Introduction
Materials and Methods
Materials
Germination Capacity of Pear Pollen
Extraction of Protein
Determination of Peroxidase Activity
Determination of Dehydrogenase Activity
Activity Staining of Peroxidase
Assay of the Reducing Sugars on TLC
Results and Discussion
Introduction
The pollen grain, a male gametophyte, plays a vital role during sexual reproduction in plants (Boavida et al., 2005). After landing on a receptive stigma of the pistil, the pollen tube emerges from the pollen grain, grows through the transmitting tissue of the style, and delivers the male gametes to the embryo sac within the ovule, for double fertilization (Wilhelmi and Preuss, 1999).
The development of reliable methods to verify pollen quality might be useful for monitoring pollen performance during storage, assessing fertility, plant breeding, research on pollen-stigma interaction, and incompatibility studies (Heslop-Harrison et al., 1984). To assess pollen quality, indirect methods based on colorimetric and enzymatic tests have been introduced. A variety of vital dyes, such as tetrazolium salts (dehydrogenase), aniline blue (callose in pollen wall), acetocarmine (nucleic acid), Alexander’s stain (cytoplasmic constituents), and fluorescein diacetate (esterase) are available (Oberle and Watson, 1953; Currier, 1957; Heslop-Harrison, 1992; Alexander, 1987; Heslop-Harrison et al., 1984). The colorimetric assays are often preferred, as these are simple and fast. However, different results might be obtained in some fruit species and cultivars (Oberle and Watson, 1953). Furthermore, they tend to overestimate the viability, due to the presence of enzymes and other substances from non-viable pollen (Rodriguez-Riano and Dafni, 2000).
Temperature and humidity have been shown to affect the viability and germination of pollen. Studies have shown that pollen can be maintained for long periods at low temperature and humidity (Johri and Vasil, 1961; Kwan et al., 1969; Linskens, 1964).
Peroxidases, which catalyze reactions that involve not only H2O2 decomposition but also hydroxyl radical (∙OH) generation, have been known to play a key role in pollen viability and cell wall formation during pollen germination and pollen tube growth (Stanley and Linskens, 1974; Chen and Schopfer, 1999; Schopfer et al., 2001; Miura, 2012). Furthermore, they are involved in diverse physiological processes such as cell wall metabolism, lignification, suberization, reactive oxygen species (ROS) metabolism, auxin metabolism, fruit ripening, and defense against pathogen attack (Welinder, 1992; Passardi et al., 2004).
Several dehydrogenases as well as peroxidases, when coupled with indicator dyes, have been used to assay pollen viability. At least 15 dehydrogenases have been well-described in pollen grains (Stanley and Linskens, 1974). Dehydrogenase activity is higher in pollen compared to other tissues (Martin, 1968). Glucose 6-phosphate dehydrogenase and isocitrate dehydrogenase activities were reported in lily pollen (Dickinson and Davies, 1971). Glutamate dehydrogenase activity has been shown in petunia (Roggen, 1967), apple (Veidenberg and Safonov, 1968), and lily (Okunuki, 1939; Desborough, and Peloquin, 1968). Lactate dehydrogenase activity has been observed in aspen (Euler, 1949) and lily (Linskens, 1966).
Pollen germination and tube elongation are closely related to important biochemical factors, such as calcium gradient within the pollen tube tip, vesicle transport, cell wall biosynthesis, ion fluxes, and actin microfilaments (Hepler et al., 2012; Steinhorst and Kudla, 2013). Recently, RNA sequencing (RNA-Seq) technology has been used to characterize the expression of genes during four developmental stages: [mature pollen grains (MP), hydrated pollen grains (HP), growing pollen tubes (PT), and stopped-growth pollen tubes (SPT)] of pear pollen (Zhou et al., 2016). Also, low risk of pollen-mediated gene flow was reported in transgenic plants under greenhouse conditions (Kim et al., 2018).
The objectives of the present study were to (1) investigate the relationship between the germination capacity of pollen and germination-related enzymes, and (2) estimate the changes in enzyme activity and expression pattern of POD and dehydrogenase in heat- and moisture-treated pear pollen.
Materials and Methods
Materials
Pollen from three pear fruit cultivars, including ‘Xuehuali’ (Pyrus bretschneideri), ‘Pea pear’ (P. calleryana), and ‘Chuwhangbae’ (P. pyrifolia) cultivars were purchased from Well Plus (Dajeon, Korea) and stored in tightly sealed containers with desiccant at -20°C until use. The pollen of ‘Imamuraaki’ (P. pyrifolia) was obtained from a pear farm (Naju, Jeonnam, Korea) and kept at -20°C for several months.
Germination Capacity of Pear Pollen
Unless otherwise stated, stored pollen was rehydrated for 12 h at 4°C in a sealed container lined with moist filter paper, prior to germination in vitro. The pollen was scattered uniformly on a medium containing 10% sucrose and 1% agar, and then incubated at 25°C in the dark, with a relative humidity of 80-90%. To investigate the effect of duration of room temperature (RT, 20°C) exposure on pollen germination, pollen was collected at various time periods (6, 12, 18, and 24 h) of exposure to RT. Subsequently, the germination rate was measured every 1 h during incubation. For moisture treatment, pollen was placed in a sealed container lined with moist filter paper and stored at 4°C for 12 h. After pre-treatment, the moisture content was measured using a moisture analyzer with infrared heat source (MB23, OHAUS Co., USA).
To assess the optimal conditions for pollen storage, pollen was subjected to five storage conditions, including heat-treated frozen storage (HF), fresh frozen storage (FF), moisture-treated frozen storage (MF), moisture-treated cold storage (MC), and moisture-treated room temperature storage (MR). For HF preparation, pollen was heated, at a temperature of 105°C using a moisture analyzer with infrared heat source, for 5 min and then kept at -20°C for 24 h. For FF preparation, pollen obtained from several companies was kept at -20°C for several months and no treatment was applied until use. For MF, MC, and MR preparation, pollen was placed in a sealed container lined with moist filter paper and stored at 4°C for 12 h. Pollen was then subdivided into three groups and each group was kept at -20°C or 4°C or 20°C, respectively, for 24 h.
Pollen germination under each set of conditions was observed using a microscope. To elucidate the role of chitinase on pollen germination, different concentrations (0, 10, and 20 nmol) of N-acetyl glucosamine (NAcGlc) were added to the germination medium. The percentage germination was determined by scoring at least 25 pollen grains from five randomly selected microscopic fields. Pollen grains with tube length equal to or greater than the diameter were considered to have germinated.
Extraction of Protein
Two types of samples were prepared using refrigerated fresh, and heat-treated (100°C for 10 min) pollen. Ten milligrams of fresh and heat-treated pollen were spread on Petri dishes containing 5 mL of germination medium [10% (w/v) sucrose and 1% (w/v) agar], and then cultured at 25°C for 3 h.
Proteins were extracted from the pear pollen (10 mg) using the method described by Persia et al. (2008) with some modifications. Pollen proteins were obtained using lysis buffer 200 µL (50 mM Tris HCl, pH 8.5, 3 mM EGTA, 3 mM EDTA, and 2 mM MgCl2) for 2 min on ice using a grinding kit (BioMasher-II, NIPPI Inc., Tokyo, Japan). The total protein concentration in each sample was determined according to the method described by Bradford (1976), with bovine serum albumin (BSA) as the standard.
Determination of Peroxidase Activity
To investigate the correlation between POD activity and germination of ‘Xuehuali’ pollen, POD activity was measured in different proportions of viable (frozen storage) and non-viable (heat-treated) pollen grains. Furthermore, changes in POD activity were estimated in pollen grains stored under the various conditions mentioned above.
Peroxidase activity was assayed based on the method described by Chance and Maehly (1955). The reaction mixture contained 50 µL of 20 mM guaiacol, 2.87 mL of 10 mM phosphate buffer (pH 7.0), and 25 µL of enzyme extract. The reaction was started with the addition of 20 µL of 40 mM H2O2. The increase in absorbance at 470 nm was measured at 1 min intervals using a UV-visible spectrophotometer (Optizen 3220UV, Mecasys Co., Korea). One unit is defined as the amount of enzyme producing 1 µmol of tetraguaiacol per minute at 25°C.
Determination of Dehydrogenase Activity
The activity of dehydrogenase was measured according to the method described previously (Cook and Stanley, 1960; Casida, 1977). Samples of pear pollen (20 mg) were taken in Eppendorf tubes. One milliliter of 100 mM Tris-HCl (pH 7.8) buffer and 500 µL of 0.4% 2, 3, 5-triphenyltetrazolium chloride (TTC) solution were added to each sample. Subsequently, the tubes were incubated at 25°C for 30 min. After incubation, 5 mL of methanol was added, and the sample extracted immediately, with triphenyl formazan. The absorbance of the red methanolic solution of formazan was read at 485 nm using a spectrophotometer (µQuant, BioTec, USA).
Activity Staining of Peroxidase
Native polyacrylamide gel electrophoresis (PAGE) was performed according to the method described by Ornstein (1964). PAGE was performed using a Bio-Rad Mini-PROTEAN (80 mm × 73 mm × 1.5 mm). The 10% native PAGE gel was stained with 0.12% Coomassie brilliant blue R-250. For active staining of POD after 10% native PAGE, the gels (30 µg proteins) were soaked for 10 min in 50 mM Tris buffer (pH 6.8), incubated with 0.46% (v/v) guaiacol and 13 mM H2O2 in the same buffer until red bands appeared, and subsequently fixed in a mixture of water/methanol/acetic acid (6.5:2.5:1, v/v) (Caruso et al., 1999).
Assay of the Reducing Sugars on TLC
To investigate the reducing sugar content of pear pollen, heat-treated and fresh ‘Xuehuali’, ‘Imamuraaki’, ‘Chuwhangbae’, and ‘pea pear’ pollen was used. Pollen extract was spotted on a thin layer chromatography (TLC) plate. After developing the plate for 3 h in a development solvent of n-propanol/water/ethyl acetate/ammonia solution (6:3:3:1, v/v), the N-acetyl- chitooligosaccharides were visualized by the aniline-diphenylamine reaction (Tanaka et al., 1999).
Results and Discussion
We examined the germination rate of the ‘Xuehuali’ pollen grains which were exposed to RT condition for various periods. The germination rate was higher after 6 h of exposure when compared with those of the other exposure times. Conversely, germination patterns of pollen exposed to RT for 12, 18, and 24 h were similar (Fig. 1A).
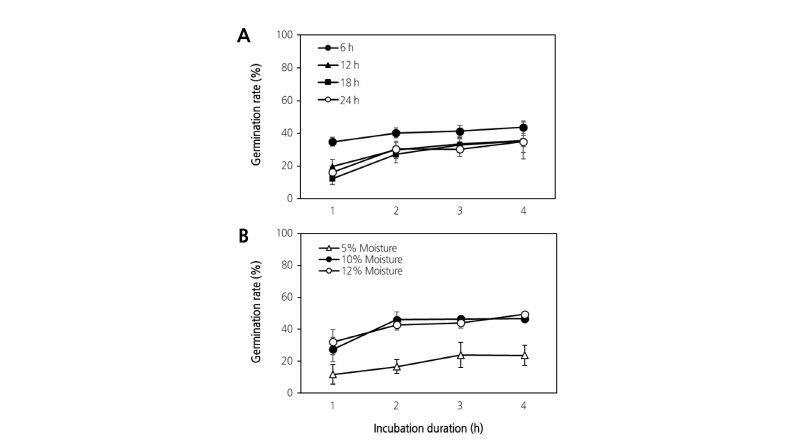
Fig. 1. Germination rate of pear pollen at various periods (6, 12, 18, and 24 h) of exposure to room temperature (RT) (A) and after moisture pre-treatment (5, 10, and 12%) for 12 h at 4°C (B). Data represent the mean value of three replicates, each performed in triplicate. Error bars indicate standard deviation (SD).
This suggests that the time after exposure to RT directly affects pollen performance, and the loss of germination occurs within 12 h of exposure, probably due to decreased activity of germination-related enzymes. This result may also be explained by impaired germination which results from the loss of pollen viability during over-exposure to RT.
To restore the vitality of pollen stored below 5% moisture content at -20°C for several months, the pollen was placed in a sealed container lined with moist filter paper and stored at 4°C for 12 h. After pre-treatment, the moisture content was measured using a moisture analyzer, the pollen with either 10% or 12% moisture content, respectively, were used in this experiment.
The effect of moisture pre-treatment on pollen germination is shown in Fig. 1B. The germination rates of moisture pre-treated (10% and 12%) and untreated pollen (5%) were observed to increase gradually during the incubation of 4 h. There was no significant difference between 10% and 12% of moisture pre-treatment on pollen germination. Overall germination rates in moisture pre-treated pollen (10% and 12% moisture content) were 20% higher than in untreated pollen (5% moisture content) over the incubation periods.
Moisture content and storage temperature are major factors in successful pollen germination. As suggested, moisture content is very important during pollen germination. In our study, we focused on differences in germination rate of pollen with an average moisture content (10-12%), at 1 h intervals during germination. Examining the differences in germination rate of pollen samples with various moisture contents may be worth further investigation.
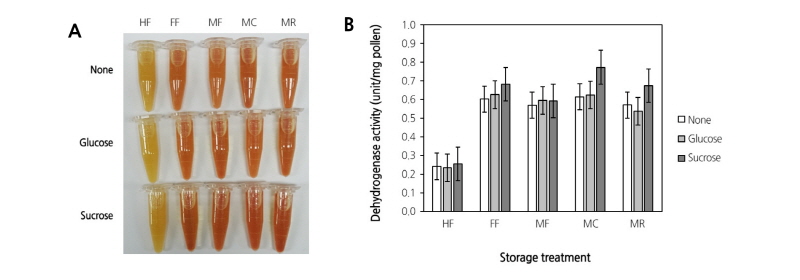
To assess the optimal storage conditions for pollen performance, various storage conditions described earlier were tested. In Fig. 2, a germination rate of 55.9% was observed in the MC treatment, whereas little or no germination (1.2%) was observed in the HF treatment. Furthermore, the germination rates of moisture pre-treatment groups (MF, MC, and MR) were higher than that of the FF treatment (21.9%), in particular that of the MC treatment, which exhibited up to a 2.5-fold increase. These results suggest a positive effect of moisture pre-treatment on pollen germination.
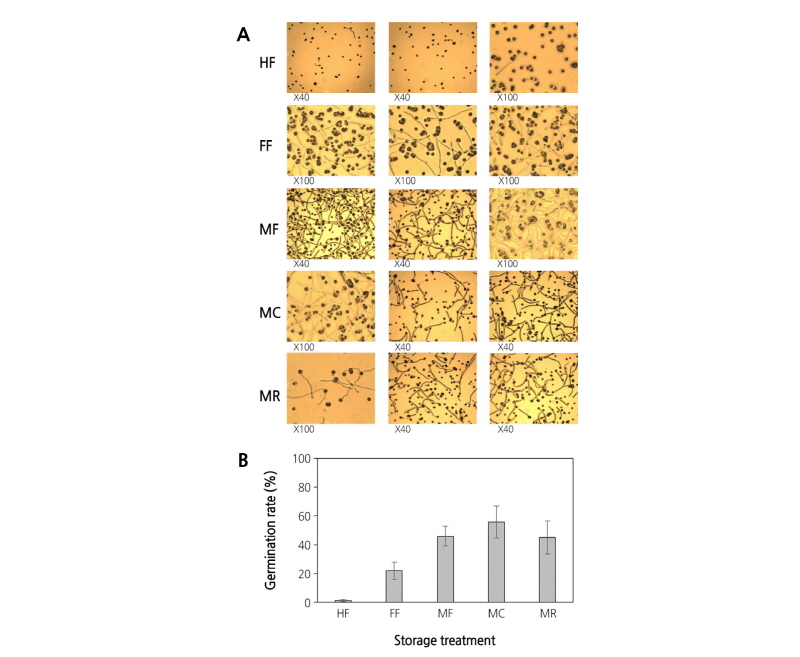
Fig. 2. Pollen germination under five storage conditions. The pollen was scattered uniformly on the medium containing 10% sucrose and 1% agar, and then incubated at 25°C in darkness for 3 h. Pollen germination was observed under a microscope (×40 and ×100). HF, heat-treated frozen storage; FF, fresh frozen storage; MF, moisture-treated frozen storage; MC, moisture-treated cold storage; MR, moisture-treated RT storage.
Germination tests of avocado pollen grains maintained at various relative humidity levels (RH), showed that the pollen retain their initial germination rate at RHs of 55% or higher, whereas the pollen completely lose their ability to germinate at a RH of 5% within 2 h (Loupassaki et al., 1997). Furthermore, there was no loss of germination for 24 h when the pollen was stored at a RH of 100%. This suggests the acute effects of moisture pre-treatment on the in vitro germination of avocado pollen. The positive effect of moisture pre-treatment on the germination of pollen has also been reported in papaya (Cohen et al., 1989), walnut (Luza and Polito, 1987), and aglaonema (Henny, 1980).
To investigate whether the activity of POD is essential for the germination of pollen, the correlation between POD activity and germination was studied in the pollen of ‘Xuehuali’. The activity of POD was measured in different proportions of viable and non-viable pollen grains. There was no linear relationship between pollen germination and POD activity (R2 = 0.3184) (Fig. 3A). The activity of POD in pear pollen revealed no significant difference among the five storage conditions tested (Fig. 3B). In the HF group, the activity of POD was observed; however, the pollen grains were considered non-viable. This might be partially attributed to the over estimation caused by the presence of enzymes in non-viable pollen. The comparison of POD activity in various pear cultivars also showed no significant difference, as expected (Fig. 3C). This suggests that the activity of POD is not required for, or associated with, the germination of pear pollen. Due to their ubiquitous distribution in pollen grains, it has been proposed that PODs are crucial to the determination of pollen viability. However, pollen has relatively low POD activity when compared with that of other tissues (Linskens, 1966; Bredemeijer, 1979; Bredemeijer, 1984).
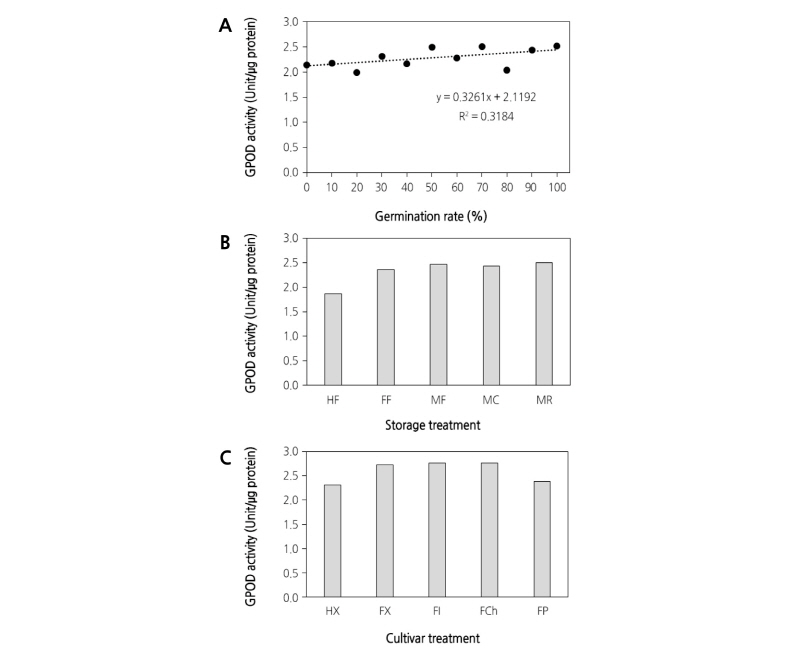
Fig. 3. Correlation between peroxidase (POD) activity and germination in pear pollen. The activity of POD was determined in different proportions of viable and non-viable pollen (A). The change in POD activity under various storage conditions (B) as described in materials and methods and in various pear cultivars (C). HF, heat-treated frozen storage; FF, fresh frozen storage; MF, moisture-treated frozen storage; MC, moisture-treated cold storage; MR, moisture-treated RT storage; HX, Heat ‘Xuehuali’; FX, Fresh ‘Xuehuali’; FI, Fresh ‘Imamuraaki’; FCh, Fresh ‘Chuwhangbae’; FP, Fresh ‘Pea pear’.
The expression of POD was examined in the crude extract of pollen from various pear cultivars (Fig. 4). Two isoform bands specific for POD migrating at approximately 66 kDa were detected among the tested pear cultivars as well as the heat-treated group (Fig. 4B). These results indicate that monitoring of POD activity is not suitable to verify the performance of pear pollen.
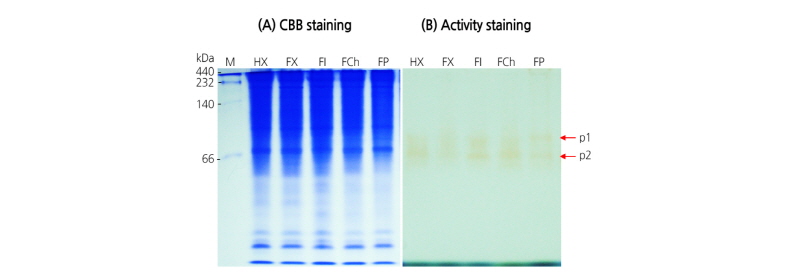
Fig. 4. Expression pattern of peroxidase (POD) in crude extract of pollen from various pear cultivars. Protein bands were visualized by Coomassie brilliant blue R-250 (A) or active staining (B). HX, Heat ‘Xuehuali’; FX, Fresh ‘Xuehuali’; FI, Fresh ‘Imamuraaki’; FCh, Fresh ‘Chuwhangbae’; FP, Fresh ‘Pea pear’; M, size marker. Arrows indicate two POD isoforms.
In the present study, to verify the role of dehydrogenase as an indicator of pollen performance, the activity of dehydrogenase was measured in pear pollen that was stored under various conditions. The results revealed that the activity of dehydrogenase in the MC treatment was at least 3 fold higher than that in the HF treatment (Fig. 5B). Except for the HF treatment, only marginal differences in the enzyme activity were observed among the tested conditions (FF, MF, MC, and MR). Furthermore, enzyme activity was enhanced marginally when either glucose or sucrose was added. The results also showed that sucrose was more effective than glucose in enhancing the activity of dehydrogenase (Fig. 5B). The pollen with germination potential can be discriminated from non-viable pollen by monitoring the activity of dehydrogenase.
The role of dehydrogenases in pollen tube growth has been reported. Pollen tube growth is essential for successful sexual reproduction and high energy-demanding processes. Pollen grains in maize have been shown to contain 20 times more mitochondria than other vegetative tissues (Lee and Warmke, 1979). During oxygen limitation in higher plants, energy metabolism switches from respiration to fermentation. The ethanolic fermentation pathway consists of two key enzymes, pyruvate decarboxylase (PDC) and alcohol dehydrogenase (ADH). This pathway serves as a pyruvate dehydrogenase (PDH) bypass, to maintain energy supply during oxygen limitation in pollen tubes (Rounds et al., 2011). The possible involvement of aldehyde dehydrogenase (ALDH) in the germination of pollen has been reported previously. According to Op den Camp and Kuhlemeier (1997), the ALDH gene was not induced under anoxia; however, it was highly expressed in tobacco pollen, and inhibition of ALDH prevents pollen growth. ALDH is important for pollen tube growth, and might be involved in PDH bypass, thus contributing to the satisfaction of energy demands during pollen tube growth.
Sorbitol dehydrogenase (SDH, EC 1.1.1.14) is an important enzyme in the metabolism of sorbitol in higher plants (Loescher, 1987). It catalyzes the oxidation of sorbitol to fructose. Chinese white pear (Pyrus bretschneideri) contains 15 SDH gene members (Dai et al., 2015).
In the present study, dehydrogenase activity was higher in the pollen of MC and MR supplemented with sucrose, when compared with that in pollen under other tested conditions (Fig. 5B). Dehydrogenase activity exhibited potential as a germination-related indicator of pear pollen (‘Xuehuali’).
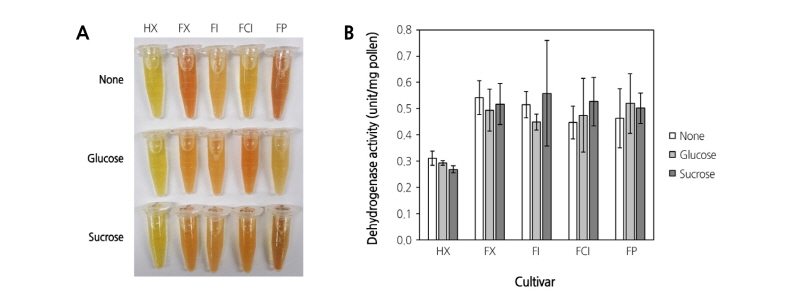
The evaluation of dehydrogenase activity is enough to discriminate between viable and non-viable pollen grains (Fig. 5B). The present study also investigated the difference in dehydrogenase activity among different pear cultivars. There were no obvious differences among the pear cultivars (Fig. 6B).
Differences in the content of reducing sugars which were extracted either by distilled water or ethanol were observed from fresh and heat-treated pollen (Fig. 7A). The result shows that the reducing sugar content in both the fresh and heat-treated pollen was higher when extracted with distilled water than with ethanol. The resulting reducing sugars extracted from different cultivars of both fresh and heat-treated pollen were determined by TLC (Fig. 7B). The production of glucose was observed in four different types of pollen with and without heat-treatment.
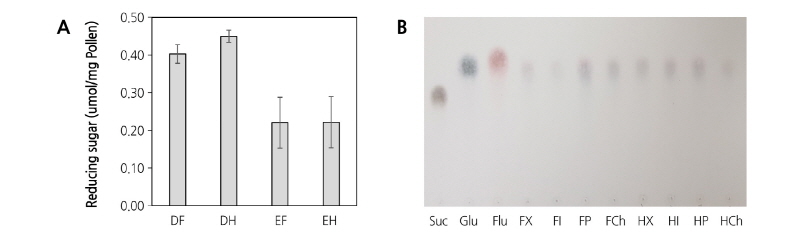
Fig. 7. Reducing sugar content in pear pollen ‘Xuehuali’ extracted with distilled water (DW) and ethanol (A) and the expression of reducing sugar on the TLC plate (B). DF, DW extraction Fresh pollen; DH, DW extraction Heat pollen; EF, 80% Ethanol extraction Fresh pollen; EH, 80% Ethanol extraction Heat pollen; FX, Fresh ‘Xuehuali’; FI, Fresh ‘Imamuraaki’; FP, Fresh Pea; FCh, Fresh ‘Chuwhangbae’; HX, Heat-treated ‘Xuehuali’; HI, Heat-treated ‘Imamuraaki’; HP, Heat-treated ‘Pea pear’; HCh, Heat-treated ‘Chuwhangbae’.
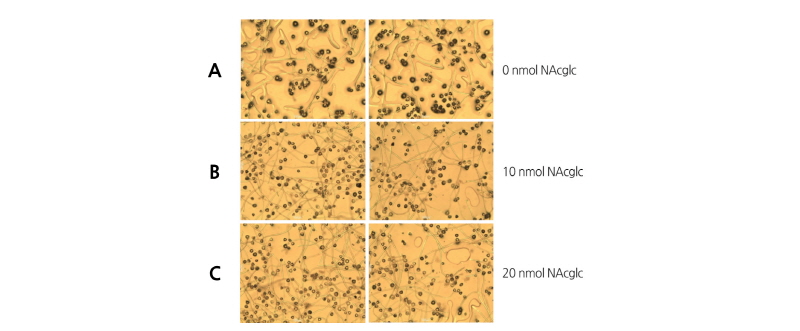
Pollen grains of ‘Xuehuali’ exhibited a germination rate above 38% in all the treatments with N-acetyl glucosamine (NAcGlc) on sucrose agar plates (Fig. 8). Pear pollen grains showed a germination rate of 38.0% in the control, 40.4% in 10 nmol NAcGlc, and 40.1% in 20 nmol NAcGlc.
In conclusion, the duration of RT exposure negatively affects pollen germination, while the moisture pre-treatment impacts positively on pollen germination. The activity of POD is not required or associated with the germination of pear pollen. The results of the present study demonstrated that dehydrogenase is a potential indicator of pollen performance in pear. Thus, the viable pollen can be discriminated from non-viable pollen by monitoring the activity of dehydrogenase.