0.01 mm; Kawasaki, Kanagawa prefecture, Japan) and recorded. The long length of calyx was measured from a horizontal position right from top to bottom of the calyx i.e, horizontally from upper tip to lower tip of calyx. The short length was measured vertically from right side to left side of calyx tip. Similarly the width of calyx was measured at lower part (at maximum area of calyx diameter) of the calyx. The number of seeds per fruit was manually counted and recorded.
Fruit firmness was measured using a 6-mm diameter probe along a horizontal axis with a rheometer (RHEO TEX SD-700, Sun Scientific Co., Tokyo, Japan) (3-mm depth). Soluble solids content was detected by wrapping fruits in four layers of cheesecloth to strain the juices, and measuring the absorbance of the juice using a portable refractometer (pocket Refractometer, pAL-1; Atago Co., Ltd., Tokyo, Japan).
phytohormonal Analysis
Hormonal analysis was performed during the 2016 season only according to previously published methods (pan et al., 2010). Briefly, approximately 50 mg of ground calyxes from ‘Fuyu’ persimmons was used to measure hormone levels. To separate individual hormones in a mixture, each analyte and internal standard was subjected to high-performance liquid chromatography (HpLC) on a reversed-phase C18 column (Agilent, Santa Clara, CA, USA), followed by electrospray ionization tandem mass spectrometry (ESI-MS/MS; Applied Biosystems, Thermo Fisher Scientific, Foster City, CA, USA). Elutes were monitored by multiple-reaction monitoring (MRM). Stock solutions of IAA (indole-3-acetic acid) (Sigma–Aldrich, St. Louis, MO, USA), GA3 (Sigma–Aldrich), jasmonic acid (JA) (Sigma–Aldrich), zeatin (Sigma–Aldrich), d6-ABA (2-cis, 4-trans-abscisic acid-[2H6]) (OlChemIm Ltd., Olomouc, Czech Republic), and SA (salicylic acid) (TOKYO Chemical Industry Co., Japan) were prepared at concentrations of 1 mg·mL-1 in 100% methanol. HpLC was performed using a 1100-series liquid chromatography system (Agilent) equipped with a degasser, pump, auto sampler, and column oven. Chromatographic separations were performed on a SunfireTM C18 (2.1 × 10 mm) column (Waters, Milford, MA, USA) using an isocratic mobile phase of 15:85 (v/v) 0.1% formic acid in water/0.1% formic acid in methanol, at a flow rate of 300 μL·min-1. The column temperature was 30°C, and the injection volume was 10 μL in all experiments.
Anatomical Analysis
Anatomical observations were performed according to previously described methods (Lee et al., 2010). Tissue samples were obtained via horizontal sections along the equatorial axis of the entire fruit. A dual fixation was performed in 2.5% glutaraldehyde and 1% osmic acid solution, and then rinsed five times at 15~20 min intervals, respectively. Fixed samples were dehydrated in a stepwise ethanol series and vitrified with propylene oxide. Individual samples were then embedded in epoxy resin in silicon molds at 60°C for 4 days. Completed samples were sliced at a thickness of 1.5 μm using an ultra-microtome (Ultracut R, Leica Co., Vienna, Austria), stained with Schiff’s reagent according to standard methods, and observed under an optical microscope (Axio scope A1, Carl Zeiss, Gottingen, Germany). Measurements of the number of cell layers and cell dimensions were digitized using image analysis software (i-Solution; IMT, Vancouver, Canada). Thirty representative replicates were selected at random and examined to count cell layers and measure cell dimensions.
Statistical Analyses
All fruits were harvested in a completely randomized design and analyses were performed on four biological replicates. Data were analyzed with SAS statistical software, release 8.2 (SAS Institute, Cary, NC, USA) following analysis of variance (ANOVA) and Tukey’s T-test. Conclusions are predicated on differences between the means, with a significance level set at p < 0.05.
Results and Discussion
physiological Changes of persimmon Fruits in Response to CppU and Urea
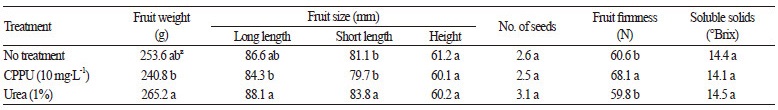
In the present study, we observed the basic physiology of persimmon fruits treated with 10 mg∙L-1 of CppU or 1% urea. The proportion of calyx cracking in CppU-treated and urea-treated fruits was significantly lower (37.1% and 30.4%, respectively) than in untreated persimmons (51.7%) (Table 1). Compared with untreated fruits, the weight and length of the long or short side of the harvested fruits were highest in urea-treated persimmons, whereas fruit firmness was significantly higher in CppU-treated persimmons (Table 2). Therefore, we conclude that spraying with CppU or urea is favorable for both increasing fruit growth and reducing calyx-end fruit cra cking in ‘Fuyu’ persimmons.
Table 1. Effect of spraying 10 mg∙L-1 N-(2-chloro-4-pyridyl)-N′-phenylurea (CPPU) or 1% urea on the proportion of ‘Fuyu’ persimmon fruits exhibiting calyx-end cracking
 |
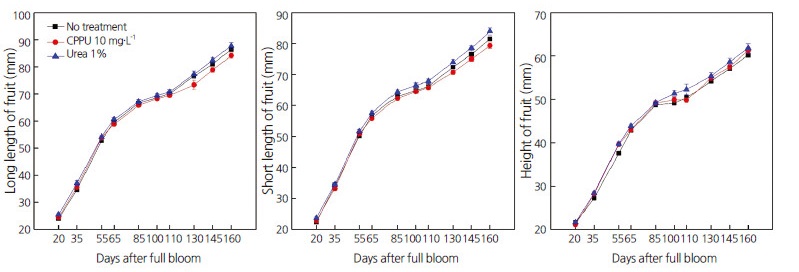
Compared with untreated persimmon fruits, application of CppU to persimmon fruits significantly increased the long and short lengths of calyxes, calyx widths, and the width of fruit stalks 35 d after full bloom (DAFB) (Fig. 1). The same parameters were slightly increased in 1% urea-treated fruits, compared with controls. After reaching the maximum at approximately 55 DAFB, these parameters remained constant from 65–160 DAFB, and were increased compared with controls. The long length, short length, and height of the fruits had similar increasing patterns in both treatment and control groups during the entire observation period (160 DAFB) (Fig. 2).
These results indicate that decreased cracking causes increased calyx dimensions, as observed in persimmons treated with CppU and urea. These findings agree with those of several reports addressing the role of growth regulators, hormones, and other chemicals in the prevention of fruit cracking in a variety of fruits (Usenik et al., 2005; Simon, 2006; Rehman et al., 2015; Khadivi-Khub, 2015). However, there are some fruits for which limited information is available regarding reducing calyx-end fruit cracking (e.g., apples) (Stern et al., 2003). We therefore suggest that CppU and urea may be used to reduce calyx-end cracking in fruit, and particularly in persimmons, which are more commercially important in Japan and Korea than in other Asian and western countries.
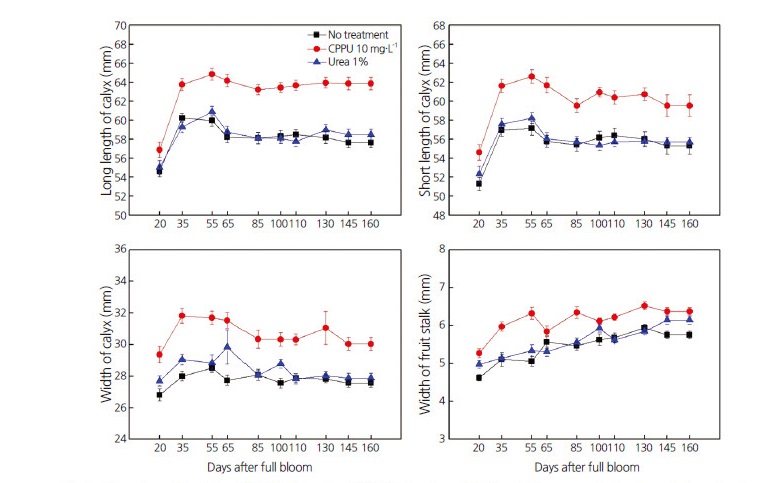
Fig. 1.
Effect of spraying 10 mg·L-1 N-(2-chloro-4-pyridyl)-N′-phenylurea (CPPU) or 1% urea on the long length, short length, and width of calyxes, and width of fruit stalks in ‘Fuyu’ persimmon fruits at different phenological stages (20–160 d after full bloom). Lines indicate means ± standard error (n=30).
Effects of CppU and Urea on phytohormones
Using HpLC, we studied the levels of six key phytohormones‒ IAA, GA3, zeatin, ABA, JA, and SA‒ in the calyxes of persimmon fruits treated with CppU or urea (Fig. 3). Compared with untreated fruits, different patterns of different phytohormone contents were seen during calyx development, and in response to both treatments, over time.
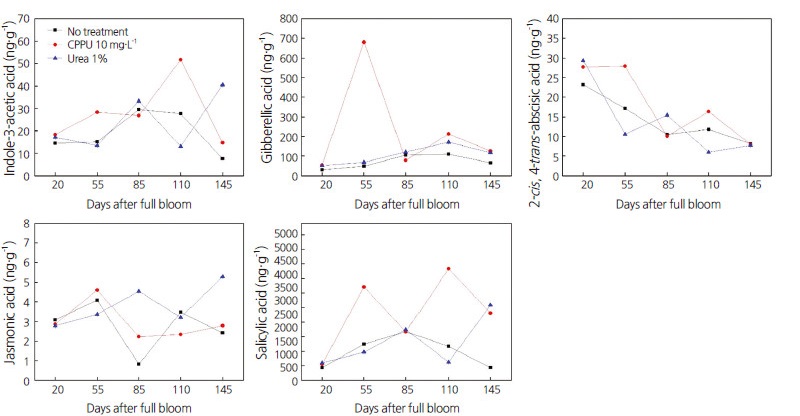
Fig. 3.
Effect of spraying 10 mg·L-1 N-(2-chloro-4-pyridyl)-N′-phenylurea (CPPU) or 1% urea on phytohormones (indole-3-acetic acid, gibberellic acid, 2-cis-trans-absessic acid, jasmonic acid, and salicylic acid) in ‘Fuyu’ persimmon calyxes at different phenological stages (20–160 d after full bloom).
The growth and development of a plant in response to any external cue is tightly regulated by phytohormones (Wolters and Jurgens, 2009; Leng et al., 2014; Wani et al., 2016). phytohormones are important in the fruit ripening process (Li et al., 2011); however, the role of phytohormones in fruit development during fruit cracking remains unknown. Although the effect of growth hormones such as GA, 2,4-D, NAA, and cytokinin on fruit cracking has been well documented (Sahu et al., 2013; Stern et al., 2003; 2013; Ginzberg et al., 2014), this is the first report to characterize phytohormone levels in persimmon calyx. Thus, we attempted to elucidate the mechanisms of the phytohormone signaling cascade during calyx development in persimmon fruits treated with or without CppU or urea. We did not detect zeatin in this experiment, thus we believe another cytokinin in involved in development of the persimmon fruit calyx.
IAA is a multifunctional phytohormone that is not only vital for normal plant growth and development, but it also governs plant growth under conditions of stress (Egamberdieva, 2009; Jung et al., 2015). IAA stimulates many genes, which have been identified in many plants. In the present study, levels of IAA increased significantly in fruit treated with CppU or urea compared with untreated persimmons. This indicates that CppU and urea promote IAA production, thus IAA might be integral to increasing calyx size in persimmons, just as it is integral to the adaptation of plants to abiotic stresses.
GAs are a large group of tetracyclic ditriterpenoid carboxylic acids; however, only a few function as growth hormones in higher plants (predominantly forms of GA1 and GA4) (Sponsel and Hedden, 2004). GAs have positive effects on seed germination, leaf expansion, flower development, and fruit development (Yamaguchi, 2008). We observed that GA levels were highest in persimmon calyx treated with CppU 55 DAFB. Levels of GA were also higher in CppU-treated and urea-treated persimmon calyx compared with control calyx. Our results indicate that CppU and urea increase the dimensions of the calyx in persimmon fruits, at least in part, by increasing levels of GAs, the main growth hormones involved in fruit development. previous studies have observed that GA4 and GA7 reduce calyx-end cracking in apples when combined with cytokinin 6-benzyladenine (Ginzberg et al., 2014).
ABA is also thought to be involved in the control of ripening, as a sharp increase in ABA accumulation has been reported during the onset of fruit ripening (Leng et al., 2014). ABA also accumulates before ethylene is released in climacteric fruits (Zhang et al., 1996; 2009a,b). Application of exogenous ABA enhances the production of several metabolites involved in fruit ripening, thereby promoting fruit ripening (Chernys and Zeevaart, 2000; Ban et al., 2003; Cakir et al., 2003; Jeong et al., 2004; Giribaldi et al., 2010). Therefore, we determined whether ABA was affected by the application of CppU and urea in persimmon fruits. Our results show wide variation in ABA levels of persimmon calyx treated with CppU or urea over time. Compared with untreated fruits, we observed that CppU increased the concentration of ABA in persimmon calyx, particularly after 55 DAFB (maximum level).
JAs are regulatory hormones derived from the metabolism of fatty acids (methyl jasmonate and free jasmonic acid). JAs are involved in reproductive processes, flowering, fruiting, senescence, secondary metabolism, and defense mechanisms (Seo and Koshiba, 2011; Jia et al., 2016). In the present study, JA levels in persimmon calyx treated with urea gradually increased at 85 DAFB and reached maximum levels at 145 DAFB.
SA is a naturally occurring phenolic compound involved in the regulation of pathogenesis-associated protein expression (Miura and Tada, 2014). Besides its role in defense, SA is essential for plant growth, fruit ripening, and fruit development. In the present study, maximum SA levels were observed in persimmon fruits at 55 DAFB and 110 DAFB in fruits treated with CppU, and at 145 DAFB in fruits treated with urea. Thus, aside from the higher levels observed in CppU-treated fruits, there was no clear pattern in SA levels between treatments.
The hormonal control of fruit ripening is complex and involves several growth regulators. Specific ripening processes, such as fruit coloration, softening, sugar accumulation, aroma development and reduction in fruit cracking are under the control of specific hormones (Fortes et al., 2015). The phytohormone analysis of persimmon calyxes undertaken in the present study represents a new platform for calyx-end fruit cracking attenuation by increasing the calyx size of persimmon fruits via the application of CppU or urea. In conclusion, we surmise that the application of CppU and urea to persimmon fruits is beneficial, not only for increasing levels of the phytohormones responsible for fruit development and ripening, but also for attenuating ripening and development to some extent.
Anatomical Changes in persimmon Fruits in Response to CppU and Urea
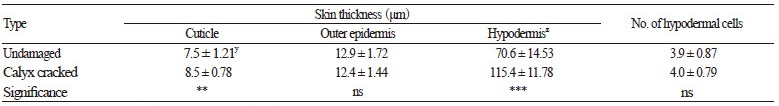
We used microscopy to analyze the anatomical differences in the skin layer of persimmon fruits. Skins of untreated ‘Fuyu’ persimmon fruits (control) comprised one layer of outer epidermis and 1–6 layers of hypodermis with or without tannins. The presence of parenchymal tissue from the outer to inner sections was common (Fig. 4). Stone cells were present between the hypodermis and parenchymal tissues. The thickness of the primary and secondary cuticle layers was 7–8 μm, and was a similar thickness to that of the outer epidermis. The cuticle also began filling the intercellular spaces appearing in the outer epidermis (Fig. 4A).
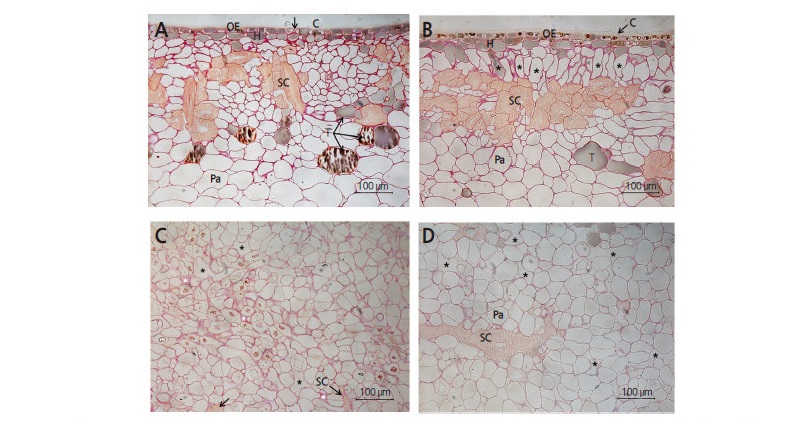
Fig. 4.
Cross-sectional characterization of ‘Fuyu’ persimmon fruit skin. (A) Undamaged fruit (no calyx-end cracking). Note the space between the outer epidermal cells indicated by black arrows. (B) Calyx-end cracked fruit with elongated hypodermal and parenchymal cells (*) directly above stone cells. (C) Abscission zone under the calyx of undamaged fruit showing the accumulation of pectin (white *) in the intercellular spaces (black *). (D) Abscission zone of calyx-end cracked fruit. Note the appearance of very large stone cells and intercellular spaces (*). C, cuticle; H, hypodermis; OE, outer epidermis; Pa, parenchyma; SC, stone cell (sclereid); T, tannin cell. Scale bars = 100 μm.
In contrast, the cuticle and fruit skin thicknesses of fruits with calyx-end fruit cracking were 8.5 and 115.4 μm, respectively; considerably thicker than undamaged fruits with no calyx-end cracking (Table 3). Microscopic analysis confirmed that the thicker skin of fruits with calyx-end cracking was caused by elongation of parenchymal cells (Fig. 4). In persimmon fruits, stone cells have previously only been observed under hypodermal cells (park et al., 2003); however, in the present study, they were also observed under the calyx. presumably, the lower part of the calyx derives from the hypodermal layer of young fruits and develops into a tissue that includes stone cells. The cells and intercellular spaces of undamaged fruits were also smaller than in fruits with calyx-end cracking, in which the intercellular spaces were filled with polysaccharides, particularly pectin (Fig. 4C).
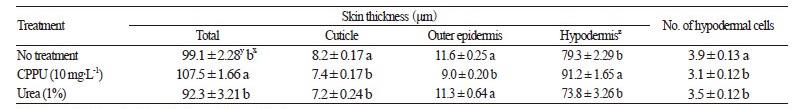
The application of foliar urea and CppU are two of several treatments that appear to inhibit the development of calyx-end cracking, as indicated by our phytohormone observations. The thickness of the skin, including the cuticle, outer epidermis, and hypodermal cell layers of urea-treated fruits was 107.5 μm, which was approximately 8–10 μm thicker than control or CppU-treated fruits (Table 4 and Fig. 5). Foliar urea treatments increase the nitrogen concentration of trees (Cheng and Fuchigami, 2003). In the present study, foliar urea treatments indirectly expanded hypodermal cells (Fig. 5B). Tannins in the hypodermal cells in CppU-treated and urea-treated fruits were present in the form of small grains (Fig. 5A and B); however, tannin cells in untreated fruits were filled with vacuoles (Fig. 5C). These results indicate that application of urea and CppU attenuates the negative effects of cracking in the Asian persimmon; however, the effects of urea and CppU on the development of fruit tissues require future research.
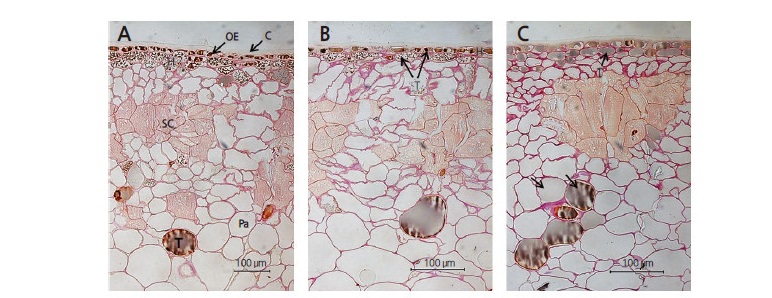
Fig. 5.
Cross-sectional characterization of ‘Fuyu’ persimmon fruit skin in response to foliar applications of CPPU or urea. (A) CPPU treatment; (B) urea treatment; and (C) control (untreated) ‘Fuyu’ persimmon fruits. C, cuticle; H, hypodermis; OE, outer epidermis; Pa, parenchyma; SC, stone cell (sclereid); T, tannin cell. Scale bars = 100 μm.
In conclusion, our study demonstrates that foliar CppU and urea treatments are efficient methods for reducing calyx-end cracking of ‘Fuyu’ persimmon fruits.