Introduction
Materials and Methods
Plants and Growth Conditions
Growth Parameters
Photosynthetic Parameters
Chlorophyll Fluorescence
Data Analysis
Results and Discussion
Conclusions
Introduction
Plants in the field are permanently exposed to various environmental conditions such as light, soil water content, temperature, and soil nutrients. Among these, light intensity is one of the most important plant growth requirements. Light energy is used to produce ATP and NADPH in the light reactions of photosynthesis (Taiz and Zeiger, 2007). Subsequently, the consumption of ATP and NADPH is coupled to reactions in which the reduction of CO2 to carbohydrates is catalyzed by a light-independent reaction. Under high irradiance, however, the photosynthetic apparatus absorbs excessive light energy, causing the inactivation or impairment of the chlorophyll-containing reaction centers of the chloroplasts (Bertamini et al., 2006). As a consequence, photosynthetic activity is hindered by photoinhibition (Osmond, 1994). Alternatively, under low irradiance, ATP is too limited to allow for carbon fixation and carbohydrate biosynthesis. This leads to decreased plant growth and development (Dai et al., 2009).
Jeffersonia dubia (Maxim.) Benth. & Hook. f. ex Baker & S. Moore (Berberidaceae) is a herbaceous perennial native to Korea. It is distributed in Northeastern Asia and ranges from Southern Korea to Northeast China and Eastern Siberia (Hutchinson, 1920). J. dubia blooms in early spring and has showy light purple flowers and attractive heart-shaped leaves. Thus, it has potential as a new ornamental crop for gardens (Huang, 1995). J. dubia has value not only as an ornamental plant, but also when used for medicinal purposes. It has also been used in Korean folk medicine as an antidote and stomachic (Bae, 2000). Extracts of J. dubia roots have berberine, which has been identified as a promising cholesterol-lowering drug that acts through different pathways than those of statins (Kong et al., 2004). However, because of extensive collection and changes in land usage, the total number and size of the populations are decreasing. One of the problems for producing J. dubia is the long cultivation period; the plant takes at least three years to bloom after sowing.
Although J. dubia has been reported to be well adapted to shade (> 90% shade of incident sunlight), because plants grow naturally in deciduous forests (NIER, 2004), we assume that J. dubia has a long cultivation period because of the lack of light in a rich forest. There is limited information on the effect of shading on the growth and physiological response of J. dubia. Thus, the objectives of this work are to observe the effects of various shading on photosynthesis, chlorophyll fluorescence, and growth of J. dubia, and to determine the proper shading level.
Materials and Methods
Plants and Growth Conditions
One-year old J. dubia plants were collected from an ex situ conservation habitat of the Korea Botanic Garden in Pyeongchang, Gangwon, Korea (37°37'S, 128°39'E) and planted on 31 Mar. 2009. Plants were planted in 10-cm pots containing a mixture of peat, perlite, and sand (4:1:5), and grown in a greenhouse under natural light conditions. Three treatments were considered, with 50, 75, and 95% shade nets erected horizontally above the plants, while unshaded plants were used as a control (0% shade). Screens were positioned 1 m high, about 0.8 m above the canopies. The experiments were conducted in a greenhouse located at the experimental farm of Seoul National University, Suwon, Korea (37°27'S, 126°99'E), from 3 Apr. 2009 to 31 Jul. 2009. We measured the diurnal variation of photo-synthetic photon flux density (PPFD) on 1 April under each treatment with a Pyranometer (LI-1400, Li-Cor, Lincoln, NE, USA) and recorded air temperature at 1-hour intervals with a thermo data logger (Watch Dog Model 450, Spectrum Technologies, Inc., Plainfield, IL, USA). Irrigation was provided manually to saturation at 09:00 AM every day. Slow-release fertilizer (23N-6P-17K, 43.5 g・m-2, Haifa Chemicals Ltd., Haifa Bay, Israel) was applied once after planting.
Growth Parameters
Plant height, leaf length and width, number of leaves, and dry matter partitioning were measured at 15 weeks after treatment. At the end of the experiment, the shoots and roots of each plant were immediately dried at 80°C in an oven for 72 hours in order to measure dry weight.
Photosynthetic Parameters
Net CO2 assimilation rates (A) were determined using a LI-6400 portable photosynthesis system (Li-Cor, Lincoln, NE, USA) after 40 days of treatment (18 June 2009). The parameters were measured on fully expanded leaves from 09:00 to 17:00 hours on 18 Jun. 2009. The leaf chamber temperature and the CO2 concentration were maintained at 25°C and 400 μmol・CO2・mol-1・s-1, respectively. PPFD was increased from 0 to 1500 μmol・m-2・s-1 (0, 20, 50, 100, 300, 500, 1000, and 1500 μmol・m-2・s-1). A was recorded at each of the seven light levels following a 10-minute acclimation period.
Chlorophyll Fluorescence
Chlorophyll fluorescence was determined during the day. Leaves were dark-adapted for 30 minutes before measurements were taken using leaf clips. Between 10:00 AM to 11:00 AM, the emitted fluorescence was recorded for the dark-adapted leaves with a portable pulse amplitude modulation fluorometer (PAM-2000, Walz, Effeltrich, Germany). A measuring light of 0.6 kHz and less than 0.1 μmol・m-2・s-1 PPFD was irradiated to obtain the minimum fluorescence in the dark-adapted state (F0), and then a saturating light pulse at about 8000 μmol・m-2・s-1 PPFD was irradiated for 0.8 s to induce the maximum fluorescence in the dark- adapted stated (Fm). Potential quantum yields in the dark- adapted states were estimated from (Fm-F0)/Fm = Fv/Fm, representing the efficiency of energy captured by open PSII (Genty et al., 1989). Chlorophyll fluorescence was measured on 12 May and 29 Jun., 2009, respectively.
Data Analysis
Experiments were conducted in a completely randomized block design with 22 plants per treatment. Collected data were analyzed by analysis of variance (ANOVA) following the general linear model procedure, SAS 9.1 (SAS Institute Inc., Cary, USA). Analysis and graph module were made using Sigma Plot 10.0 (SPSS, Inc., Chicago, USA). When significant differences were found, the means were separated using Duncan’s multiple range test at the 5% level.
Results and Discussion
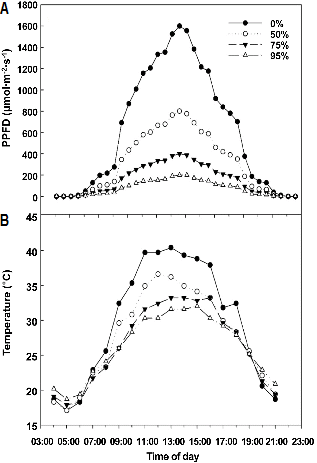
The PPFD level was reduced by the utilization of black mesh cloth. Compared to the 0% shade treatment, the PPFD level was reduced by 50.6, 74.5, and 95.9% in the 50, 75, and 95% shade treatments, respectively, at 12:30 on a sunny day in the greenhouse (Fig. 1A). Shading also led to a decrease in the air temperature. During the hottest hours (11:00-13:00 hours), air temperatures under 50, 75, or 95% shade were approximately 4, 8, or 12°C lower than those under 0% shade, respectively (Fig. 1B).
|
Fig. 1. Diurnal change of canopy PPFD (A) and air temperature (B) under different shading levels in a greenhouse on 1 Apr. 2009 in Suwon, Korea. |
Plants grown under 75% and 95% shade were 1-2 cm taller than plants from the other treatments (0 and 50% shade) (Table 1). Under shaded conditions, the plant height, shoot length, and flower stem length were also increased compared to unshaded conditions in Hibiscus syriacus L. (Yoo and Kim, 1997), Hydrangea serrata for. acuminate (Lee et al., 2008), lily (Sorrentino et al., 1997), and ferns (You et al., 2005), indicating that plants show a shade avoidance response under shaded conditions. Shading had no significant effects on leaf length and width. However, the number of leaves and dry weight were affected by the shade treatments (Table 1). Plants treated with 0% and 50% shade produced more leaves compared with 75% and 95% shade treated plants. The dry weights of shoots and roots under 50% shade were 187% and 420% higher than those under 95% shade, respectively (Table 1 and Fig. 3). The shoot:root ratio was increased with increasing shading rate. Under 95% shade, the shoot:root ratio was 0.97, two times higher than the 0 or 50% shade treatments. In Rhamnus alaternus, a decrease in light induces an increasing shoot:root ratio, and this phenomenon was explained by a reduction in evaporative demand due to shading, affecting the water absorption needs, which possibly diminished the root growth (Miralles et al., 2011).
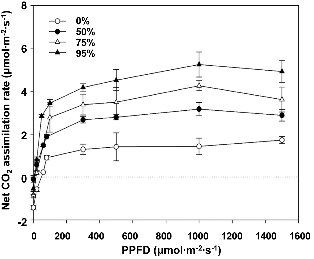
Regardless of the shading treatment, the net CO2 assimi-lation rates (An) increased rapidly as PPFD increased to 150 μmol・m-2・s-1, increasing slowly to a maximum, followed by a slow decrease as PPFD was increased to 2000 μmol・m-2・s-1 (Fig. 2). A PPFD higher than the saturation point for photo-synthesis induces non-radiative de-excitation mechanisms at the chlorophyll level in the PSII complexes, resulting in a decreased maximum quantum yield of photosynthesis (Pastenes et al., 2003). The light compensation points (LCPs) in 0% shade treated plants were slightly higher than those of the 50%, 75%, and 95% shade-treated plants. It was also reported that Guzmania monostachia damaged by high light intensities had increased LCPs (Maxwell et al., 1992), indicating that J. dubia was also injured by high light intensities under 0% shade. Both An and maximum An varied significantly (p < 0.05) among the light intensity treatments. The maximum An under 0, 50, 75, and 95% shade was approximately 1.9, 2.9, 3.7, and 4.7 μmol CO2・m-2・s-1, respectively. High light intensity causes a decrease in the rate of PSI-mediated electron transport (Reichenauer et al., 1997), and it is linearly related with inhibition of PSII photochemistry (Long et al., 1994). These results indicate some degree of decline of photosynthetic capacity under high light above 95% shade.
|
Fig. 2. Net CO2 assimilation rate curves from leaves of Jeffersonia dubia under different shading levels in a greenhouse on 18 Jun. 2009. Vertical bars represent standard errors of the means. |
|
Fig. 3. Photograph showing the growth of Jeffersonia dubia under different shading levels in a greenhouse at 15 weeks after treatment. |
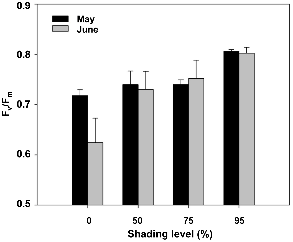
The combination of high light and high temperatures can also affect photosynthetic efficiency (Barber and Andersson, 1992), leading to photoinhibition (Demmig-Adams, 1990), which can be measured by chlorophyll a fluorescence. A very good correlation has been observed between photo-inhibition and a decreased ratio of variable to maximum fluorescence, i.e., the Fv/Fm ratio (Bolhar-Nordenkampf and Öquist, 1993). The Fv/Fm parameter has been correlated with the maximum quantum yield of photosynthesis (Björkman and Demmig-Adams, 1987; Bolhar-Nordenkampf and Öquist, 1993; Cornic and Briantais, 1991), reaching a maximum theoretical value of 0.85 (Bolhar-Nordenkampf et al., 1991). Fluorescence measurements were performed on 12 May and 29 Jun. 2009. In 95% shade, Fv/Fm values remained near the optimal values of 0.8 for both dates, indicating that the heavy shading was effective in preventing photoinhibition.
Therefore, this shade-tolerant plant could adapt to grow under a light intensity of > 90% shade in nature, and such high An and Fv/Fm were shown under this low light intensity condition. On the other hand, a decrease in the Fv/Fm value indicated photoinhibition in the 0, 50, and 75% shade treated plants. Under 50 and 75% shade, the Fv/Fm value declined moderately, holding steady in both May and June. However, exposure to the high irradiance conditions of the 0% shade treatment resulted in a greatly reduced Fv/Fm value, which was apparent in June. Under 0% shade, the Fv/Fm value was 0.72 ± 0.01 in May and 0.62 ± 0.05 in June, showing severe chronic photoinhibition of photo-synthesis.
|
Fig. 4. Changes in the maximum quantum efficiency (Fv/Fm) of Jeffersonia dubia on 12 May and 29 Jun. 2009 under 0, 50, 75, and 95% shade levels. Vertical bars represent standard errors of the means. |
This kind of photoinhibition causes a decrease in photo-synthetic efficiency, and may lead to limitations in daily carbon gain with a lower capacity for plant dry mass accumulation (Ögren and Sjöström, 1990). In this study, the dry weight of the shoots and roots under 0% shade was 24% and 22% lower than that under the 50% shade (Table 1). In Tetrastigma hemsleyanum, the low rate of electron transport through PSII and the high non-photochemical quenching of chlorophyll fluorescence values combined with unhealthy leaf morphology (smaller leaf size and yellow leaf color), as well as the low An of the plants grown under high light intensity suggested that excess light energy damaged the plants (Dai et al., 2009). This is thought to be due to the formation of destructive oxidative molecules, resulting in damage to the photosynthetic apparatus via photoinhibition (Aro et al., 1993; Krause, 1988). Shading resulted in lower air temperature compared to 0% shade treated plants throughout the day (Fig. 1B). Leaf temperature increases cause increased susceptibility to photoinhibition (Gamon and Pearcy, 1990). Similarly, decreases in the Fv/Fm value in the 0% shade treatment were probably dependent on PPFD levels associated with high temperatures.
According to the value of A and Fv/Fm, the photosynthetic ability of J. dubia was the best when plants were grown under 95% shade. This characteristic might allow J. dubia to be used as indoor plants. However, 95% shade-treated plants had the lowest dry weight of the shoots and roots, whereas the 50% shade had the greatest (Table 1). These results indicated that a light intensity under 95% shade led to diminished production of carbohydrates. At low light intensities (below the light saturation point), the rate of photosynthesis increases proportionately as the light intensity increases. However, under 95% shade, the light coming through the shade cloth was quite low, below 100 μmol・m-2・s-1 PPFD on a sunny day (Fig. 1A). The light saturation point of plants grown under 95% shade was around 1000 μmol・m-2・s-1 PPFD (Fig. 2).
Conclusions
Shading levels significantly affected the growth of J. dubia. Plant growth (dry matter and number of leaves) was optimal when plants were grown under 50% shade. With high light intensity under 0% shade, photosynthetic activity was depressed, likely due to photoinhibition. On the other hand, light intensity reductions greater than that due to 75% shade resulted in irradiation insufficient to maintain An, influencing carbon balance and consequently leading to a decline in plant growth. Thus, approximately 50% shade is concluded to be the optimum light irradiance condition for J. dubia cultivation. In view of the low light acclimation capacity of this plant, wild J. dubia is not expected to occur in the shaded understory where the light intensity is greater than 50% of ambient values. The light intensity of natural habitat areas with less than that coming from 10% ambient light will decrease the pro-ductivity of J. dubia. For agricultural purposes, in order to obtain high yields of J. dubia, we recommend trying to achieve approximately 50% ambient light with a shade net, which reduces incident PPFD and leaf tempera-tures.