Introduction
Material and Methods
Plant Materials
Measurement of Ethylene Production
RNA and DNA Extraction and cDNA Synthesis
Internal Conserved Fragment Cloning of Two Ethylene Biosynthetic Genes
5’- and 3’-RACE (the Rapid Amplification of cDNA Ends) of HvACS and HvACO
Cloning of the Full-length cDNAs and DNAs of HvACS and HvACO
Sequence Analysis
Semi-quantitative RT-PCR Assay
Results
Ethylene Production by Whole Flowers and Wounded Leaves
Isolation and Characterization of HvACS and HvACO from H. ventricosa Flowers
Expression of Two Ethylene Biosynthetic Genes during Flower Development and in Wounded Leaves
Discussion
Introduction
Hosta Tratt. is an herbaceous perennial with about 45 species distributed mainly in Japan, as well as a few in China, Korea and Russia (Rao et al., 2012). Hosta ventricosa is the only natural tetraploid species in the genus Hosta (Cao et al., 2011). This species has been widely used in city afforestation, garden landscape and home decoration as a globally important ornamental plant for both beautiful foliage and flowers (Shadrack and Shadrack, 2011). The main reason for its wide cultivation in China is not only for its ornamental value, but also for its consideration as a medicinal herb and its economical value as food (Cao et al., 2011). H. ventricosa is generally easy and long-lived garden plant, relatively disease free, requiring little care other than watering and some fertilizer to enhance growth (http://belflora.com/en/catalog/hosta/). This species is adichogamous and self-compatible, but pollinator visitation is necessary for pollination. Flowers are produced on a raceme inflorescence and open sequentially from basal to distal positions (acropetal blooming). Commonly, only one or two flowers open on the same day within an inflorescence. However, longevity of individual flowers is only one day (Cao et al., 2011).
Ethylene, an internal signal molecule, plays a pivotal role in flower development, senescence and abscission. Disruption of ethylene biosynthesis in carnation and petunia resulted in delayed flower death (Rogers, 2006). In higher plants, ethylene is made via a two-step biosynthetic route that starts with the conversion of the methionine derivative S-adenosyl-L-methionine to 1-aminocyclopropane-1-carboxylic acid (ACC) by ACC synthase (ACS, EC 4.4.1.14). Then ACC is oxidized by ACC oxidase (ACO) to release ethylene (Lin et al., 2009). The activity of ACS is considered to be the rate-limiting step in the pathway (Wang et al., 2007), and the ACO activity contributes to the regulation of ethylene production (Lasserre et al., 1996; Xue et al., 2008). Both enzymes are encoded by multigene families, the members of which are differentially regulated in a tissue-specific manner by a variety of signals including wounding and senescence (Jones and Woodson, 1999; Lasserre et al., 1996; Lin et al., 2009; Trusov and Botella, 2006; Xue et al., 2008). Although there is evidence for the regulation of ACS and ACO at the posttranscriptional level, expression analyses indicate that the induction of their activity is most often the result of the increased accumulation of the mRNAs (Jones and Woodson, 1999; Weterings et al., 2002). The sensitivity of flower senescence to endogenously produced ethylene is species specific (Rogers, 2006). In ethylene- sensitive species, flower senescence is regulated through the expression of genes encoding ACS and ACO enzymes (ten Have and Woltering, 1997). Their transcript levels are up-regulated as petals senesce (Jones, 2003; Weterings et al., 2002).
The flowers of H. ventricosa produce nectar and have six tepals, six stamens of similar size, and a gynoecium consisting of three carpels. They produce seed by pseudo-gamous apomixis (Cao et al., 2011). The role of ethylene in its flower senescence is unclear. There has been no attempt to determine if H. ventricosa flowers produce ethylene, nor have the related genes been cloned. An under-standing of the role of ethylene in the control of floral deterioration in H. ventricosa has commercial relevance for post-harvest handling, as well as furthering understanding of the regulation of floral senescence. Accordingly, our main objectives in this study were to (i) evaluation on ethylene production from whole flowers and wounded leaves of H. ventricosa; (ii) cloning of two genes encoding the ethylene biosynthetic enzymes ACS and ACO from blooming flowers and analysis of gene sequences; and (iii) comparison of their expression during flower development with that in wounded leaves.
Materials and Methods
Plant Materials
Hosta ventricosa (Salisb.) Stearn (Hostaceae) was planted in soil and grown under natural conditions at the experimental farm at Zhejiang Normal University, China. Flowers were sampled at four flowering stages (Fig. 1): A, small bud. Petaloid tepals started to emerge from bracts; B, mature bud. Petaloid tepals reached their full length and were ready to open; C, blooming. Flower fully opened. Six stamens were visible. The anthers were all undehisced and positioned below the stigma; and D, wilting. Petaloid tepals were wilted. The flower appearance had deteriorated and all the anthers were dehisced. Stem, young leaf and root samples of H. ventricosa were randomly collected periodically. For gene expression analysis in wounded leaves, mature leaves were cut across the midrib with a sterile razor blade for 2 h, 6 h, 12 h and 24 h. All samples were frozen immediately in liquid nitrogen and stored at -80°C.
|
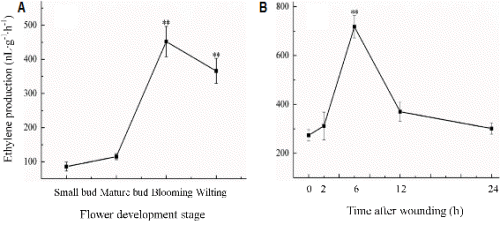
Fig. 1. Stages of H. ventricosa flower development. A, small bud; B, mature bud; C, blooming; D, wilting. |
Measurement of Ethylene Production
Ethylene production was measured on three replicates each of four detached whole flowers (eight for blooming flowers) or wounding leaves at the given sampling times. The samples were detached and enclosed in 50 or 280 mL (for blooming flowers) airtight containers and incubated at 25°C for 2 h following which 1 mL of the headspace was withdrawn. Ethylene concentration in the gas sample was analyzed by gas chromatography (GC; model SP 6800, Lunan Chemical Engineering Instrument, Shandong, China) equipped with a GDX-502 column and a flame-ionization detector for ethylene determination. The injector, detector, and oven/column temperatures were 110°C, 140°C, and 90°C, respectively.
RNA and DNA Extraction and cDNA Synthesis
Total RNA was extracted from whole flower with improved hot borate method (Hunter and Reid, 2001), while that was isolated from root, stem and leaf using Trizol Reagent (Life Technologies, Carlsbad, CA, USA). Genomic DNA was extracted by cetyltriethylammnonium bromide method. The quality and concentration of RNA and DNA were checked by agarose gel electrophoresis and evaluated by a NanoDrop 2000 Spectrophotometer (Thermo Scientific, San Jose, CA, USA). First-strand cDNA was synthesized using an oligo (dT)-18 primer and Superscript II kit (Invitrogen, Carlsbad, CA, USA) according to manufacturer’s instructions.
Internal Conserved Fragment Cloning of Two Ethylene Biosynthetic Genes
The primers were designed based on the conserved sequence of each gene from the National Center for Bio-technology Information (NCBI) database to amplify two gene fragments and were synthesized by SBS Genetech Co., Ltd. (Shanghai, China). The degenerated primers TA(T/C) CC(G/A/T)GGATT(T/C)GA(T/C)AGAGA and AACCA(A/T/G/C) CC(A/C/T)GG(T/C)TC(G/C/A)(A/G)(A/C/T)GCA were used for cloning of HvACS partial cDNA, and the specific primers 5’-GGGGCTTCTTTGAGATTCTGAACC-3’ and 5’-TCATGGTCT CAAATCTTGGCTCC-3’ for HvACO. 1 μL of the diluted cDNA from blooming flowers was amplified in 20 μL reaction mixtures containing 2 μL 10×PCR buffer with MgCl2, 2 μL 2.5 mM dNTPs, 0.2 μL rTaq polymerase (Takara, Dalian, China), 0.5 μL 10 μM each primer and 13.8 μL distilled H2O. The PCR was conducted for 30 cycles each consisting of 30 s at 95°C, 30 s at 52°C or 55°C and 1 min at 72°C. Purified amplified products of appropriate length were ligated to the pMD18-T vector for sequencing (Sangon, Shanghai, China).
5’- and 3’-RACE (the Rapid Amplification of cDNA Ends) of HvACS and HvACO
The 5’- and 3’-RACE cDNAs of two genes were isolated according to the SMART RACE cDNA Amplification Kit protocol (Clontech, Mountain View, CA, USA). For HvACS we used the specific primer 5'-GAACTCCTCATCAGACAACA TTGAGGC-3’ and UPM for the first round of 5'-RACE, and 5'-CACCGAGGCTTCAGCCGCAAAGATCAC-3’ and UPM for 3'- RACE both at the annealing temperature of 58.5°C. The PCR product was diluted 40-fold as a template for the second round. The nested primer (5'-CGATCACGAAGTCGA GTGCATCTTCGAGG-3’) and NUP (provided in the kit) were used for 5'-RACE at the annealing temperature of 59.2°C, 5'-CTCGGGGGTACGACAACGCAGAGAGAGTCC-3’ and NUP for 3'-RACE at the annealing temperature of 61.0°C. The 20 μL reaction mixture was subjected to an initial denaturation at 95°C for 5 min, and subsequently 30 cycles of amplification (95°C for 30 s, annealing for 30 s and 72°C for 1 min).
For HvACO, the first round of amplification was performed using the specific primer 5'-GGACTCGGGGAGATGGCGCAAG AAG-3’ and UPM for 5'-RACE at the annealing temperature of 58.0°C, and 5'-GGGGCTTCTTCGAGATGGTGAACCACGGAA-3’ and UPM for 3'-RACE at the annealing temperature of 56.5°C. The nested primer (5'-TTGCTCCATGCCCTTCTTGTA GTGCCCC-3’) and NUP (provided in the kit) were used for 5'-RACE at the annealing temperature of 57.2°C, 5'-CTTCTT GCGCCATCTCCCCGAGTCCAACAT-3’ and NUP for 3'-RACE at the annealing temperature of 58.0°C. All RACE products were ligated to the pMD18-T vector for sequencing.
Cloning of the Full-length cDNAs and DNAs of HvACS and HvACO
After alignment and assembly of the sequences of the internal conserved sequence, the 5’-RACE and 3’-RACE products, the full-length cDNA seqences of two genes were deduced and subsequently obtained by RT-PCR in a total volume of 20 μL. The primers 5’-GGCCTTCTCTTAATATTTC ATTAGTTGTG-3’ and 5’-AACAAGACTGGATGCATAATTACG ATCC-3’ were used for HvACS, 5’-ACGCGGGGACAACACAAG CACTAA-3’ and 5’-AGTAGCTCATGATCAACAACACTTA-3’ for HvACO. The PCR annealing temperature was 60.5°C for HvACS and 54.6°C for HvACO. And the extension time was 2 min instead of 1 min.
The full-length DNAs of two genes were amplified using genomic DNA from leaves as templates and the same primers as above for HvACS, and 5’-GCCATGGACGTTCTTC GTGATGCTTGCGAG-3’ and 5’-CACTTATGAGTCTCTCTCTC TCTCATGCAG-3’ for HvACO at the annealing temperature of 61.5°C.
Sequence Analysis
Nucleotide sequence and the deduced amino acid sequence were identified by the NCBI BLAST program (http://www. ncbi.nlm.nih.gov/BLAST/). Prediction of ORF was made using the NCBI ORF Finder program. The full-length cDNA and DNA sequences were assembled with ContigExpress software (Invitrogen, Carlsbad, CA, USA). Gene structure was analyzed using the GSD Server (http://gsds.cbi.pku. edu.cn/index.php). Amino acid multiple alignments were made with the Clustal X 2.0 program under default para-meters. Theoretical molecular weight of deduced polypeptide was calculated using the Compute Mw tool (http://www. expasy.ch/tools/pi_tool.html). Predictions of subcellular localization of the deduced polypeptides were conducted by using PSORT (http://wolfpsort.org) and TargetP (http:// www.cbs.dtu.dk/services/TargetP/).
Semi-quantitative RT-PCR Assay
Semi-quantitative RT-PCRs were performed using the primers 5’-CACCGAGGCTTCAGCCGCAAAGATCAC-3’ and 5’- GAACTCCTCATCAGACAACATTGAGGC-3’ for HvACS at the annealing temperature of 59.5°C, and 5’-TGAACCACGGAAT CTCGC-3’ and 5’-AGCCATTGAAGGCACTCT-3’ for HvACO at the annealing temperature of 55.0°C. HvActin (Accession No. JX274300) mRNA was tested for control purposes using the primers 5’-GAACTGGTATGGTCAAGGCTG-3’ and 5’-ACACGGAGCTCGTTGTAGAAG-3’ at the annealing tem-pera-ture of 52.0°C. PCR amplification was performed in duplicates in three independent assays for 30 cycles with denaturation at 95°C for 30 s, annealing for 30 s and elongation at 72°C for 30 s, followed by 72°C for 10 min. The relative expression was given by a ratio between the target gene and the reference gene.
Results
|
Fig. 2. Ethylene production from H. ventricosa flowers (A) and wounded leaves (B). Error bars indicate SE from three replicates. |
Ethylene Production by Whole Flowers and Wounded Leaves
In small buds of H. ventricosa, the level of ethylene pro-duction was approximately 85 nL・g-1・h-1, which is much higher than other ethylene-sensitive species such as carnation (ten Have and Woltering, 1997) and Alstroemeria (Wagstaff et al., 2005). Ethylene concentration increased significantly (p < 0.01) at blooming stage to a maximum over 450 nL・g-1・h-1 and thereafter declining slightly to 365 nL・g-1・h-1 at wilting stage (Fig. 2A). The increase in ethylene production occurred before symptoms of senescence were apparent.
As shown in Fig. 2B, the ethylene production was signi-fi-cantly (p < 0.01) enhanced at 6 h after mechanical wounding in mature leave, and seemed to decline at a similar rate to that at which it increased.
Isolation and Characterization of HvACS and HvACO from H. ventricosa Flowers
On the basis of the conserved amino acid sequences of two genes from different plant species, two pairs of primers were designed. Two cDNA fragments of 777 bp and 822 bp were obtained by PCR using cDNA aliquots of blooming flowers of H. ventricosa. To clone the full-length cDNA, 3’- and 5’-RACE reactions were conducted. The 5’-RACE-PCRs generated a fragment of 771 bp for the HvACS and 274 bp for the HvACO gene. 3’-RACE reactions generated fragments of 702 bp and 1071 bp for HvACS and HvACO, respectively. On the basis of the above sequences, specific primers were designed for the 3’- and 5’-untranslated regions of two genes, and the full-length cDNA and genomic DNA were amplified.
The cloned HvACS was 1562 bp in length and contained a 1347 bp ORF encoding a protein of 448 amino acids. The calculated molecular mass of the protein was 49.9 kDa with an isoelectric point of 5.81 (Table 1). The putative protein had a high identity with other known from Musa acuminata (CAA11158.1) (79%). Seven highly conserved regions among all ACS were detected in HvACS (Fig. 3). The Lys276 present in the most heavily conserved region serves as the pyridoxal 5’-phosphate (PLP)-binding site by reducing the double bond between PLP and the enzyme itself in tomato (Lin et al., 2009). Eleven invariant residues that were conserved in all ACS and certain aminotransferase (Wang et al., 2007) also present in HvACS. Four important amino acid residues (Tyr240, Lys276, Arg284 and Arg410) were detected here (Fig. 3), which were involved in interac-tions with the PLP cofactor in apple and tomato (Wang et al., 2007). The C-terminal end, the least conserved region, may correspond to the different protein dimerization and stability of ACS (Wang et al., 2007). The genomic sequence of HvACS was 1915 bp and interrupted by two introns (257 bp and 643 bp respectively) located near the 5’-end of gene, similar to ACS genes from other higher plants (Wang et al., 2007).
The full-length HvACO harbored an ORF of 957 bp encoding a polypeptide of 318 amino acids with calculated molecular mass 36.1 kDa (Table 1). The HvACO amino acid sequence showed 83% identity with ACO of Narcissus tazetta (AEZ06406.1). Seven regions indicated in box (Fig. 4) were conserved in other ACO isoforms. The nine amino acid residues (Pro5, Ala27, His39, His177, Asp179, Gly218, His234, Arg244 and Ser246) (Fig. 4) conserved across all members of the Fe/ascorbate-dependent dioxygenase family (Lasserre et al., 1996) were found in HvACO. Three residues (His177, Asp179 and His234) were ligated to a single Fe(II) to form the active site. The side chains of Arg175 and Arg244 were involved in binding bicarbonate, which leads to the activation of the ACO enzyme (Zhang et al., 2004). HvACO is similar to the tomato, petunia and melon genes, with three introns located at the responding positions (Lasserre et al., 1996).
Expression of Two Ethylene Biosynthetic Genes during Flower Development and in Wounded Leaves
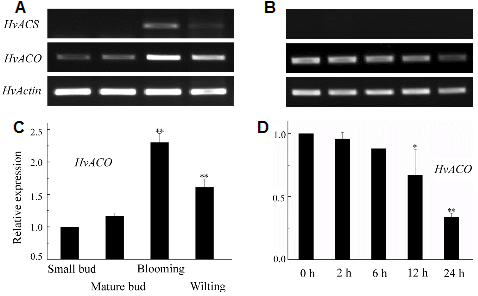
In flowers, the HvACS transcript was below the level of detection at small bud stage and at mature bud stage, strongly induced at blooming stage and remained at moderate level at wilting stage (Fig. 5A). The relative expression of HvACO significantly increased (p < 0.01) by over 2.2-fold at blooming stage and 1.5-fold at wilting stage compared to small bud stage (Fig. 5C). The maximum expression of HvACS and HvACO was observed at blooming stage which was accompanied by a peak of ethylene production (Fig. 2).
In wounded leaves, there were not much changes in HvACO expression within 6 h, but a significant decrease after 12 h of treatment (Fig. 5D). Interestingly, the HvACS transcript was not detected throughout the 24-h period after mechanical wounding (Fig. 5B).
Discussion
Flower deterioration of H. ventricosa is manifested as tepal wilting (Fig. 1). There was a significant increase in the rate of ethylene production from blooming and wilting flowers (Fig. 2). The expression analyses of two cloned genes involved in ethylene biosynthesis indicated that the timing of the induction of HvACS expression was correspond to the timing of the increase of ethylene production, and that the up-regulation of HvACO transcript was closely correlated with elevated ethylene production. In order to determine whether the accumulation of HvACS and HvACO mRNAs identified from whole flowers were flower-specific, the expression of two genes was also investigated in the wounded leaves that stimulated the generation of ethylene, and in different vegetative tissues. HvACS transcripts were detectable only in stems and roots (Fig. 6), and strongly induced in blooming and wilting flowers (Fig. 5A), whereas not detectable in young leaves and in wounded mature leaves (Fig. 5B). The results indicated that HvACS exclusively expressed during flower senescence. The transcripts of HvACO, which were present in flowers and young leaves and not in roots and stems (Figs. 5A and 6), displayed a significant up-regulation during the late stages of flower development from a basal level (Fig. 5C) but underwent a down-regulation at 12 h and 24 h after wounding in mature leaves (Fig. 5D). These data suggested that HvACS and HvACO here were specifically regulated by flower senescence although they are encoded by multigene family (Lin et al., 2009), and ethylene played an important role in flower senescence of H. ventricosa. There are likely additional members of HvACS and HvACO gene family responsible for the responses to wounding in mature leaves.
ACS and ACO have been reported in several species to be involved in flower development or senescence. In rose, the expression of the ACS gene increased in a short-lasting cultivar, but remained at a constant and low level in a long-lasting one during the petal senescence process of flower, while the expression of the ACO gene increased in the final stage of flower development in both cultivars (Mu¨ller et al., 2000). In tobacco, both ACS2 and ACO transcripts were not detected at early flower stage and the levels increased at later stages when ethylene release was induced by the pistil (Weterings et al., 2002). In Alstroemeria, ALACO1 showed very little change over the first six stages of development, but then increased by over 4.5-fold in the last two stages of development prior to tepal abscission (Wagstaff et al., 2005). In tomato, pollination caused an increase in ethylene synthesis, an enhanced rate of senescence, and was accompanied by changes in ACS and ACO expression (Llop-Tous et al., 2000). In carnation, the abundance of ACS and ACO mRNAs increased concomitantly with the ethylene climacteric in senescing petals (ten Have and Woltering, 1997; Woodson et al., 1992). Continuous monitoring of the flowering dynamics of transgenic and control pineapple plants showed that suppression of the ACS2 gene resulted in significantly delayed flowering (Trusov and Botella, 2006). Flowers of transgenic carnation containing antisense ACO gene exhibited low climacteric ethylene production and a markedly delayed petal senescence (Savin et al., 1995).
In carnation, ethylene produced from the pollinated stigma is translocated, via the style and ovary, to the petals. Here it up-regulates ethylene biosynthetic genes and induces the production of ethylene in the petals (ten Have and Woltering, 1997). Once initiated, the production of ethylene becomes autocatalytic (Wagstaff et al., 2005). In tomato, translocated ACC from the pistil may be important for regulating the initial burst of ethylene production during petal senescence (Llop-Tous et al., 2000). Further detailed study will be needed to more accurately identify the site of ethylene biosynthesis, and assess the regulatory mechanisms of ethylene biosynthesis in the senescing flower of H. ventricosa.