Introduction
Materials and Methods
Plant materials
Stomatal characteristics
Statistical analysis
Results
Stomatal shape and characteristics
Interspecific grouping based on PCA and cluster analysis
Discussion
Introduction
The genus Hosta (Asparagaceae, APG IV) comprises herbaceous perennials native to Northeast Asia, including the Korean Peninsula (Lee et al. 2018). It is widely appreciated for its exceptional shade tolerance, cold-hardiness, and ornamental foliage (Jones 1989; Armitage 2020; Yang and Kim 2025). Despite the global popularity of Hosta cultivars –7,064 registered with the American Hosta Society (AHS 2025) – relatively few breeding attempts have been made in South Korea, which is the center of origin for several wild Hosta species (Ko et al. 2018). Understanding the environmental adaptability of these native taxa is essential for their utilization in horticulture and for the development of novel cultivars suited to future climatic conditions (Yang et al. 2022).
According to Plants of the World Online (Kew 2025a), 30 Hosta species are distributed across Southeastern Russia, China, Japan, and South Korea. Six species are native to Korea – H. capitata (Irwol-bi-bi-chu in Korea), H. clausa (Ju-qeok-bi-bi-chu), H. jonesii (Dadohae-bi-bi-chu), H. minor (Jom-bi-bi-chu), H. venusta (Halla-bi-bi-chu), and H. yingeri (Heuksando-bi-bi-chu) – of which H. jonesii, H. minor, H. venusta, and H. yingeri are endemic (Chung et al. 2017; Lee et al. 2018). These species exhibit distinct differences with regard to plant size, leaf shape, floral traits, and ecological niches, reflecting a wide range of adaptive strategies (Lee et al. 2018). They are distributed in various habitats (Table 1). This ecological and morphological diversity represents valuable genetic resources for landscape and breeding purposes (Kim and Kim 2024).
Table 1.
Natural habitats of six Korean Hosta species, as reported by Chung and Kim (1991), Jo and Kim (2017), and Lee et al. (2018)
Among the various adaptive traits, stomatal characteristics influence plant responses to environmental stressors, particularly under changing climatic conditions. Stomatal shape, size, and density are key determinants of gas exchange efficiency and water use regulation. They may reflect species-specific ecological strategies. Although a study focusing on the stomatal traits of Hosta species inhabiting western Siberia has been conducted, no research has been carried out on the Hosta species native to Korea (Sedelnikova 2021). Therefore, we hypothesize that the stomatal traits vary significantly among the six Korean Hosta species. The aim of this study was to characterize the interspecific variation in stomatal traits among the six Korean Hosta species. These findings can provide fundamental insights into their morphological differentiation and ecological adaptability, contributing baseline data for both conservation and horticultural utilization.
Materials and Methods
Plant materials
Seedlings of H. capitata, H. clausa, H. jonesii, H. minor, H. venusta, and H. yingeri were obtained from the Korea National Arboretum in May of 2020 and cultivated under greenhouse conditions at the University of Seoul (UOS) until November of 2022. To meet their natural dormancy requirements, the plants were then exposed to ambient cold conditions outdoors for approximately two months. In January of 2023, they were returned to the greenhouse and transplanted into rectangular plastic pots (11.5 × 11.5 × 10 cm) filled with a 1:1 (v/v) mixture of perlite (Kebifarm, Korea) and a commercial potting medium (Baroker, Seoul Bio, Korea). Each pot contained a single seedling, and commercial garden soil was added to ensure adequate nutrient availability.
In April of 2023, stomatal traits were investigated using fully expanded leaves of healthy, mature plants. During this period, the average environmental conditions were as follows: the air temperature was 18.4 ± 4.1°C, relative humidity was 50.6 ± 21.2%, and the mean daily light integral (DLI) was 6.7 mol·m-2·day-1 under natural photoperiod conditions. Irrigation and fertilization followed general greenhouse practices for potted plants, including daily watering and solid fertilizer application to prevent drying and leaf yellowing.
Stomatal characteristics
The stomatal characteristics were analyzed using field-emission scanning electron microscopy (FE-SEM) (SU8010, Hitachi High-Tech, Tokyo, Japan). Fully expanded leaves were sampled from each species at 10:00 a.m. on April 17, 2023. From each species, two individuals were sampled, and one leaf was collected from each individual. Leaf sections approximately 1 cm in width were excised vertically, with the midrib as a central reference. Each sample was mounted on a metal stub with the abaxial (lower) surface facing upward and was secured using carbon adhesive tape to ensure flat positioning for imaging. Mounted samples were dehydrated using a freeze-dryer (FD 8505, IlShinBioBase Co. Ltd., Dongducheon, Korea). After being coated with platinum, the specimens were examined using FE-SEM at the UOS Center for Research Facilities. Stomatal traits including the density, guard cell length, guard cell width, and morphology were assessed at an accelerating voltage of 10 kV. Images were captured at 100×, 400×, and 2000× magnifications. To access the stomatal size, five stomata were randomly examined per leaf, resulting in a total of ten stomata measured per species at 400× magnification. Stomatal density (number per mm2) was calculated by examining ten points randomly chosen per species, consisting of five points of each leaf at 100× magnification.
Statistical analysis
All of the statistical analyses were performed using SPSS Statistics 27 for Windows (IBM Corp., Armonk, NY, USA). A one-way analysis of variance (ANOVA) followed by Duncan’s new multiple range test (p < 0.05) was used to compare the mean differences in stomatal traits among the species. The ANOVA was conducted under the statistical assumptions of normality, homogeneity of variance, and independence. Pearson’s correlation analysis was conducted to evaluate the relationships among the stomatal parameters. The correlation analysis was conducted under the assumptions of normality, linearity, independence, and absence of outliers. Principal component analysis (PCA) and hierarchical cluster analysis (HCA) were used to identify variation patterns and classify species based on their stomatal characteristics. Univariate descriptive statistics initially were conducted to examine the distributions of the variables. The suitability of the dataset for a factor analysis was assessed using the Kaiser–Meyer–Olkin (KMO) measure of sampling adequacy and Bartlett’s test of sphericity based on the correlation matrix. Factor extraction was performed from the covariance matrix, with the number of factors fixed at two. Orthogonal varimax rotation was applied to enhance the interpretability of the factor loadings. Factor scores were estimated using Bartlett’s method.
Results
Stomatal shape and characteristics
All six Hosta species exhibited anomocytic stomatal complexes (Table 2 and Fig. 1). The stomata were oval in shape and located at the junctions between the epidermal cells, with a generally uniform orientation. Although the stomatal morphology was consistent across species, significant interspecific variation in the stomatal size and density was observed (Table 2).
Table 2.
Variation in stomatal density and size among the six Korean Hosta species
| Species | Stomata type | Stomatal size |
Stomatal density (stomata per mm2) | ||
|
Guard cell length (µm) |
Guard cell width (µm) |
L × Wz (µm2) | |||
| H. capitata | Anomocytic | 27.3 ± 2.05 bcy | 17.2 ± 1.99 b | 471.4 ± 69.95 b | 43.0 ± 1.86 d |
| H. clausa | Anomocytic | 25.7 ± 2.48 c | 11.6 ± 2.28 d | 300.0 ± 70.74 c | 111.0 ± 6.24 a |
| H. jonesii | Anomocytic | 28.8 ± 2.81 b | 14.8 ± 2.51 c | 429.2 ± 109.72 b | 44.9 ± 5.13 d |
| H. minor | Anomocytic | 29.3 ± 3.26 b | 19.5 ± 1.72 a | 569.9 ± 62.07 a | 54.8 ± 4.01 c |
| H. venusta | Anomocytic | 26.4 ± 3.01 bc | 15.9 ± 2.62 bc | 423.5 ± 105.09 b | 73.2 ± 3.89 b |
| H. yingeri | Anomocytic | 32.1 ± 4.25 a | 19.4 ± 1.47 a | 624.9 ± 115.78 a | 41.1 ± 6.80 d |
| Significance | *** | *** | *** | *** | |
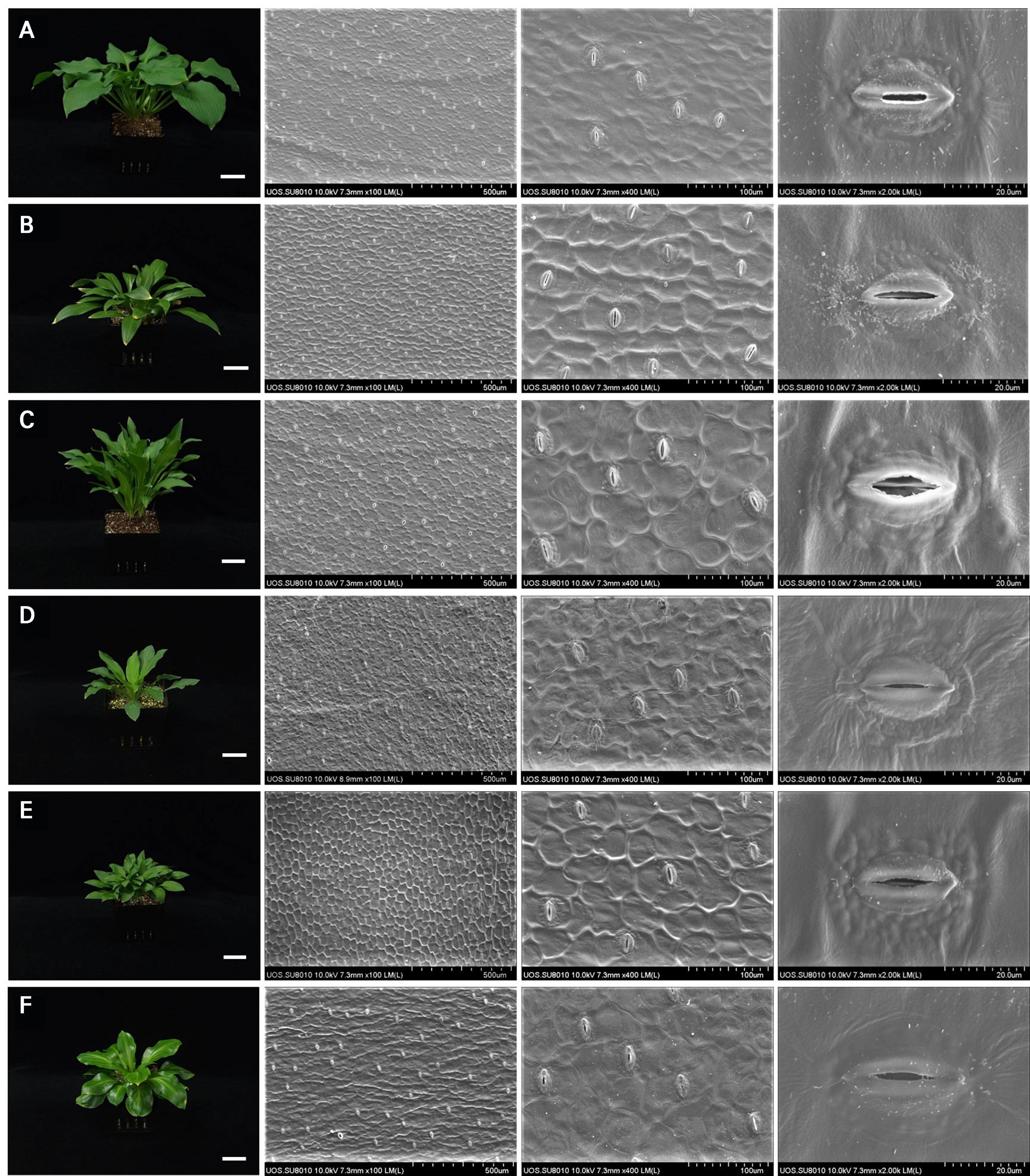
Fig. 1.
Inter-specific variation in the stomatal density and size among the six Korean Hosta species with different shoot shapes. A: H. capitata; B: H. clausa; C: H. jonesii; D: H. minor; E: H. venusta; F: H. yingeri. Stomatal images were captured from the abaxial leaf surface by means of field scanning electron microscopy (FE-SEM) at magnifications of 100× (scale bar = 50 mm), 400×, and 2000× in order, respectively.
Among the species, H. yingeri had the longest guard cells (32.1 ± 4.25 µm), which were found to be significantly longer (up to 1.2-fold) than those of the other five species. In contrast, H. clausa exhibited notably shorter guard cells (25.7 ± 2.48 µm) compared to H. jonesii, H. minor, and H. yingeri. With respect to the guard cell width, both H. yingeri and H. minor had broader guard cells (19.4 ± 1.47 µm and 19.5 ± 1.72 µm, respectively), exceeding those of the remaining species by as much as 1.68-fold. H. clausa, on the other hand, had the narrowest guard cells (11.6 ± 2.28 µm).
In terms of the stomatal area (length × width), H. clausa again exhibited the smallest mean value (300.0 ± 70.74 µm2), whereas H. yingeri and H. minor had the largest values (624.9 ± 115.78 µm2 and 569.9 ± 62.07 µm2, respectively). Regarding the stomatal density, H. clausa displayed the highest density (111.0 ± 6.24 stomata/mm2), while H. yingeri, H. capitata, and H. jonesii exhibited the lowest values (41.1 ± 6.80, 43.0 ± 1.86, and 44.9 ± 5.13 stomata/mm2, respectively).
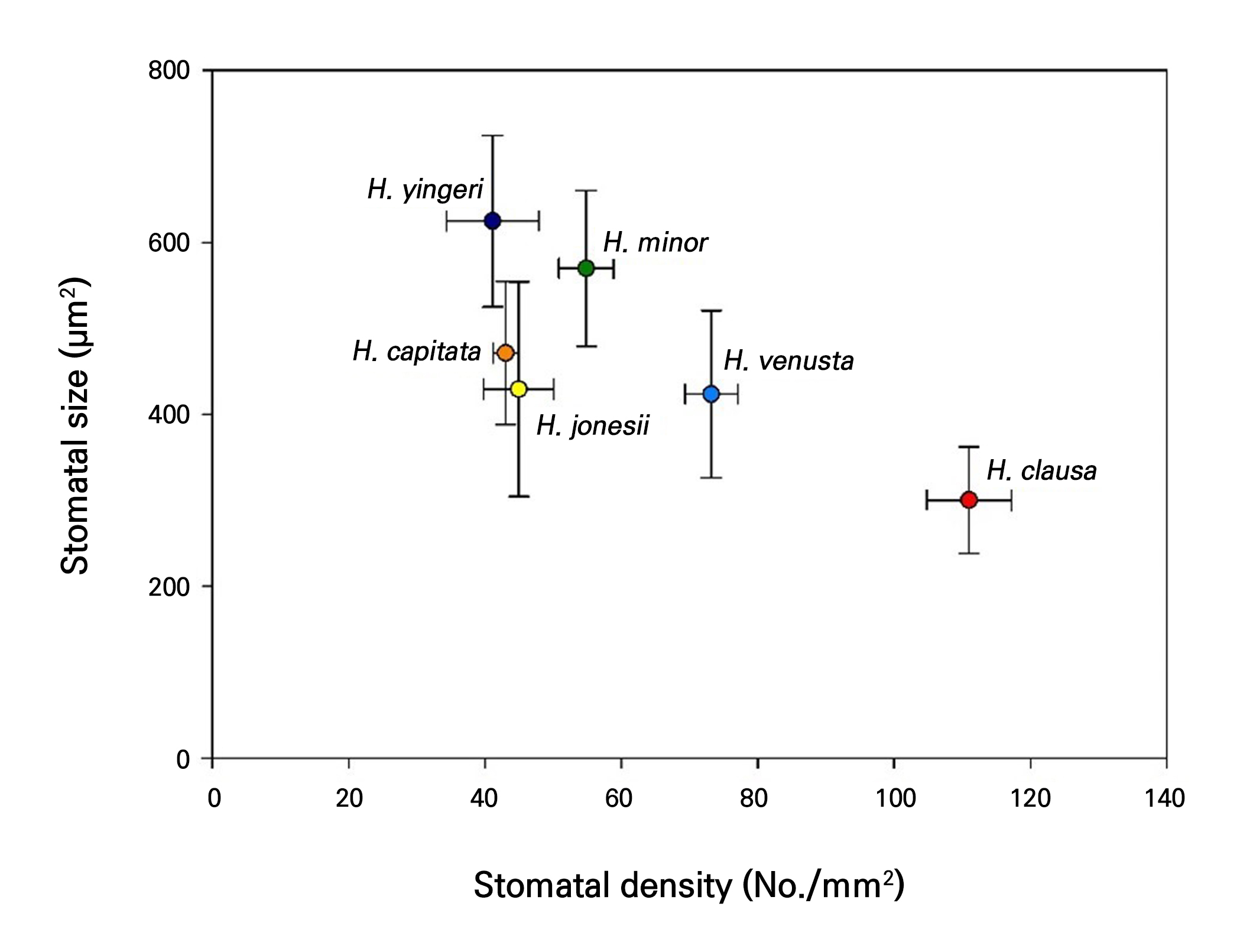
The stomatal density, length, and width were strongly correlated (Table 3). Stomatal density was significantly negatively correlated with stomatal size variables: length (r = –0.44***), width (r = –0.63**), and area (length × width, r = –0.61***). A significant positive correlation was also found between the stomatal length and width (r = 0.49***), and a strong positive correlation was observed between the stomatal width and area (r = 0.90***). Distinct stomatal characteristics were observed when the stomatal size and density were compared across species (Fig. 2). H. clausa demonstrated a tendency toward smaller but denser stomata and was clearly separated from the other species. H. yingeri exhibited the most contrasting characteristics. H. venusta exhibited a stomatal density pattern that differentiated it from the other five species; however, in terms of stomatal size, it generally matched H. capitata and H. jonesii.
Table 3.
Pearson correlation coefficients among the stomatal traits of the six Korean Hosta species
| Variable | Stomatal size | ||
| Guard cell length (L) | Guard cell width (W) | L × Wz | |
| Stomatal density | ‒0.44*** | ‒0.63** | ‒0.61*** |
| Guard cell length (L) | 0.49*** | 0.80** | |
| Guard cell width (W) | 0.90** | ||
Interspecific grouping based on PCA and cluster analysis
The PCA results revealed that the first two principal components (PC1 and PC2) accounted for 46.8% and 42.7% of the total variance, respectively, explaining 89.5% of the variation in the dataset (Fig. 3A). Individual samples were grouped by species, and confidence ellipses were drawn around each group. H. clausa was clearly distinguished from the other five species, whereas H. jonesii, H. capitata, and H. minor showed substantial overlap, suggesting a high degree of morphological similarity.
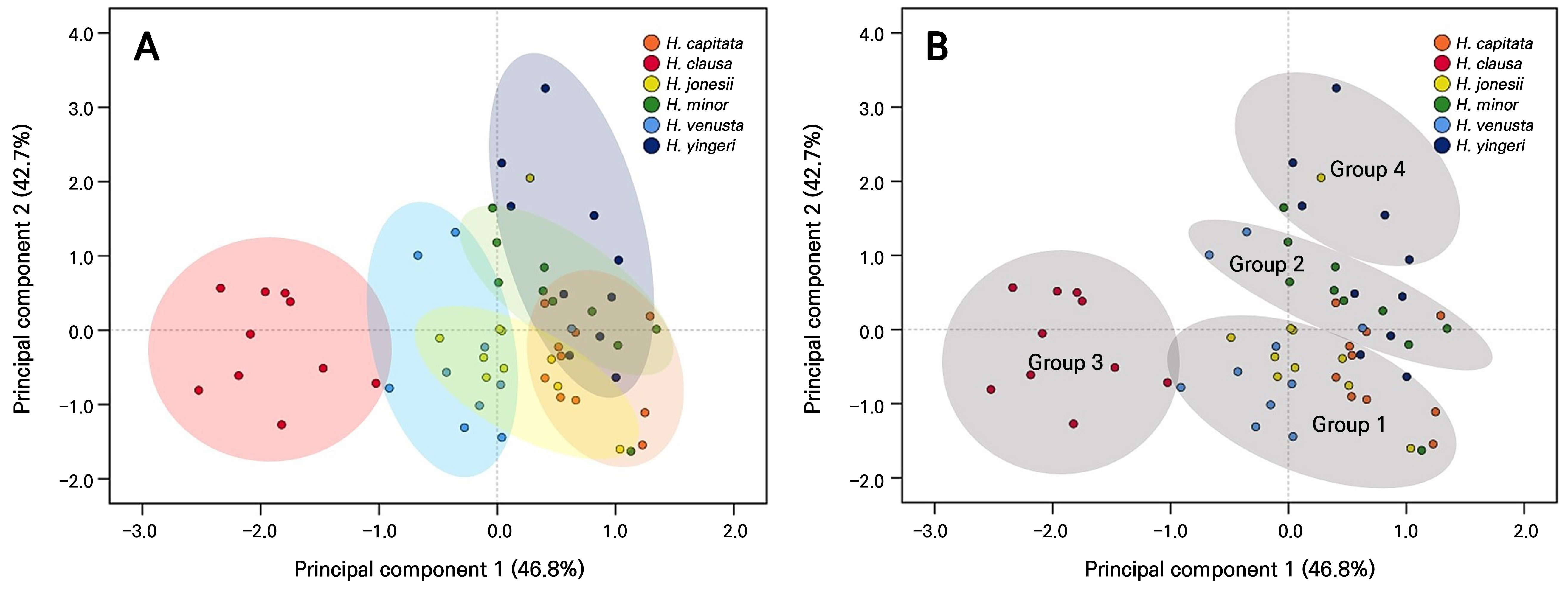
Fig. 3.
Principal component analysis (A) of stomatal traits among the six Korean Hosta species and hierarchical clustering analysis (B) illustrating species groupings. Stomatal traits include the stomatal density and size (guard cell length, width, and length × width). N = 60 (10 samples per species).
The cluster analysis further divided the 60 samples into four distinct groups, each characterized by specific stomatal traits. PCA revealed that the stomatal density loaded strongly and negatively on PC1 (–0.89), whereas the stomatal length showed the highest positive loading on PC2 (0.95). The stomatal width (0.80 on PC1, 0.45 on PC2) and stomatal area (L × W; 0.62 on PC1, 0.76 on PC2) contributed substantially to both components (Table 4). Group 1 consisted of 26 samples characterized by a relatively small stomatal size and low stomatal density (Table 5 and Fig. 3B). This group was primarily composed of H. jonesii (90%) and included approximately 70% of the populations of both H. capitata and H. venusta. Group 2 comprised 17 samples with relatively high stomatal densities and small guard cell dimensions. This group included eight samples of H. minor (80%), indicating that most individuals of this species shared a common stomatal profile. Group 3 was composed exclusively of ten samples of H. clausa (100%) exhibiting very high stomatal density and a small stomatal size. Uniformity within this group reflects the morphological distinctiveness of H. clausa relative to other species. Group 4 consisted exclusively of H. yingeri (50%) and was characterized by large guard cells and low stomatal density. This group exhibited the most pronounced contrast in traits when compared to Group 3.
Table 4.
Factor loadings of variable on principal component analysis
| Variable | Principal component | |
| 1 | 2 | |
| Stomatal density | ‒0.89 | ‒0.17 |
| Guard cell length | 0.20 | 0.95 |
| Guard cell width | 0.80 | 0.45 |
| L × Wz | 0.62 | 0.76 |
Table 5.
Means and standard deviations of the stomatal traits for four groups identified among six Korean Hosta species through a hierarchical clustering analysis
| Group | Statistics | Stomatal density | Guard cell length | Guard cell width | L × Wz |
| Group 1 | Mean | ‒0.34 | ‒0.50 | ‒0.28 | ‒0.46 |
| N | 26 | 26 | 26 | 26 | |
| SD | 0.538 | 0.597 | 0.598 | 0.382 | |
| Group 2 | Mean | ‒0.30 | 0.38 | 0.84 | 0.73 |
| N | 17 | 17 | 17 | 17 | |
| SD | 0.442 | 0.445 | 0.520 | 0.277 | |
| Group 3 | Mean | 1.96 | ‒0.70 | ‒1.40 | ‒1.23 |
| N | 10 | 10 | 10 | 10 | |
| SD | 0.246 | 0.684 | 0.664 | 0.514 | |
| Group 4 | Mean | ‒0.79 | 1.94 | 1.00 | 1.68 |
| N | 7 | 7 | 7 | 7 | |
| SD | 0.359 | 0.630 | 0.425 | 0.604 | |
| Total | Mean | 0 | 0 | 0 | 0 |
| N | 60 | 60 | 60 | 60 | |
| SD | 1 | 1 | 1 | 1 |
Discussion
Water loss through leaf transpiration and CO2 uptake for photosynthesis are primarily regulated by the stomata (Lawson and Blatt 2014). Thus, the stomata play a crucial role in leaf thermoregulation and water use efficiency (Faralli et al. 2022). Understanding interspecific variations in the stomatal density and size is essential for elucidating species-specific environmental adaptability, as these traits are influenced by both genetic factors and diverse growth conditions (Bertolino et al. 2019).
All of the six Hosta species examined in this study exhibited the anomocytic stomatal complex type (Fig. 1), which is characterized by guard cells surrounded by ordinary epidermal cells without distinct subsidiary cells (Rudall et al. 2013). This observation is consistent with previous reports on stomatal morphologies in Asparagaceae (Rudall et al. 2017; Ibrahim and Ismaeel 2022) and other Hosta species (Sedelnikova 2021). Despite this morphological consistency, significant interspecific variations were observed in terms of the stomatal density and size (Table 2).
Our results have revealed a clear inverse relationship between the stomatal density and stomatal size (Table 3), consistent with findings from studies on Arabidopsis epidermal patterning factor mutants and Oryza sativa (Ohsumi et al. 2007; Doheny-Adams et al. 2012). This trade-off stems from the need to minimize the negative effects caused by stomata being in close proximity or direct contact and to maintain sufficient epidermal allocation to the stomata to support an optimal gas exchange capacity (de Boer et al. 2016). However, as previous findings on stomatal traits have largely been derived from non-Hosta species, their interpretation in the context of Hosta species may be constrained by physiological differences.
PC1 primarily represents variation in the stomatal density, while PC2 reflects the stomatal size (Table 4). The separation of density and size into different components indicates that these stomatal characteristics can be regulated independently, potentially reflecting adaptive trade-offs between maximizing the gas exchange capacity and maintaining water use efficiency. Among the six species, H. clausa exhibited high stomatal density coupled with a small stomatal size, whereas H. yingeri showed the opposite pattern, with low stomatal density but a large stomatal size (Table 5, Figs. 2 and 3). Generally, smaller stomata are associated with faster stomatal responses and higher photosynthetic efficiency rates under fluctuating environmental conditions (Harrison et al. 2020). The small, densely distributed stomata of H. clausa may contribute to its enhanced adaptability to environmental variability, which may partly explain its broader habitat range than that of H. yingeri (Lee et al. 2018; Kew 2025b). H. clausa commonly occurs along rivers and valleys, consistent with its intolerance to dry conditions and, by extension, its observed stomatal traits (Jo and Kim 2017; Kim et al. 2025). However, as direct measurements linking water availability in its natural habitat to stomatal function are lacking, further research is needed.
Moreover, the substantial overlap observed among the five Hosta species in the PCA corresponded to their closer phylogenetic relationships, as inferred from the plastome data, in contrast to the more distinct H. clausa (Yang et al. 2021; Yoo et al. 2021). Morphologically, H. clausa is also differentiated by its long rhizomes and predominant clonal propagation (Lee et al. 2018).
This study has certain limitations, as the plants were cultivated under greenhouse rather than natural habitat conditions, and the relatively small sample size may also restrict the generalization of the findings. To elucidate stomatal adaptations in Hosta species further, future studies should focus on assessing stomatal responses under a wider range of environmental conditions; such studies can also include controlled stress experiments. Comprehensive ecological investigations of the native habitats of each species are essential to clarify the relationship between stomatal traits and environmental adaptation.