Introduction
Materials and Methods
Reagents
Plant Material
Preparation of Plant Extracts
Alpha-Glucosidase Inhibition Assay
Conditions for High Performance Liquid Chromatography (HPLC)
Statistical Analysis
Results and Discussion
Introduction
Diabetes mellitus is a chronic metabolic disorder caused by an absolute or relative lack of insulin. It is characterized by hyperglycemia and disruptions in carbohydrate, protein, and fat metabolism. It is also associated with several complications, such as retinopathy, neuropathy, and peripheral vascular insufficiency (Chehade and Mooradian, 2000), and represents a global health problem (Pandey et al., 2011). There has been a sharp increase in the number of diabetic patients worldwide, including Korea, because of changes in lifestyle and diet. High carbohydrate diets elevate postprandial hyperglycemia, which can progress to type II diabetes (Gerrits and Tsalikian, 1993). Inhibitors of α-glucosidase and α-amylase have been suggested as an effective means of lowering postprandial hyperglycemia, which is
known to be the earliest defect occurring in type II diabetes (Matsui et al., 2006).
Pepper (Capsicum annuum L.), belonging to the Solanaceae plant family, is the most highly consumed season-free crop in the world (Wahyuni et al., 2013). The pepper fruit, widely used in dried or fresh forms, is one of most important ingredients in traditional Korean fermented foods, including various kinds of kimchi (Ku and Kang, 2010). The red pepper fruit contains a range of essential nutrients and bioactive compounds, including provitamin A and flavonoids that are known to have antioxidant, antimicrobial, antiviral, anti-inflammatory, and anticancer effects (Minguez-Mosquera and Hornero-Mendez, 1993; Lee et al., 1995; Ku et al., 2009; Luo et al., 2011). Capsaicin, known to confer a spicy taste, is a main constituent of the pepper fruit, and has been extensively studied for its pharmacological relevance (Luo et al., 2011).
A previous study found that phenolic compounds from pepper leaves, including polyamine phenolic conjugates, chlorogenic acid derivatives, anthocyanin, and flavone glycosides, have antioxidant activities (Kim et al., 2014). Recently, Park et al. (2016) reported that pepper (C. annuum L. cv. Dangjo) leaf extract inhibited the activities of α-glucosidase and α-amylase-possibly caused by the presence of luteolin 7-O-glucoside (L7G), a flavonoid glucoside. To date, few studies have analyzed α-glucosidase inhibitory (AGI) activity using pepper fruits. This study, therefore, set out to investigate the AGI effects of pepper fruits.
Materials and Methods
Reagents
Alpha-Glucosidase from Saccharomyces cerevisiae, p-nitrophenyl glucopyranoside (pNPG), acarbose, and L7G were purchased from Sigma-Aldrich (MO, USA). All other unnoted chemicals were purchased from Sigma-Aldrich.
Plant Material
Fruits of 14 pepper cultivars (C. annuum) were obtained from Asia Seed Corporation, Seoul, Korea (Table 1). Three fresh fruits per cultivar were wiped with a paper towel to remove contaminants, and were cut into pieces before being placed in liquid nitrogen. Fruits were dried at 55°C for 2 d, and ground to a fine powder using a mortar and pestle. The powdered samples were stored at 4°C until use.
Preparation of Plant Extracts
Two hundred and fifty milligrams of powdered sample was divided into two portions for extraction with 2 mL of 70% ethanol (EtOH) and water (H2O), and incubated at 55°C for 16 h. Extracts were filtered through the Empty PD-10 column (GE Healthcare, PA, USA), and concentrated in a speed vacuum centrifuge (CVE-2200, Eyela, Japan). Samples were extracted in triplicate, and the dried extracts were stored at 4°C until use.
Alpha-Glucosidase Inhibition Assay
The effect of pepper fruits on α-glucosidase activity was determined using the procedure described by Kim et al. (2005), with some modifications. The pNPG substrate solution was prepared in 20 mM phosphate buffer (pH 6.9). One hundred microliters of α-glucosidase (1.0 U·mL-1) was preincubated with 50 µL of 100%, 50%, and 25% extracts (70% EtOH and H2O) for 10 min. Then, 50 µL of substrate (3.0 mM pNPG dissolved in 20 mM phosphate buffer at pH 6.9) was added to start the reaction. The reaction mixture was incubated at 37°C for 20 min, and stopped by adding 2 mL of 0.1 M Na2CO3. Alpha-glucosidase activity was determined by measuring the yellow-colored paranitrophenol released from pNPG at 405 nm. The blank control was prepared using the same procedure, except that α-glucosidase was replaced with 20 mM phosphate buffer (pH 6.9).
Percentage inhibition was calculated as follows:
% Inhibition=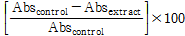
Conditions for High Performance Liquid Chromatography (HPLC)
Extracts from the cultivars Mee-In (MI) and Soon-Han-Gil-Sang (SG) were prepared using the same procedure as described above for the AGI assay. Dried extracts were reconstituted in 150 µL methanol (MeOH) before being injected into the HPLC system. The HPLC system comprised an LC-20AD pump, SPD-20A ultraviolet/visible light detector, CBM-20A communications bus module, and an SIL-20AC autosampler (Shimadzu, Kyoto, Japan), which was used in combination with a 4.6 × 250 mm Syncronis C18 column (Thermo Scientific, MA, USA). The mobile phases contained two solvents: (A) 0.1% formic acid in H2O, and (B) 0.1% formic acid in acetonitrile. Samples were injected in a total volume of 20 µL, and the flow rate of the solvent was maintained at 1 mL/min. Separation of the sample extracts was carried out using the following gradient elution profile: 5% B for 10 min, which was increased to and maintained at 70% and 100% B for 35-40 min and 45-50 min, respectively, and then restored to and maintained at 5% B for 55-60 min. UV signals were monitored at 350, 300, and 210 nm. All reagents used were of analytical grade.
Statistical Analysis
Version 3.4.3 of the R program was used to perform analysis of variance (ANOVA) and t-tests. The latter was used to compare between the means of data obtained from different concentrations of SG and MI fruit extracts. Tukey’s ANOVA test was used to assess differences between the means of data obtained from extracts of different varieties of pepper fruit.
Results and Discussion
The purpose of this study was to investigate the AGI effects of 14 pepper cultivars commonly consumed in Korea. The α-glucosidase activity assay was conducted using different concentrations of extracts obtained from different solvents (70% EtOH and H2O). A list of pepper cultivars used in this study is given in Table 1, and their appearances are presented in Fig. 1. Results of the AGI assays conducted with two different solvents are shown in Fig. 2. Acarbose diluted to 25 mM was used as positive control. Undiluted extracts using 70% EtOH had almost 100% AGI activity, except for extracts of Nok-Gwang (NG), Morning (MR), and Long-Green (LG) cultivars. However, AGI activity decreased sharply when extracts from NG, MR, SG, Gil-Sang (GS), LG, Sweety (ST), Hyul-Jo K (HJ), Shin-Hong (SH), and Ai-Mat (AM) were diluted to 50%. Interestingly, there was almost no change in AGI activity between undiluted and diluted extracts (50%) of MI and Dda-Go-Ddo-Dda-Go (DG); the degree of change was lower for extracts from Shin-Jo-Hap (SJ), Mee-Nam (MN), and Dus-Shyu-Bo-Ra (DS) extracts. All of the extracts diluted to 25% showed inhibition rates of than 10% (Fig. 2A).
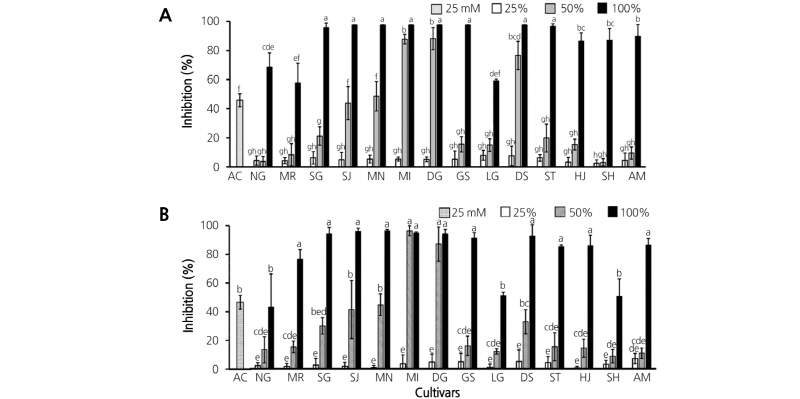
Fig. 2. Inhibitory effects of pepper fruit extracts on α-glucosidase activity. Seventy percent ethanol (A) and H2O (B) were used for extraction. Twenty-five millimoles of acarbose (AC) served as the positive control. NG, Nok-Gwang; MR, Morning; SG, Soon-Han-Gil-Sang; SJ, Shin-Jo-Hap; MN, Mee-Nam; MI, Mee-In; DG, Dda-Go-Ddo-Dda-Go; GS, Gil-Sang; LG, Long-Green; DS, Dus-Shyu-Bo-Ra; ST, Sweety; HJ, Hyul-Jo K; SH, Shin-Hong; AM, Ai-Mat. Vertical bars represent the mean ± SD value (n = 3). Different lower case letters indicate significant difference (p<0.05) according to Tukey’s test.
The AGI activities of samples extracted with H2O were also determined to compare the extraction efficiencies of 70% EtOH to H2O. There was no significant difference between 70% EtOH and H2O extraction solvents; the cultivar-dependent AGI activity associated with undiluted and 50% diluted H2O extracts followed the same trends as the corresponding samples extracted with 70% EtOH (Fig. 2B).
Interestingly, different patterns of decreasing AGI activity were detected between the two sample groups. The first group, comprising MI, DG, and DS extracts, showed almost no change upon dilution; the second group, comprising SG, SJ, MN, GS, ST, HJ, SH, and AM extracts, showed sharply decreasing AGI activity after dilution to 50%. These results suggest the presence of an unknown compound or compounds in pepper cultivars that inhibit α-glucosidase in a concentration- dependent manner. To elucidate this possibility, AGI assays of SG and MI sample extracts (which, having different decreasing AGI activity patterns were selected to represent the two groups) were compared. As shown in Fig. 3, the MI extract maintained almost 100% AGI activity up to 40% extract concentration. However, the AGI activity of SG extract began to decrease at 80% extract concentration, and continued to decrease to around 30% of AGI activity at 50% extract concentration. These patterns reflect the results in Fig. 2A. Taken together, these results suggest that the MI extract contains a higher concentration of an unknown AGI compound.
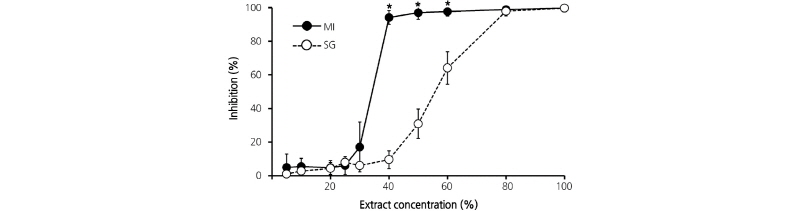
Fig. 3. Alpha-glucosidase inhibition assay of Mee-In (MI) and Soon-Han-Gil-Sang (SG) pepper fruit extracts diluted from 100% to 80%, 60%, 40%, 30%, 25%, 10%, and 5%. Ethanol (70%) was used for extraction. Vertical bars represent the mean ± SD (n = 3). Asterisks (*) indicate significant difference between MI and SG (two-sided t-test, p<0.05).
Park et al. (2016) reported that LG7, a compound in the extract of pepper leaves, inhibits α-glucosidase and α-amylase activity, and reduces blood glucose levels in diabetic mice. Extracts of MI and SG were analyzed using an HPLC system to determine LG7 levels, but no significant difference in LG7 concentrations were found between the two samples (Fig. 4A). A previous study identified active antioxidants in pepper leaves, namely, a phenolic compound, a polyamine phenolic conjugate, a chlorogenic acid derivative, anthocyanin, and a flavone glycoside (Kim et al., 2014; Bhandari et al., 2016). Telagari and Hullatti (2015) reported that Adiantum caudatum L. and Celosia argentea L. extracts rich in triterpenoids, phenolics, and flavonoids were effective α-glucosidase and α-amylase inhibitors. One or more of these phenolic or flavonoid compounds might be a key effector of AGI in pepper fruit extracts. In this study, extracts of MI and DG had the highest AGI effect of all the pepper extracts studied. NG, MR and LG showed relatively lower AGI activity, even at 100% extract concentration. After separating the components of MI and SG using HPLC, their chromatograms yielded no evidence of any major AGI compounds (Fig. 4A, B, and C). To identify AGI effectors in pepper fruits, further research is required using precise analytical methods and instruments such as HPLC, gas chromatography, and liquid chromatography-tandem mass spectrometry. This study provides an exciting opportunity to advance our knowledge of the usefulness of pepper fruits as a nutraceutical to control diabetes.

Fig. 4. Determination of alpha-glucosidase inhibitory compounds by comparing high performance liquid chromatography (HPLC) chromatograms. (A) HPLC chromatograms of L7G standard, Mee-In (MI) extract, and Soon-Han-Gil-Sang (SG) extract; UV signals monitored at 350 nm. (B) Chromatogram comparison of MI and SG extracts; UV signals monitored at 300 nm and (C) 210 nm.