Introduction
Materials and Methods
Plant material and culture
Drought stress treatment
Determination of water potential of leaves
Determination of free proline content
Determination of total sugar content
Determination of lipid peroxidation
Determination of antioxidant enzyme activities
Statistical analysis
Results
Leaf water potential
Free proline content
Sugar content
Malondialdehyde (MDA) content
Antioxidant enzyme activity
Discussion
Introduction
Drought is one of the most serious the environmental factor affecting plant growth and production. Reduced water availability immediately affects cellular water status, solute transport, photosynthesis and transpiration (Murshed et al., 2013). Drought induces higher leakage of electrons toward O2 during photosynthesis and respiration; this generates cellular oxidative stress, which enhances the production of reactive oxygen species (ROS) (Sánchez-Rodríguez et al., 2012). ROS, superoxide anion radical (O2・-), and hydrogen peroxide (H2O2), seriously disturb the normal metabolism by making oxidative damage to lipids, proteins, nucleic acids, and membrane function (Meloni et al., 2003). Plants have developed several antioxidant systems which counteract the cytotoxic effects of oxidative damage (Rahnama and Ebrahimzadeh, 2005), including superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD). These enzymes react with ROS to keep them at a low-level, and subsequently regenerate cellular antioxidants including glutathione reduc-tase and ascorbate peroxidase. For example, SOD converts O2・- to H2O2; subsequently, CAT and POD reduce H2O2 to water and molecular oxygen in the cytoplasm and other cellular compartments (Howarth, 2005).
Adjusting cellular osmotic status by accumulating osmoti-cally active solutes is an important adaptive response of drought-resistance plant to water deficit (Morgan, 1984). Maintaining water potential for plants under water deficit is important for growth. This can be realized by accumulating of compatible cytoplasmic solutes such as soluble sugars, proline, organic acids, and glycine betaine (Hare et al., 1998). The most important plant response to drought and other abiotic stress is one of the excessive productions of different types of metabolites and solute (Ashraf and Harris, 2008). Proline and total sugars accumulate to high levels in drought-stressed plants of many speciess (Hessini et al., 2008). These solutes are hypothesized to benefit drought-stressed plant cells by acting as the cell osmotic regulation substances and contribute to water retention (Hare et al., 1998) and achieving the protection and stabilization of macromolecules from drought-stress induced damage (Bohnert and Jensen, 1996). Proline is compatible solutes in the protection for the folded protein structure against denaturation, interacts with phospholipids to stabilize cell membranes, function as a hydroxyl radical scavenger, and play a role as a source of energy and nitrogen (Samaras, 1995).
Strawberry (Fragaria × ananassa) is a commercially important fruit growing market demand, but it is sensitive to water deficit and salinity. Strawberry plants are shallow roots, large leaf area and high water content of fruit; these physiological characteristics determine large quantities of water are needed for its normal growth and fruit yield (Klamkowski and Treder, 2006). A rapid decrease of leaf water potential was found in pot-grown strawberry plants under water deficit (Blanke and Cooke, 2006). In field studies, fruit yields of strawberry failed under soil water- deficit conditions (Krüger et al., 2002; Liu et al., 2007; Terry et al., 2007; Kim et al., 2007). Previous reports found many changes in physiology and biochemistry of strawberry in response to stress. Sun et al. (2013) found that the salicylic acid affects physiological characteristics of strawberry leaves when soil water deficit. Grant (2010) reported that water potential in strawberry leaf was significantly de-creasing when water-deficit treatment. Li et al. (2014) and Kim et al. (2011) reported that UV-C affected the quality, anthocyanin contents and antioxidant capacity of strawberry fruit. However, a detailed understanding of strawberry antioxidant systems and responses to water-deficit stress are still lacking. The purpose of this work was to study the response of Osmotic regulation substances (proline and sugars) and antioxidant enzymes (SOD, CAT and POD) of strawberry to drought stress condition.
Materials and Methods
Plant material and culture
This study used the ‘Toyonoka’ strawberry. The experi-ments were conducted at the Jiangsu Normal University, Xuzhou, Jiangsu Province, China (N34°15'48", E117°11'16"; an elevation of 46.4 m) under natural conditions in mid- March to late-May 2014. The strawberry transplants were initially grown in the experimental field of the Jiangsu Normal University, and then planted in pots in the green-house. The culture medium for potting was yellow fluvo- aquic soil of Yellow River sediment, which was a slightly high-level of calcium carbonates and soluble salts as detected by tests for alkalinity. 12 kg of culture medium was put into each pot which average soil moisture content was 12.5% and maximum soil moisture content was 29.8% (after watering to saturation). Sufficient base fertilizer (compound fertilizer purchased from the market, 42%, N:P:K was 16:8:18, Jiangsu Zhongnong Fertilizer Co., Ltd., Lianyungang, China) was added to support plant growth. Potted plants were cultivated in a greenhouse following the conditions: 25-27°C was average daily temperatures during the day and 15-17°C was the night temperatures; natural light which maximum photosynthetic photo flux was 1,087 μmol・m-2・s-1; and relative humidity was 40-65% (in March 11th-March 19th 2014).
Drought stress treatment
Strawberry plants grew to the six to seven leaf stage, plants with identical growth were randomly block designed to four replicates (10 plants per block), and drought stress was imposed (Bordonaba and Terry, 2010) by controlling irrigation (on May 12th 2014). The source of the irrigation water was state-supplied water and sub-irrigation was used to supply water to soil. The soil water-saturation content was determined by measuring the weight of the pots; these values were used to control the soil-moisture capacity according to the experimental requirements. The four sets were exposed to different soil-moisture treatments as follow: soil of the control group contained 70-85% of maximum water holding capacity; soil of the mild drought-stress group contained 50-60% of maximum water holding capacity; soil of the moderate drought-stress group contained 40-50% of maximum water holding capacity; and soil of the severe drought-stress group contained 30-40% of maximum water holding capacity. The pots were weighed at 18:30 every day, and the moisture lost during the day was replenished to maintain the specified soil-moisture content. A soil hygro-meter (ZYN-TSW1, Beijing Zhiyunda Science and Technology Co., Ltd., Beijing, China) was used to verify and maintain the target of soil-moisture contents. The plants were sampled and physiological characteristics were measured on 0, 2, 4, 6, 8, and 10 days after the commencing of drought stress. Leaves were randomly chosen for sampling at the specified time points. Five replicate samples were processed simul-taneously for measuring each experimental parameter during the drought-stress treatment. The average value of the five replicated samples was used for further analysis.
Determination of water potential of leaves
The leaves water potential was determined with a pressure chamber (Model 3005, Soil Moisture Equipment Co., Santa Barbara, CA, USA). The leaves were inserted into the pressure chamber in such a way that 3-5 cm of the petiole remains outside and slowly air pressure was increased with nitrogen gas flow. Pressure at which xylem sap flow was initiated at the cut end was noted as water potential of leaves samples.
Determination of free proline content
Proline content was measured using the method of Claussen. (2005). Sample of 0.2 g leaves was placed into a glass vial and 5 mL of aqueous solution of 3% sulfonic acid solution was added. The vials were closed and placed in boiling water with Digital thermostat water bath (TZL-5006, Experimental Equipment Co., Ltd., Suzhou Percival, Suzhou, China) for 30 min, and cooled to room temperature. Then, 2 mL ninhydrin and 2 mL glacial acetic acid were added to 2 mL of the supernatant extract; the vial with the reaction mixture was kept in a boiling water bath for 30 min and then cooled. After cooling, each vial was added to 5 mL of toluene and shaken vigorously. The reactions were carried out in the dark for 5 h to allow chromophore formation. The chromophore was placed in a quartz cuvette, and the extraction was determined with UV-Visible spectrophotometer at 520 nm (Model 754, Shanghai Third Analytical Instrument Factory, Shanghai, China). A standard calibration curve was used to estimate the proline content and expressed as mg・g-1 FW.
Determination of total sugar content
Samples of 0.2 g leaf was ground a mortar with liquid nitrogen and pestle after the addition of 10 mL of phosphate buffer (200 mmol・L-1, pH 7.0). 10,000×g was used to centrifuge the homogenate for 30 min at 4°C. The sugar concentration of the supernatant was determined, which was estimated using the method of Luo and Huang (2011) and slightly modified. The supernatant was serially diluted 10 to 50-fold with phosphate buffer. Then, 5 mL anthracene- H2SO4 and 1.8 mL distilled H2O were added to 0.2 mL of each serial dilution and mixed. The mixture was heated in boiling water for 10 min, and then cooled to room temperature. The content of soluble sugars was measured by determining absorbance using spectrophotometer at 620 nm (Model 754, Shanghai Third Analytical Instrument Factory, Shanghai, China). Glucose was used to prepare the standard curve. The express of results was as mg soluble sugar/g FW.
Determination of lipid peroxidation
Lipid peroxidation was estimated by measuring the malon-dialdehyde (MDA) concentration according to the method of Wang and Jin (2005). 0.5 g fresh leaf was homogenized with 5 mL 20% trichloroacetic acid (TCA). The homogenate was centrifuged at 10000×g for 5 min. 1 mL supernatant was mixed after the addition of equal volume of 0.6% (w/v) thiobarbituric acid solution containing 10% trichloroacetic acid. The mixture was reacted for 30 minutes in a boiling water bath and rapidly cooled with an ice bath. The absor-bance of the mixture was measured at 450, 532, and 600 nm. The calculation of MDA concentration used the Formula: MDA(μmol・g-1FW)=6.45(A532-A600)-0.56 A450.
Determination of antioxidant enzyme activities
The activity of SOD was measured using the method of photochemical nitro blue-tetrazolium (NBT) according to Yin et al. (2009). Inhibition of NBT reduction per min per mg leaf tissue (FW) by 50% was expressed as one unit of the SOD activity. The activity of POD was estimated by the change of absorbance at 470 nm produced by the oxidation of guaiacol (Chance and Maehly, 1995). The quantity required to degrade 0.01 mol of guaiacol per min per mg leaf tissue (FW) was expressed as one unit of POD activity. The activity of CAT was measured by the consumption of H2O2 at 240 nm using the method of Aebi (1984). One unit of CAT activity was expressed as the degradation of 0.1 mol of H2O2 per min per mg leaf tissue (FW).
Statistical analysis
The SPSS 13.0 statistical software package (standard released version 13.0 for Windows; SPSS Inc., Chicago, IL, USA) was used for statistical analyses. Analysis of variance was used to evaluate the significance of the experiment. Significantly difference of plants treated with different drought levels was analyzed by Duncan’s multiple range test (p < 0.05).
Results
Leaf water potential
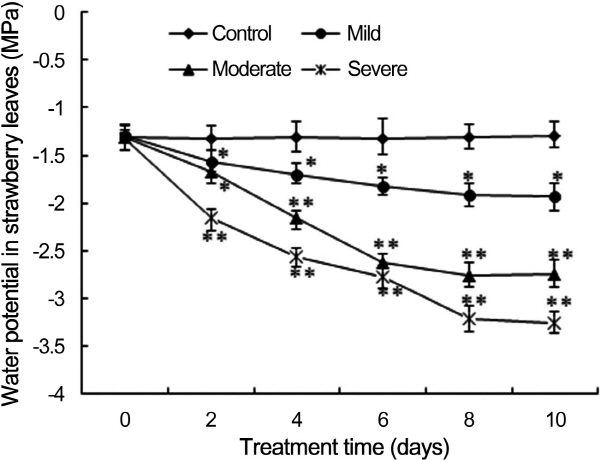
Water potential of strawberry leaves presented in Figure 1 is a good reference index of plant water stress, which maintained a constant in the control plants during the period of study. The leaves water potential decreased from the mild, moderate, and severe stress plants during the course of the water stress treatment and the difference was significant with the control. The leaf water potential of mild, moderate, and severe stress reached -1.93, -2.75, -3.25 MPa at 10 days after stressing treatment, respectively.
|
Fig. 1. Effects of drought stress on water potential in strawberry leaves (*: p < 0.05, **: p < 0.01). |
Free proline content
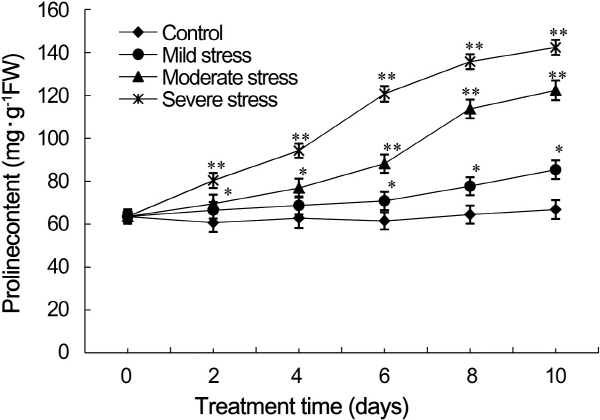
The accumulation of osmoprotectants in strawberry leaves induced by drought stress (Fig. 2). Free proline levels were higher in leaves subjected to drought stress than that of control leaves. The proline elevated levels as severity and duration of drought stress increased. Proline levels were significantly different between leaves subjected to drought stress and control except mild stress on day two. On day four, the proline content of leaves subjected to severe drought stress increased dramatically to 1.5-fold higher than that of control leaves (p < 0.05). On day six, the proline content of leaves subjected to moderate drought stress increased dramatically to 1.4-fold higher than that of control leaves (p < 0.05), and the content of leaves under severe drought stress increased to 1.9-fold higher than that of control leaves (p < 0.01). On day eight, the accumulation of proline in leaves under mild drought stress was not significantly different from that of control leaves, but accumulation of leaves under moderate and severe drought stress and controls were significantly different (1.8- and 2.1-fold higher than control respectively, p < 0.01). On day ten, a significant difference of proline accumulation in leaves under all the drought stress levels with that of control leaves appeared (p < 0.05). The proline contents of leaves subjected to mild, moderate, and severe drought stress were 1.3-, 1.9-, and 2.3-fold higher than that of control leaves, respectively.
|
Fig. 2. Effects of drought stress on proline levels in strawberry leaves (*: p < 0.05, **: p < 0.01). |
Sugar content
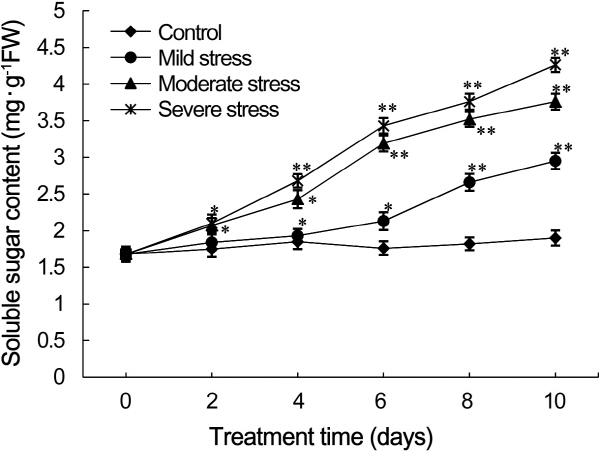
To further identify osmotic regulatory pathways to drought stress, we measured the soluble sugar content of strawberry leaves subjected to drought stress. The accumulation of soluble sugars in strawberry leaves induced by drought stress (Fig. 3), and the levels of soluble sugars increased as the severity and duration of drought stress increased. On day two, the contents of soluble sugar between drought- stress treated leaves and control leaves had no significant differences. On day four, the difference of sugar level in leaves subjected to mild drought stress was not significant than that of control leaves; however, the soluble sugar content of leaves under moderate and severe drought stress significantly increased to 1.5- and 1.6-fold higher than that of control leaves, respectively. On day six to ten, the sugar content of leaves subjected to moderate and severe drought stress increased sharply compared with that of control leaves, whereas they increased gradually in leaves under mild drought stress. On day ten, the soluble sugar content of leaves treated with mild, moderate, and severe drought stress were 1.6-, 2.2-, and 2.5-fold higher than that of control leaves (p < 0.01), respectively.
Malondialdehyde (MDA) content
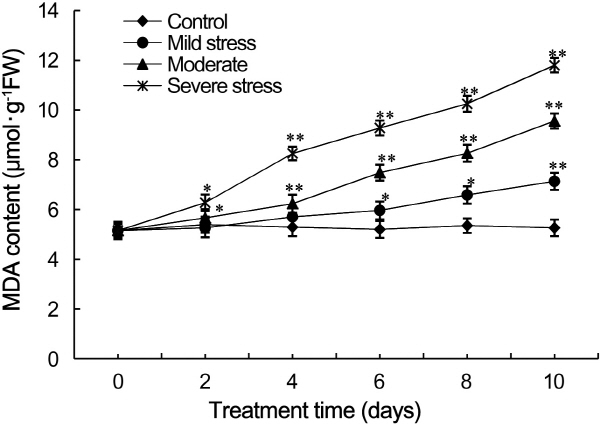
The MDA content of strawberry leaves subjected to drought stress increased as the severity and duration of drought stress increased (Fig. 4). On day two, except MDA content of the drought-treated and control leaves were not signifi-cantly different,the other differences are obvious. The content of MDA in leaves subjected to severe drought stress was 1.6-fold higher than that of control leaves on day four (p < 0.01), and increased to 1.8- and 2.0-fold higher than that of control leaves on day six and eight, respectively (p < 0.01). On day six, the MDA content of leaves under moderate drought stress was significantly different from that of control leaves. On day ten, the MDA content of leaves under moderate and severe drought stress was the 1.9- and 2.3-fold higher than that of control leaves, respectively.
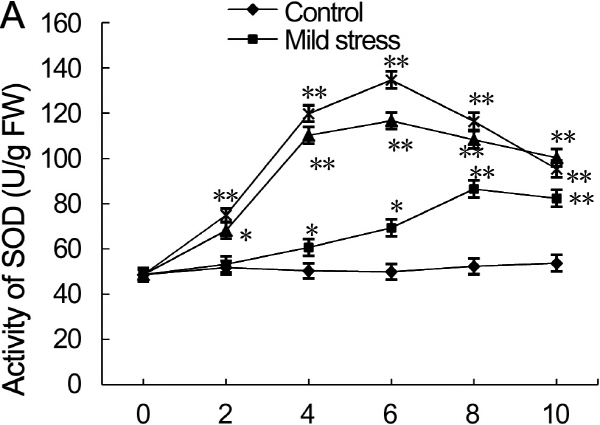
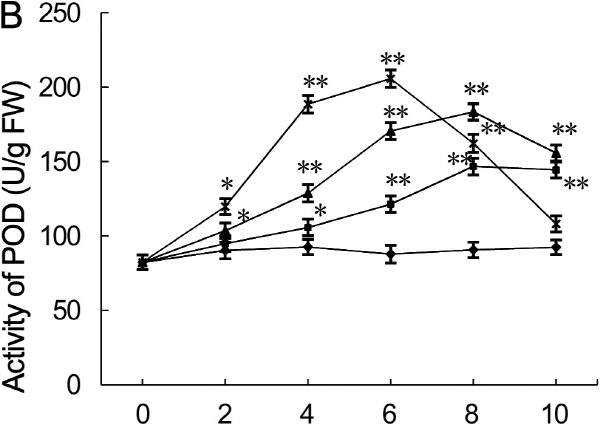
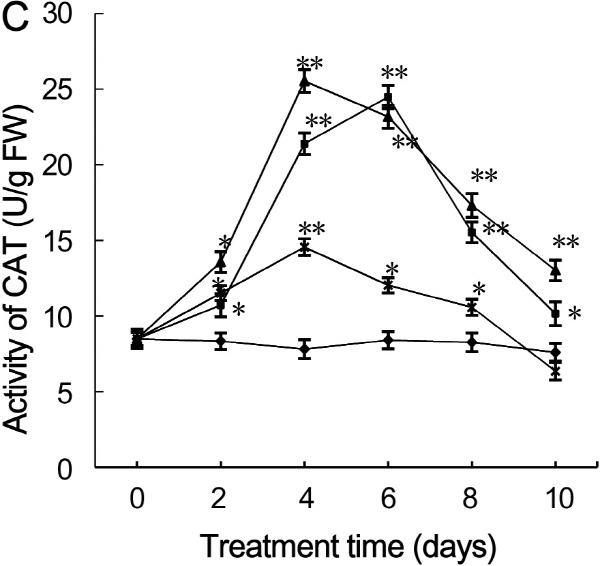
Antioxidant enzyme activity
SOD activity displayed dynamic responses to drought stress. SOD activity in leaves subjected to drought stress first increased on day four, reached maximum on day six, and then decreased during day eight to ten (Fig. 5A). On day two, SOD activity in leaves subjected to drought stress and control leaves was not significantly different. SOD activity increased rapidly in drought-stress treated leaves during day four to six, and reached maximum value and was 2.4- and 2.8-fold higher for moderate and severe drought stress than that in control leaves (p < 0.01), respectively. On day eight to ten, SOD activity in leaves under severe drought stress decreased rapidly. SOD activity increased gradually in leaves under mild drought stress, and reached maximum value and was 1.7-fold higher than that of control on day eight (p < 0.05).
POD activity displayed dynamic responses to drought stress. POD activity in the leaves under severe drought stress first increased on day two, reached maximum value on day six, and then decreased during day eight to ten (Fig. 5B). On day two, the difference of POD activity in leaves subjected to mild and moderate drought stress was not significant than the activity of control leaves; but POD activity of leaves subjected to severe drought stress had 1.3-fold higher than the activity of control leaves (p < 0.05). POD activity in leaves subjected to moderate drought stress was 1.6-fold higher than the activity of control leaves (p < 0.05) on day four, and POD activity in leaves subjected to severe drought stress was 2.8-fold higher than that of control leaves (p < 0.01). On day six, POD activity in leaves subjected to mild, moderate, and severe drought stress was 1.5-, 2.1-, and 2.5-fold higher than the activity of control leaves, respectively. POD activities in leaves subjected to mild and moderate drought stress reached maximum value and was 1.8- and 2.2-fold higher than the activity of control leaves on day eight (p < 0.01), respectively and declined thereafter. Whereas, POD activities of leaves subjected to severe drought stress reached maximum value and was 2.3-fold higher than that of control leaves on day six and declined thereafter.
CAT activity displayed dynamic responses to drought stress. CAT activity in leaves under severe drought stress first increased on day two, reached maximum value on day four, and sharply declined from day six to ten (Fig. 5C). On day two, CAT activity in leaves under mild, moderate, and severe drought stress increased significantly and was 1.3-, 1.6-, and 1.4-fold higher than that of control leaves, respectively. On day four, CAT activity in leaves subjected to moderate and severe drought stress reached maximum value and was 3.0- and 1.7-fold higher than that of control leaves (p < 0.01), respectively. Thereafter, CAT activity in leaves subjected to severe and moderate drought stress declined sharply; CAT activity in leaves treated with severe stress and was 0.8-fold lower than the activity of control leaves on day ten. On day six, CAT activity in leaves subjected to mild drought stress reached maximum value and was 2.9-fold higher than that of control leaves (p < 0.01); there-after, CAT activity sharply declined.
Discussion
Water potential is a direct index to reflect the plant water status or water deficit and can be used to estimate the drought-resistant ability of plants under drought stress (Na et al., 2014). The study found that the water potential of strawberries leaves decreased with drought stress intensity increased, which is consistent with previous research (Yang et al., 2006). The results indicate that strawberry has a certain ability to adapt to drought by reducing cellular water potential under water stress, improving their absorption of moisture in the dry environment in soil, thereby improving drought resistance.
Osmotic adjustment is an important physiological adap-ta-tion of plants associated with drought resistance. The intra-cellular proline can be used as osmotic substance with high compatibility with enzymes and other cellular macro-molecules, thus to protect them from damage induced by drought stress (Hare et al., 1998). Osmotic adjustment produced by proline causes a drop of the osmotic potential in plant tissues (Nanjo et al., 1999). A very important role of proline in response to osmotic stress may promote the biosynthesis of cell wall matrix proteins, such as extensin, which has an important role in maintaining cell morphology and provides a mechanical support of plant cells subjected to stress conditions (Nanjo et al., 1999). The current study observed higher levels of proline accumulation in strawberry leaves under drought stress from two to ten days, and the proline levels increased as the severity and duration of drought stress increased. Therefore, these results indicate that proline accumulation can be correlated with drought stress in strawberry, and may function as an adaptive response that mediates drought tolerance. The sugar accumulation in plant tissues have been found in response to water deficit, and was considered to have an important role in osmotic adjustment (Hare et al., 1998; Martinez et al., 2004). Bacelar et al. (2006) reported that the sugars in three olive cultivars increased subjected to water deficit. The concentration of soluble sugars of strawberry leaves subjected to drought stress increased as the severity and duration of drought stress increased in this study. The results suggest that soluble sugars are important osmotically active solutes that mediate osmoregulation and reduce water-deficit damage.
The definition of lipid peroxidation is the oxidation deg-radation of polyunsaturated lipids containing two or more carbon - carbon double bond (Mishra and Singha, 1992). MDA is often used as suitable biomarkers of membrane peroxidation damage (Baily et al., 1996). The increase of MDA content olive plants subjected to water stress also found, and was accompanied by a corresponding increase of lipoxygenase activity (Sofo et al., 2004). The intensity of water stress and the rate of MDA formation with the intensity of water deficit regulate the peroxidation process. Petridis et al. (2012) reported that lipid peroxidation of olive tree increased subjected to drought stress as the duration of drought stress. Remorini et al. (2009) reported that content of MDA also respond to salt stress and sunlight irradiance. In the current research, the content of MDA in strawberry leaves increased as the severity and duration of drought stress increased. These results indicate that membrane lipid peroxidation of the strawberry leaves caused by drought stress.
Increasing ROS levels of plant subjected to water stress has been shown. Antioxidant enzymes, including CAT, POD and SOD play a major role in detoxification of ROS (Pourtaghi et al., 2011). Plants alter the activities of antioxidant enzymes when subjected to stress. A recent study reported that one of the adaptive mechanisms in drought-tolerant crops subjected to drought-stress was up-regulating the activities of SOD and CAT (Devi et al., 2012). Drought-tolerant crops such as common bean, sunflower, and sorghum subjected to drought-stress conditions often have higher activity of POD than sensitive plants (Shigeoka et al., 2002). The results of the study showed that the activities of SOD, POD and CAT in strawberry leaves up-regulated with the increase of intensity and duration of drought stress. These results suggest that SOD, POD, and CAT in strawberry plants could mitigate the oxidative stress induced by drought. These antioxidant enzymes may have crucial roles in plant adap-tation to drought conditions. But SOD, POD and CAT activity began to decline six days after drought stress, indicating that strawberry adaptability to drought stress had a limit. In summary, this study showed that the strawberry plants subjected to drought stress could alter the activities of antioxidant enzyme and increase the levels of osmotically active solutes in strawberry leaves. Dynamic increase of SOD, CAT, and POD activity in strawberry leaves occurred in parallel with increases in free proline and soluble sugars. These results suggest that strawberry plants may use anti-oxidant enzymes and osmotically active solutes for adaptation responses to drought stress. Therefore, these systems would be excellent candidates for future biotechnology approaches to improve strawberry resistance to drought stress.