Introduction
Materials and Methods
Plant Material and Experimental Design
Growth Parameters
Measurement of Leaf Relative Water Content
Measurement of Lipid Peroxidation, H2O2
Electrolyte Leakage
Determination of the Enzyme Activities of SOD, CAT, POD, and APX
Staistical Analysis
Results
Growth Status
Effects of Salinity Stress on Leaf RWC
Lipid Peroxidation
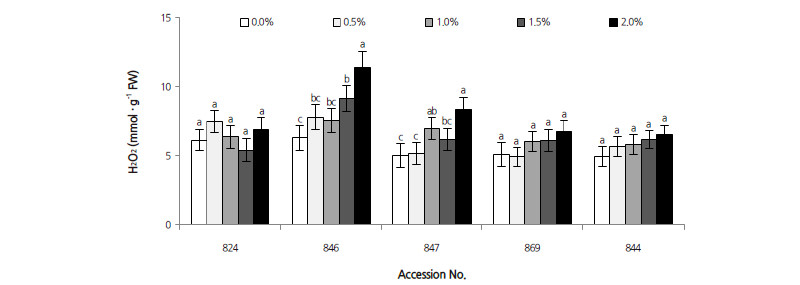
H2O2 Generation
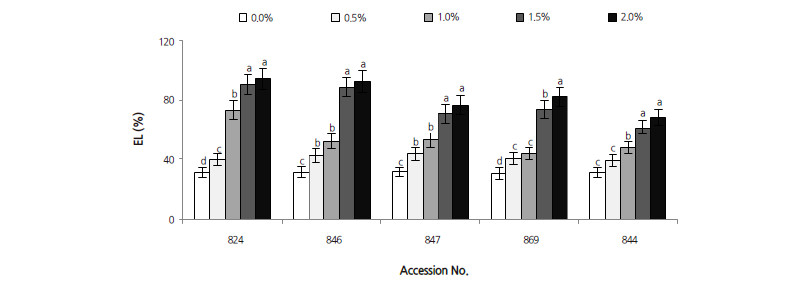
Electrolyte Leakage
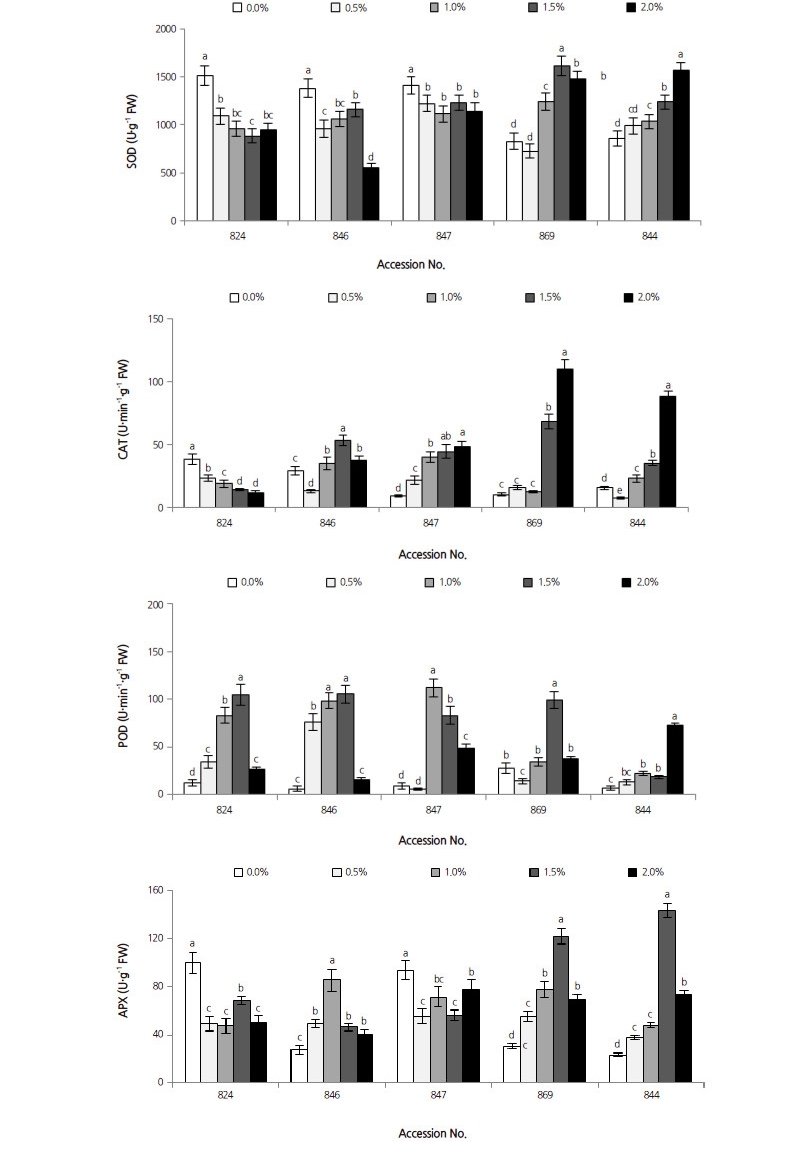
Antioxidant Enzymes
Discussion
Introduction
Salt stress is a major environmental limiting factor for agricultural production that determines the geographical distribution of plants in arid and semi-arid areas all over the world (Flowers, 2004). Salinity affects 19.5% of irrigated land and 2.1% of dry land agriculture according to recent statistics (FAO, 2000). In China, there are approximately 100 million hectares of saline soil, mainly in the northern, northeast, and northwest regions. Many researchers have reported that salinity stress induces the expression of specific genes and affects physiological and biochemical parameters in many plants, such as rice (Khan and Panda, 2008; Lee et al., 2013), alfalfa (Wang and Han, 2009), perennial ryegrass (Hu et al., 2012), moraceae (Zhang et al., 2013) and grapevine (Ikbal et al., 2014). However, the mechanisms of plant salt tolerance are not fully understood. Salt affects plant growth and crop yield. High salt concentrations can also cause cellular imbalance, leading to ionic and osmotic stress, and increased production of reactive oxygen species (ROS) (Cramer et al., 1994; Demiral and Türkan,2005; Mandhania et al., 2006). ROS are highly reactive and toxic, and lead to damage to proteins, membrane lipids, cellular structures, and other vital molecules, such as RNA and DNA (Parida and Das, 2005; Türkan and Demiral, 2009; Adly, 2010). Examples of ROS include hydrogen peroxide (H2O2), the radicals superoxide (O2·-), hydroxyl (OH·) and alkoxyl (RO·), and singlet oxygen (1O2) (Halliwell and Gutteridge, 1985; Foyer and Noctor, 2003).
Plants have developed resistance mechanisms such as osmotic adjustment and alterations of photosynthesis, and thus they are able to enhance their antioxidative capacity to prevent damage caused by salt stress (Gibson et al., 1984; Dionisio-Sese and Tobita, 1998). Analyses of these protective strategies provide us with a basic understanding of plants’ tolerance and resistance. Antioxidant defense machinery scavenges free radicals to improve plants’ salt tolerance (Shigeoka et al., 2002). ROS accumulation induced by various stresses is counteracted by antioxidant molecules, such as glutathione (GSH), ascorbic acid (AsA), alkaloids, α-tocopherol, and tocopherol, and by various enzymes including superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX), peroxidase (POD), glutathione reductase (GR), glutathione peroxidase (GPX) and guaiacol peroxidase (GOPX) (Noctor and Foyer, 1998; Hernandez et al., 2000; Prochazkova et al., 2001; Bor et al., 2003; Ashraf and Arkam, 2009). These enzymes and metabolites not only protect plants from cellular damage, but also fine-tune ROS concentrations to optimize their functions in metabolism (Becana et al., 2010). Since high ROS concentrations are harmful to plant cells, several studies have explored the role of ROS at a steady-state level as substrates and signals in plant cells (Sarvajeet and Tuteja, 2010; Foyer and Shigeoka, 2011), which contribute to the control of plant growth and development; as beneficial transduction signaling molecules during normal metabolism; and in response to different stresses (Nauseef, 2008).
Species of the same genus can show wide variation in their responses to salinity stress (Dionisio-Seseand Tobita, 1998; Bor et al., 2003). In the first study of its kind, we chose five accessions of persimmon rootstock to determine their responses to salinity, and examine the differences between them, ultimately aiming to improve the salt tolerance of Diospyros. Accessions selected were: D.lotus, which is frequently used as persimmon rootstock in the North of China because of its good tolerance to salinity; D. kaki var. sylvestris, which is generally used as persimmon rootstock in the South of China; and D. virginiana, originally from the Americas, used in Israel, and now as a rootstock for Japanese persimmon because it is particularly well adapted to poor soil (Tao and Sugiura, 1992). We investigated the effects of different soil NaCl concentrations on growth, H2O2 accumulation, lipid peroxidation, electrolyte leakage, and antioxidant enzyme activities of leaves from young seedlings of these persimmon rootstock accessions.
Materials and Methods
Plant Material and Experimental Design
Five accessions of Diospyros: three of D. lotus, and D. kaki var. sylverstris and D. virginiana were employed in this study (Table 1). Mature seeds were collected in December 2011 from the National Field Genebank for Persimmon of Northwest A&F University, located in Yangling (34′20″ N, 108′24″ E), Shaanxi Province, China. After washing with clean water, seeds were dried in a ventilator, placed in bags and stored in the dark. The surface of the seeds was sterilized with 0.58% sodium hypochloride solution and washed several times with deionized water. Seeds were then germinated in the dark on 90 mm glass Petri dishes in an artificial climate incubator with a constant or diurnally alternating temperature regime of 12 h 25°C/12 h 20°C for 7 d. Germinated seeds were then transferred to pots filled with aerated plant nutrient solution. Pots of each species were divided into four groups (four replicates each) and grown in a climate-controlled growth chamber, at a temperature of 25°C during the light day and 20°C during the night, relative humidity of 70%, and light intensity of 400 μmol·m-2·s-1. Seedlings were grown under non-saline conditions for 15 d to fully prepare them for salinity treatment. After 15 d, all plants were exposed to salinity by adding NaCl solution to the growth medium, adding 0.5% increments every 24 h to avoid osmotic shock, until the final NaCl concentrations reached 0.5, 1.0, 1.5, and 2.0% (0.0% for control). Nutrient solutions were refreshed every 5 days and deionized water was added daily to replace the water lost by transpiration. Salinity treatment lasted 25 d; plants were harvested on the 25th day after salt stress, leaves were collected and immediately frozen in liquid nitrogen, and stored at –70°C for subsequent analyses.
Growth Parameters
Relative plant height growth was measured using the following formula:
Rg(5) = (Ht1 - Ht0 ) / (Hc0 - Hc1 ) × 100
where: Rg, relative height growth (%); Ht0 and Ht1 , initial and final height of treatment seedlings, respectively; Hc0 and Hc1 , initial and final height of control seedlings, respectively.
Measurement of Leaf Relative Water Content
Leaf relative water content (RWC) was determined using the method by Barrs and Weatherley (1962) shown below:
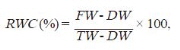
where: FW, leaf fresh weight; DW, leaf dry weight; TW, leaf turgid weight.
First, a fresh leaf was weighed then soaked in deionized water for 12 h. Turgid tissue was then quickly and carefully blotted dry before weighing again to determine its turgid weight. Dry weight was determined after drying the leaf in an oven at 80°C for 72 h until it reached a constant weight.
Measurement of Lipid Peroxidation, H2O2
The level of lipid peroxidation in the leaf tissue was detected in terms of malondialdehyde (MDA) content using the thiobarbituric acid reaction (Madhava Rao and Sresty, 2000), as described by Gao (2000). A leaf sample weighing 0.5 g was used. The absorbance value at 532 nm and the value for nonspecific absorption at 600 nm were detected to calculate the MDA content using its extinction coefficient of 155 mM-1·cm-1.
The H2O2 concentration was measured using the methods of Alexieva et al. (2001). A 0.5 g leaf sample was homogenized in 5 mL ice-cold 3% trichloroacetic acid followed and centrifuged at 12,000 ×g for 20 min at 4°C. A total of 1 mL supernatant was transferred to a new tube and mixed with 0.1 mL 10 mM phosphate-buffered saline (PBS), pH 7.0 and 2 mL 1 M potassium iodide. The absorbance of the solution was read at 410 nm.
Electrolyte Leakage
Leaf discs weighing 0.3 g were cut to a uniform size (5 mm diameter) and placed into test tubes containing 10 mL deionized water. The tubes were incubated in a water bath at the constant temperature of 32°C for 2 h and the initial electrical conductivity of the medium (EC1) was determined. Thereafter, the samples were autoclaved at 121°C for 20 min to release all electrolytes, cooled to 25°C, and the final electrical conductivity (EC2) was measured (Dionisio-Sese and Tobita, 1998). Electrolyte leakage (EL) was calculated using the following formula:
EL= (EC1/EC2 ) ×100
Determination of the Enzyme Activities of SOD, CAT, POD, and APX
Leaf samples weighing 0.5 g were ground with a pestle in a chilled mortar and then homogenized in 8 mL of 50 mM pre-cooled potassium phosphate buffer (pH 7.8) containing 0.1 M ethylenediaminetetraacetic acid (EDTA) and 1% (w/v) polyvinylpyrrolidone (PVP). Homogenates were centrifuged at 13,000 ×g for 20 min at 4°C, and the supernatants used for the enzyme assays described below.
SOD activity was assayed using the nitroblue tetrazolium (NBT) method. The 3 mL reaction mixture contained 50 mM potassium phosphate buffer (pH 7.8), 13 mM methionine, 75 μM NBT, 20 μM riboflavin, 0.1 mM EDTA-Na2, and 25 μL enzyme extract. The reaction mixture without the addition of enzyme extract was used as the control. The reaction was initiated by adding riboflavin, and mixed well in the dark. Reaction mixtures in glass test tubes were then irradiated under fluorescent lamps (4000 lx) for 36 min. When the color of the reaction mixture turned from yellow to blue-black, the absorbance was read at 560 nm. The reaction mixture without enzyme and illumination was used to zero the absorbance at 560 nm. One unit of SOD activity was defined as the amount of enzyme causing 50% inhibition of the reduction of NBT, as monitored at 560 nm.
CAT activity was analyzed following a slightly modified version of the method described by Gao (2000). The reaction solution (3 mL) contained 50 mM PBS (pH 7.0) with 15 mM H2O2 and 50 μL enzyme extract. The reaction was started by the addition of H2O2. Absorbance changes at 240 nm were followed for 3 min. One unit of CAT activity was defined as the change in absorbance of 0.01 U·min-1.
POD activity was also determined using a modified version of the method described by Gao (2000). The 3 mL reaction mixture contained 100 mM PBS (pH 6.0), 25 mM guaiacol, 15 mM H2O2, and 50 μL enzyme extract. The reaction was initiated by the adding H2O2. The increase in absorbance at 470 nm was followed for 6 min. Decomposition of 1 mM H2O2min-1 was defined as one unit of POD activity.
APX activity was estimated according to the method described by Nakano and Asadak (1981). Samples were monitored to observe the decrease in absorbance at 290 nm as the ascorbate was oxidized. The reaction solution contained 50 mM PBS (pH 7.0), 9 mM reduced ascorbate (AsA), 15 mM H2O2, and 50 μL enzyme extract in a final volume of 3 mL. The reaction was started by adding H2O2. Measurements showing 1 mM ascorbate oxidized per min was defined as one unit of APX activity.
Staistical Analysis
Each data point was the mean of four replicates (n = 4). All data were subjected to ANOVA, and means were compared using Duncan’s multiple range test; significant differences occurred at the 5% level. In all figures, the spread of values is shown as error bars representing standard errors (SE) of the means. Calculation of the correlation coefficient and multiple linear regression analysis were carried out using the statistical software package SPSS 18.0.
Results
Growth Status
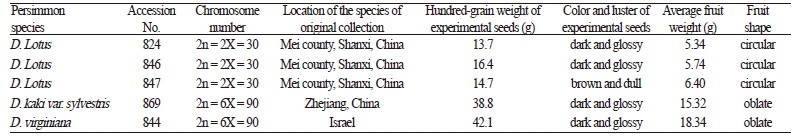
The correlation coefficient between relative height growth and salt stress was 0.913 (p < 0.001). Significant changes in relative height growth occurred in all Diospyros accessions. Curves of relative height growth in D. virginiana 844 and D. kaki var. sylvestris 869 both peaked at 0.5% NaCl, which were higher than those of 0.0% NaCl by 15.4% and 10.5%, respectively. Curves continued to decrease with increasing salt concentration, and percentages were reduced to 33.1% and 21.1% at 2.0% NaCl, respectively (Fig. 1). For three accessions of D. lotus (824, 846, and 847), relative height growth significantly decreased by 72.7%, 82.1%, and 73.9% under severe salt stress (2.0% NaCl) compared with their corresponding controls, (Fig. 1).
Effects of Salinity Stress on Leaf RWC
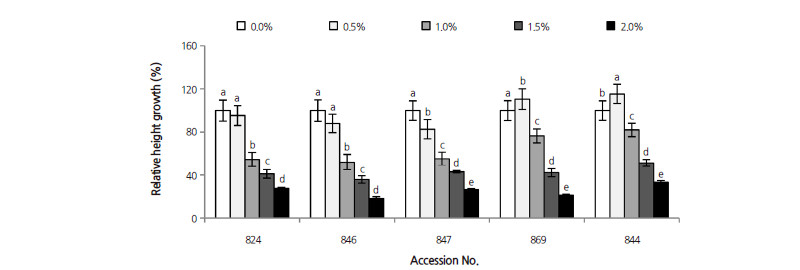
The correlation coefficient between leaf RWC and salt stress was 0.721 (p < 0.001). Leaf RWC in D. virginiana 844 declined with increased salinity, although this was statistically insignificant. In contrast to D. virginiana 844, D. kaki var. sylvestris 869 showed a marked change in leaf RWC, decreasing by 33.4% at 2.0% NaCl (Fig. 2). Among the three accessions of D. lotus, the minimum decrease in the RWC was observed in accession number 847, which decreased by 28.7%; this was followed by accession numbers 824 and 846, which decreased significantly by 35.4% and 45.7%, respectively. D. virginiana 844 maintained leaf RWC better than other persimmon rootstocks.
Lipid Peroxidation
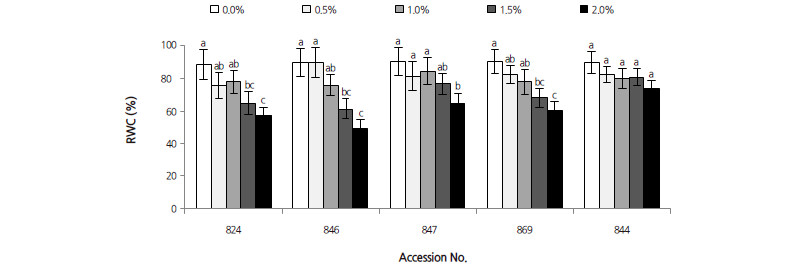
Lipid peroxidation levels were determined in terms of MDA content (see Fig. 3). The correlation coefficient between MDA and salt stress was 0.613 (p < 0.001). NaCl concentration and different persimmon rootstocks were significant factors for MDA content; the MDA contents of all accessions of Diospyros increased with increasing NaCl concentration. MDA levels in D. virginiana 844 and D. kaki var. sylvestris 869 both peaked at 2.0% NaCl, and increased by 25.3% and 70.2% compared with their corresponding controls, respectively. In D. lotus, the MDA content of accession numbers 824, 846, and 847 increased significantly by 125.1%, 95.2%, and 65.9% at 2.0% NaCl, respectively. These results indicate that leaf cells of D. lotus were more easily damaged compared to other Diospyros accessions.
H2O2 Generation
The correlation coefficient between H2O2 and salt stress was 0.399 (p < 0.001). H2O2 concentration in the leaves of all accessions progressively increased when young seedlings were subjected to salinity stress (Fig. 4). Leaves of D. virginiana 844 and D. kaki var. sylvestris 869 showed no significant change in H2O2 content, and increased by 32.5% and 33.7% at 2.0% NaCl, respectively. In the three D. lotus accessions subjected to increasing salinity stress, H2O2 levels increased remarkably (except for 824), reaching 111.9%, 181.5%, and 167.3% at 2.0% NaCl in 824, 846, and 847, respectively, compared to their corresponding controls. These results indicate that salt stress induced all three accessions to accumulate H2O2. D. lotus accumulated more H2O2 than other persimmon rootstock accessions.
Electrolyte Leakage
Solute leakage from cells was assessed to measure the extent of membrane damage in saline conditions. The correlation coefficient between EL and salt stress was 0.894 (p < 0.001). Analysis revealed that electrolyte leakage from the leaves of the five Diospyros accessions increased significantly with increasing salinity (Fig. 5). The smallest increase was measured in D. virginiana 844, with an amount exceeding that of control seedlings by 122.0% at 2.0% NaCl. This was followed by D. kakivar. sylvestris 869, which increased by 175.5%. The largest increasing magnitude of electrolyte leakage occurred in D. lotus under salinity stress, which increased markedly by 203.9%, 198.9%, and 152.1% at 2.0% NaCl in accession numbers 824, 846, and 847, respectively.
Antioxidant Enzymes
Clear differences in SOD activity in response to salinity were observed in the leaves of all accessions (Fig. 6). The correlation coefficient between SOD activity and salt stress was 0.055 (p > 0.05). At 2.0% NaCl, leaves of D. lotus accession numbers 824, 846, and 847 showed significant decreases in SOD activity of 37.1%, 60.2%, and 19.1%, respectively. SOD activity in the leaves of D. lotus accession numbers 824, 846, and 847 showed significant decreases in SOD activity of 37.1%, 60.2%, and 19.1%, respectively. SOD activity in the leaves of D. kaki var. sylvestris 869 increased to the maximum at 1.5% NaCl, which was markedly higher than that of the control by 95.7%, and then declined at 2.0% NaCl. SOD activity in the leaves of D. virginiana 844 increased to a maximum level at 2.0% NaCl and was 83.1% higher than that of the control plants.
Stress-induced CAT activity in leaves was different for all accessions (Fig. 6). The correlation coefficient between CAT and salt stress was 0.582 (p < 0.001). CAT activity in leaves of D. virginiana 844, D. kaki var. sylvestris 869, and the 847 accession of D. lotus, all increased with the increasing salt concentrations, and all peaked at 2.0% NaCl; this was significantly higher than the corresponding controls by 469.0%, 984.2%, and 429.4%, respectively. CAT activity in the 824 accession of D. lotus dropped with increased salinity and was 69.4% lower than that of the corresponding control, whereas CAT activity in the 846 D. lotus accession increased by 81.8%, with the curve peaking at 1.5% NaCl.
There was a striking difference in leaf POD activity in all accessions subjected to salinity stress (Fig. 6). The correlation coefficient between POD and salt stress was 0.401 (p < 0.001). POD activity increased significantly at 2.0% NaCl in the leaves of D. virginiana 844, which was around 10.5 times that of the control plants. The curve of D. kaki var. sylvestris 869 had a peak at 1.5% NaCl, with a level exceeding that of control plants by 257.7%. In D. lotus, leaf POD activity for the three accessions 824, 846, and 847 increased by 765.5%, 1550.0%, and 1135. 3% at 1.0%, 1.0%, and 1.5% NaCl, respectively, then declined.
Changes in salt-induced APX activity were different between accessions (Fig. 6). The correlation coefficient between APX and salt stress was 0.252 (p < 0.05). When plants were subjected to salinity stress, D. virginiana 844 and D. kaki var. sylvestris 869 both peaked at 1.5% NaCl and significantly exceeded the APX activity levels of their corresponding controls by 301.8% and 513.6%, respectively. In D. lotus, leaf APX activity in the 846 D. lotus accession increased to the maximum at 1.0% NaCl, which was higher than that of the control by 48.1%; APX activity at 2.0% NaCl in both the 824 and 847 D. lotus accessions decreased by 49.5% and 17.1% compared with corresponding controls.
From a series of linear regression analyses, these results indicate that the variables Rg, RWC, MDA, EL, and CAT have a strong relationship with salt stress; the correlation coefficient of each variable was more than 0.5. Some variables (H2O2, SOD, POD, and APX) with a correlation coefficient less than 0.5 have a weak relationship with salt stress. Fitting all of these variables into multiple regression analyses, the multiple correlation coefficient between all variables and salt stress was 0.949 (p < 0.001), implying that, overall, these variables can adequately explain the responses of young seedlings of persimmon rootstock to salt stress.
Discussion
Salt stress affects both plant growth and water status (Hernández et al., 2000; Sekmenet al., 2007). Leaf RWC was shown to be an appropriate parameter to represent the stress level of plants, and indicates the plant’s water status. High leaf water content is usually caused by a plant’s strong water-holding ability; likewise, a strong resistance to stress gives rise to a lesser degree of damage to the cell membrane. In the present study, D. lotus exhibited a stronger decrease in leaf RWC, while no significant change was found in RWC of D. virginiana 844 under salt stress. Decrease in leaf RWC seems to suppress the growth of all Diospyros accessions after high-salt treatment. Low-salt treatment did not affect the growth of D. virginiana 844 or D. kaki var. sylvestris 869, while growth in D. lotus was inhibited most severely at a high NaCl concentration. RWC level can be reflected in the morphological appearance of seedlings; D. virginiana 844 had the strongest salt tolerance.
Many researchers have reported that salt stress induces oxidative damage in plant tissues, expressed as enhanced MDA content, H2O2 level, and the degree of EL (Dionisio-Sese and Tobita, 1998; Chaparzadeh et al., 2004; Carrascoon-Ríos and Pinto, 2014). This opinion is supported by our study, which also found evidence of salt-induced oxidative stress in Diospyros leaves. MDA, the product of lipid peroxidation, was used to evaluate plants’ tolerance to oxidative damage provoked by salt stress. In this study, MDA content increased to different degrees in the leaves of different persimmon rootstocks subjected to high salinity. D. virginiana 844 accumulated the least MDA, while the highest accumulation of MDA was found in D. lotus. Numerous studies have shown that maintaining low levels of MDA is related to high salt tolerance in many plants, e.g., cotton, Cakile maritime, and myrtle (Meloni et al., 2003; Amor et al., 2006; Acosta-Motos et al., 2015). Over-production of H2O2 in plant tissues is toxic, may reduce plant growth, and causes damage in macromolecules, especially in membrane lipids; D. lotus accumlated more H2O2 compared with other accessions. Enhanced membrane permeability leads to an increase in solute leakage, thus decreased plant resistance to salinity. EL provides an indication of cell membrane injury index; fewer electrolytes were lost from the cells of D. virginiana 844, while the biggest increase in membrane permeability was observed in D. lotus. These results suggest that salinity indeed induces oxidative damage in the different persimmon rootstocks observed, with D. lotus being more affected by salinity, compared with the other accessions.
Salinity tolerance in plants is related to their antioxidant response (Shalata et al., 2001). In many cases, tolerant species have higher activity of antioxidant enzymes (SOD, CAT, POD, and APX) to protect themselves from ROS accumulation caused by salt treatment (Panda and Khan, 2004; Sekmen et al., 2007). SOD is considered the first line of defense against ROS by converting O2.- into O2 and H2O2 (Scandalios, 1993; Gómez et al., 2004). Furthermore, changes in SOD activity are different in different plant species, as well as in different intensities and durations of exposure to salinity. Meloni et al. (2003) reported that SOD activity increased in both cotton cultivars under salinity stress, and in the salt-tolerant cultivar ‘Pora’, SOD activity was higher than that of the salt-sensitive cultivar ‘Guazuncho’. A reduction in SOD activity caused by salt stress has also been found in rice and corn (Khan and Panda, 2008; Carrasco-Ríos and Pinto, 2014). In our results, SOD activity increased significantly in D. virginiana 844 (by 83.1% at 2.0% NaCl), and in D. kaki var. sylvestris 869 (by 95.7% at 1.5% NaCl), while remarkable decreases in SOD activity were observed in salt-sensitive varieties, as well as in the three accessions of D. lotus (by 37.1%, 60.2%, and 19.1% in 824, 846, and 847 at 2.0% NaCl, respectively). Our results demonstrate that D. virginiana 844 is a salt-tolerant accession with a better superoxide radical-scavenging ability. Our results were similar to those observed in other salt-tolerant plants, such as salt-tolerant genotypes of rice, foxtail millet, barley, Cakile maritima, tomato, and Plantago maritima (Dionisio-Sese and Tobita, 1998; Sreenivasulu et al., 2000; Acar et al., 2001; Amor et al., 2006; Koca et al., 2006; Sekmen et al., 2007).
SOD activity decreased in leaves of D. lotus salt-sensitive rootstock, which impaired seedlings’ ability to scavenge O2.- radicals. Hence, more oxygen radical species accumulated, which could cause damage to the membrane. The degree of membrane damage was detected by measuring the amount of MDA, which is produced when polyunsaturated fatty acids in the membrane undergo peroxidation. The structure and properties of the membrane were affected by changes to unsaturated fatty acids that increased lipid peroxidation and free radical formation in salt-sensitive species after salt treatment. The D. lotus leaf cells may have also increased in membrane permeability or suffered a loss of membrane integrity, as manifested by increasing solute leakage. Previous studies have reported salt stress-induced electrolyte leakage in foxtail millet and corn (Sreenivasulu et al., 2000; Carrasco-Ríos and Pinto, 2014).
CAT converts H2O2 into H2O (Meloni et al., 2003). CAT activity levels were different between accessions in response to salt stress. Bor et al. (2003) reported that CAT activity increased in sugar beet and wild beet. Conversely, a decrease in CAT activity has been reported in both salt-sensitive and salt-tolerant rice cultivars (Khan and Panda, 2008). CAT activity was almost unchanged in Catharanthus roseus (Jaleel et al., 2007). In our study, CAT activity increased in all Diospyros accessions (except for the 824 accession of D. lotus), and CAT activity in D. virginiana 844 and D. kaki var. sylvestris 869 was markedly higher than that in D. lotus. Our experimental results are in agreement with those described by Wang and Han (2009), who showed higher CAT activity in a salt-tolerant cultivar of alfalfa.
Dionisio-Sese and Tobita (1998) revealed that POD is not only involved in scavenging H2O2 produced in chloroplasts, but also in growth and developmental processes. POD activity of plants was shown to be up-regulated in order to reduce oxidative stress, cell membrane damage and altered Ca2+ concentrations caused by ROS generated by salinity treatment (Lucrezia et al., 2012). In our study, the biggest increase in POD activity was measured in D. virginiana 844; this increase of 946.15% occurred at 2.0% NaCl. Meanwhile, POD activity in other accessions began to decline at 1.0% or 1.5% NaCl. This may indicate that D. virginiana 844 possesses a higher capacity to decompose H2O2. We found a positive correlation between POD activity and salt resistance of Diospyros persimmon rootstock. However, other research has reported decreased POD activity in a salt-tolerance rice variety, and a significant increase in POD activity in a salt-sensitive rice variety (Dionisio-Sese and Tobita, 1998; Demiral and Türkan, 2005). This suggests a negative correlation between POD activity and salt tolerance.
APX functions in the ascorbate-glutathione cycle, which is induced by high levels of intercellular H2O2 (Lee et al., 2001); APX activity also plays an important role in protecting plants from H2O2 resulting from environmental stress (Noctor and Foyer, 1998). In our study, increased APX activity was reported in D. virginiana 844 and D. kaki var. sylvestris 869 accessions, whereas a decrease was reported in D. lotus (except for accession 846). Similarly, higher APX activity has been reported in salt-tolerant tomato, foxtail millet, and rice (Shalata and Tal, 1998; Sreenivasulu et al., 2000; Demiral andTürkan, 2005).
We compared rootstocks of Diospyros L. differing in their responses to salinity stress by adding different salt concentrations to healthy young plants for 25 days. In this study, we identified and compared the effects of salt treatment on growth and leaves in terms of MDA, H2O2, EL, and antioxidative enzymes (SOD, CAT, POD, and APX) in different persimmon rootstocks. After salt treatment, the variables identified in different persimmon rootstock samples were found to be closely related to salinity tolerance. It was clear that lower MDA, H2O2 contents, and EL, and increased SOD, CAT, and POD activities are linked to increasing NaCl concentration (except for APX, which reached peak at 1.5% NaCl) in D. virginiana 844. These activities can effectively scavenge ROS and thus improve the salt tolerance of plants. For the three accessions of D. lotus, higher MDA content, EL, H2O2 concentration, and SOD activity decreased in all NaCl treatments, and CAT, POD, and APX activities were up-regulated at 0.5%, 1.0% and 1.5% NaCl. Moderate MDA content, H2O2 concentration, EL and the activities of SOD, CAT, POD, and APX increased at 0.5%, 1.0% and 1.5% NaCl (except for CAT activity) in D. kaki var. sylvestris 869. The activities of protective enzymes increased at low salt concentrations to adapt to salinity stress, which can be used effectively to scavenge ROS and improved the salinity tolerance of plants. There was an imbalance between the generation of ROS and effective antioxidant systems when plants were subjected to a high concentration of salt, similar to other studies on Calendula officinalis and Cakile maritime (Chaparzadeh et al., 2004; Amor et al., 2006). Some differences were noted in the damage caused to different rootstocks in salinity-tolerant and salinity-sensitive seedlings; the main reason for this salt stress damage was the disturbance of oxygen metabolism. Higher activities of protective enzymes are highly favored by salinity-tolerant species, which scavenge free oxygen to reduce damage to the membrane system.
From the above results, we have confirmed that D. virginiana 844 had higher salinity resistance than the other persimmon rootstocks studied. Future studies should address persimmon rootstock responses to salinity stress at different stages of growth and ROS concentrations, and elucidate the mechanisms of the ROS-inducing antioxidant system.