Introduction
Materials and Methods
Experimental field and apple tree selection
Treatments
Determination of the incidence and severity of stem bowl cracking
Storage conditions and duration
Determination of fruit quality characteristics
Quantification of cuticle weight in apple skins
Cuticle and epidermal cell image analysis
Statistical analysis
Results
Incidence rate and severity index of stem bowl cracking in ‘Picnic’ apples
Cuticular properties of apple skins
Fruit quality characteristics at harvest
Fruit quality characteristics during postharvest storage
Discussion
Introduction
‘Picnic’ (Malus × domestica Borkh.) is a popular Korean apple cultivar known for its excellent flavor and attractive color, and its production is steadily increasing (Kwon et al. 2015). However, this cultivar is highly susceptible to stem bowl cracking (also referred to as stem-end cracking or splitting) at harvest. This disorder can manifest as single or multiple cracks, developing during fruit maturation, at harvest, or during postharvest storage (Lu and Lin 2011). The incidence of stem bowl cracking in apple fruits is influenced by various factors, including cultivar characteristics, temperature, humidity, plant nutrient levels, rootstock type, irrigation practices, and fruit maturity stage (Opara et al. 2000; Fallahi et al. 2013; Wang et al. 2021). Postharvest storage exacerbates the severity of cracking, increasing susceptibility to fruit decay and pathogen attack, while reducing flavor quality and storability (Lee et al. 2019b). Therefore, implementing effective management strategies to mitigate apple stem bowl cracking is crucial to ensure optimal fruit quality and economic value both at harvest and during postharvest storage.
Fruit cracking is a physical failure of the skin, appearing as fractures in the cuticle or epidermal layers (Opara et al. 1997). The apple skin comprises cuticle, epidermal cells, and multiple hypodermal cell layers (Lara et al. 2014, 2015). The cuticle, the outermost protective layer, plays a critical role in minimizing moisture loss, infection, physical damage, and transpiration (Lara et al. 2014, 2015). Previous studies have shown that cuticle deposition ceases during fruit maturation and ripening, resulting in reduced cuticle thickness and area, which increases the risk of cracking. Furthermore, cuticle changes during storage contribute to fruit water loss and shriveling (Konarska 2013; Fernández-Muñoz et al. 2022).
Foliar applications of mineral nutrients and plant growth regulators are widely used in orchards to enhance fruit quality and color (Lee et al. 2019a; Lee et al. 2020), reduce fruit drop and sunburn, control pests and diseases (Peng et al. 2025), and improve the postharvest storability of fruits (Kim et al. 2017; Tanou et al. 2017). Calcium chloride (CaCl2) is a commonly used treatment, applied as a pre-harvest foliar spray or as a postharvest fruit dip, to improve fruit quality and extend storability. Recent studies have demonstrated that pre-harvest foliar application of CaCl2 reduces fruit drop and sunburn incidence while enhancing the fruit weight and quality of apples at harvest (Irfan et al. 2020; Khadavi and Hosseini 2024). Additionally, CaCl2 treatments have been shown to improve postharvest physiological characteristics and cell wall metabolism in apples (Ortiz et al. 2011; Gago et al. 2016) and reduce cracking in sweet cherries during postharvest storage (Wang and Long 2015).
Glycine betaine (GB) is another potential treatment, known for stabilizing and maintaining the integrity of fruit cell membranes during storage (Wang et al. 2019; Habibi et al. 2022a). GB acts as a biological agent, reducing disease incidence, wounds, and decay in apples (Sui et al. 2012; Zhang et al. 2017). It also mitigates oxidative stress by decreasing reactive oxygen species accumulation and lipid peroxidation, while inducing antioxidant enzymes under stress conditions (Zhang et al. 2017). Furthermore, GB enhances cold stress tolerance in papaya and banana fruits during storage (Pan et al. 2019; Chen et al. 2021). Despite its potential, studies on GB as a foliar treatment for apple cracking and cuticular metabolism remain limited, particularly in comparison with CaCl2 applications. Furthermore, the effects of these treatments on postharvest storability and fruit quality have not been extensively investigated.
Therefore, this study aimed to evaluate the effectiveness of pre-harvest CaCl2 and GB treatments in mitigating stem bowl cracking in ‘Picnic’ apples. It also evaluated the effects of these treatments on cuticular metabolism in apple skins. Additionally, this study investigated the influence of pre-harvest treatments on apple fruit quality at harvest and during postharvest storage.
Materials and Methods
Experimental field and apple tree selection
This experiment was conducted in a commercial apple orchard located in Jibo-myeon, Yecheon-gun, Gyeongsangbuk-do Province, Korea. Six-year-old ‘Picnic’ apple trees with similar-sized canopies, planted in the same environment and soil conditions with a 3.0 m × 1.2 m spacing, were selected for this study. The trees were grafted onto M.9 rootstocks and irrigated using a drip irrigation system. The orchard followed an integrated pest management system. The apple trees fully bloomed on April 25, 2022, and the fruits were harvested 155 days after full bloom, on September 27, 2022.
Treatments
Three pre-harvest treatment groups were tested in this study, with a total of 27 trees selected from the orchard. Each treatment consisted of nine apple trees, with three replicates per treatment. The first group received a 0.5% calcium chloride (CaCl2, Sigma Aldrich, Seoul, Korea) spray at 45, 30, and 15 days before harvest (DBH). The second group received a 10 mL·L‐1 of glycine betaine (GB, Fitomaat SL, Futureco Bioscience, Barcelona, Spain) spray at the same intervals. The third group was sprayed with water as a control. An electrodeposit agent (Silwet L-77, Agrotech Ltd., Seoul, Korea) was used during foliar applications to enhance the efficacy of the treatments. A hand sprayer fitted with a halocon nozzle (ATR60, Solcera, Evreux Cedex, France) was used for all treatments. Foliar application was performed in the morning around 8:00 a.m. After harvest, fruit samples were sent to the crop quality management laboratory of Kyungpook National University, Daegu, Korea, within 1.5 h for further physiological analyses conducted at harvest and during cold storage.
Determination of the incidence and severity of stem bowl cracking
The incidence and severity of stem bowl cracking were assessed in all harvested apples from each tree, following the method of Lee et al. (2019b), with slight modifications. Fruits exhibiting stem bowl cracking symptoms were classified as cracked, and the incidence rate was calculated using the formula: Fruit cracking rate (%) = (Number of cracking fruits/Total number of harvested fruits) × 100. Representative symptoms of stem bowl cracking observed in ‘Picnic’ apples are shown in Fig. 1A–1D.
Cracking severity was scored on a 0–5 scale based on the percentage of relevant cracking size or area damaged by stem bowl cracking: 0 = 0%, 1 = 1–20%, 2 = 21–40%, 3 = 41–60%, 4 = 61–80%, and 5 = 81–100% of the apple skin.
Storage conditions and duration
After determining the cracking incidence at harvest, 180 apples (60 fruits per treatment) were selected for quality evaluation during cold storage. Only fruits without cracking symptoms were selected to evaluate the occurrence of stem bowl cracking in apples during postharvest storage at 0 ± 1°C temperature and 90% ± 2% relative humidity, with physiological changes measured at 1-month intervals for up to 3 months.
Determination of fruit quality characteristics
Fruit physiological characteristics were measured to assess the effects of pre-harvest treatments at harvest and during storage intervals. Fruit weight was measured individually at harvest and after storage using a digital scale. Weight loss during storage was calculated as: Fruit weight loss (%) = [(Fruit weight before storage – Fruit weight after storage)/ Fruit weight before storage] × 100, as described by Kim et al. (2018). IEC was measured by extracting 1 mL of internal gas with a hypodermic needle and analyzing it using a gas chromatograph (GC-2010, Shimazu, Japan), with oven, flame, and detector temperatures set to 90°C, 100°C, and 200°C, respectively (Yoo et al. 2021). Fruit color values (L*, a*, and b*) were measured on sun-exposed and sun-shaded sides of each fruit using a chroma meter (CR-400, Konica Minolta, Tokyo, Japan) (Shin et al. 2023). Starch pattern index (SPI) was scored on a scale of 1–8 using the Cornell SPI method (Blanpied and Sisly 1992). Fruit firmness was measured three times per fruit using a Rheometer (Compac-100Ⅱ, Sun Scientific, Tokyo, Japan) with an 11-mm (diameter) plunger. The juice extracted from the fruit was used for soluble solids content (SSC) and titratable acidity (TA) determination. SSC was measured using a portable refractometer (PR-201ɑ, Atago, Tokyo, Japan), while TA was measured by titrating malic acid with 0.1 NaOH solution using a titrator (DL-15, Mettler Toledo, Mississauga, Canada), following the method of Yoo et al. (2022). A total of 15 fruits were analyzed for each quality attribute.
Quantification of cuticle weight in apple skins
Cuticle weight was measured using the method given by Joshi et al. (2018). A 10 mm diameter skin sample was collected from the equator of the fruit using a cork borer (Korea Ace Scientific Co., Seoul, Korea). The sample was incubated in 0.1 M sodium acetate buffer (pH 3.8) at 60 rpm and 37°C temperature for 1 week. Subsequently, the buffer solution was replaced with a lysis solution containing 4.73 unit·mL‐1 pectinase and 1.57 unit·mL‐1 cellulase (Sigma Aldrich, St. Louis, MO, USA) in 0.1 M sodium acetate buffer, and incubation was continued under the same conditions for another week. Then, epidermal and parenchymatous cells adhering to the cuticle were removed, and the cuticle membrane was dried at 40°C in a programmable forced convection oven (JSOF-150, JS Research Inc., Gongju, Korea). The weight of the cuticle was measured per square centimeter of fruit surface. Each fruit was measured three times, and the results were averaged. Five biological replicates per treatment were analyzed.
Cuticle and epidermal cell image analysis
The cuticle layer and epidermal cells in the cracking surface area near the stem-end region of the fruit were analyzed. Samples (0.5 mm × 5 mm × 5 mm area) were examined using an optical microscope (Olympus BX53, Tokyo, Japan) equipped with a 10× eyepiece (SWH10X-H) and a 10× objective lens (UPlanSApo 10X/ 0.40). Photographs were captured and displayed via CCD (CV-M4+CL, JAI, Miyazaki, Japan) using the Cytovision program. The boundary between the cuticle layer and epidermal cells was delineated using the Image J program. The area and thickness of the cuticle layer were measured at five locations on each fruit and averaged. Five biological replicates per treatment were analyzed.
Statistical analysis
Statistical analyses were performed using IBM SPSS statistics 26 (IBM Corp., Armonk, NY, USA). Data were analyzed using analysis of variance (ANOVA), and mean differences among treatments at harvest and during each storage interval were compared using Tukey’s HSD test (p < 0.05).
Results
Incidence rate and severity index of stem bowl cracking in ‘Picnic’ apples
At harvest, the incidence of stem bowl cracking was 21.41%, 10.46%, and 6.74% in the control, CaCl2, and GB treatments, respectively (Fig. 2A). The control fruits exhibited 10.95% (over 2-fold) and 14.67% (over 3-fold) higher cracking incidence compared to fruits from the CaCl2 and GB treatments, respectively. Additionally, the incidence rate in the CaCl2 treatment was 3.72% (1.5-fold) higher than that in the GB treatment (Fig. 2A). The severity index of stem bowl cracking was significantly higher in the control compared to both the CaCl2 and GB treatments (Fig. 2B). Additionally, the severity index in the CaCl2 treatment was higher than that in the GB treatment at harvest (Fig. 2B).
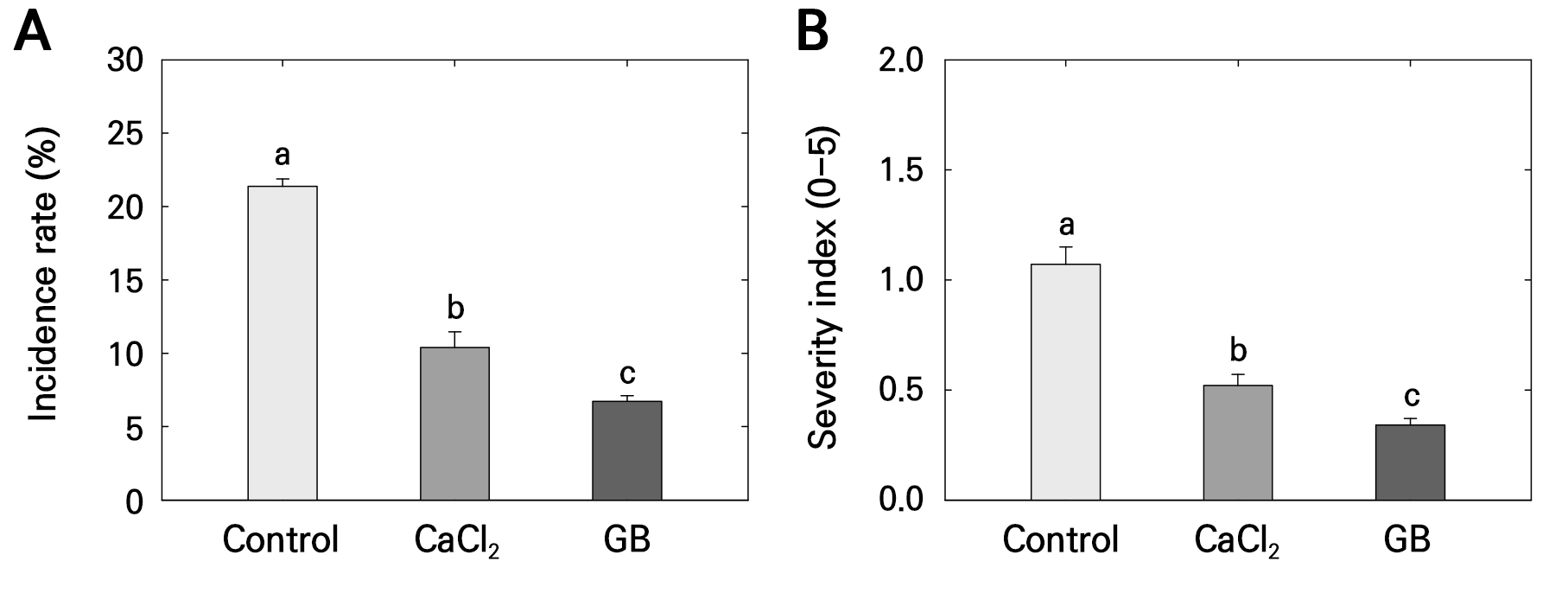
Fig. 2.
Incidence rate (A) and severity index (B) of stem-end cracking in ‘Picnic’ apples following pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments, observed at harvest. Bars represent means ± standard errors (n = 15). Different lowercase letters indicate significant differences based on Tukey’s HSD test at p < 0.05. The incidence rate is assessed based on the number of cracking fruits observed in the total number of harvested fruits. Cracking severity is scored on a 0–5 scale based on the percentage of relevant cracking size or area damaged by stem bowl cracking: 0 = 0%, 1 = 1–20%, 2 = 21–40%, 3 = 41–60%, 4 = 61–80%, and 5 = 81–100% of the apple skin.
Cuticular properties of apple skins
At harvest, cuticular properties, including thickness, area, and weight, were analyzed (Table 1). Both CaCl2 and GB treatments increased the cuticle area and weight compared to the control (Table 1). The cuticle thickness also increased in fruits from these treatments, although CaCl2-treated fruits had a thinner cuticle than GB-treated fruits, with no significant difference in cuticle area (Table 1). Additionally, optical microscopy revealed visually thicker cuticle layers in CaCl2- and GB-treated fruits compared to the control (Fig. 3A–3C).
Table 1.
Cuticular properties (area, weight, and thickness) of ‘Picnic’ apple skins observed at harvest after pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments
| Treatments |
Area of cuticle (mm2) |
Weight of cuticle (mg·cm‐2) |
Thickness of cuticle (µm) |
| Control | 0.37 ± 0.04z by | 2.50 ± 0.06 b | 18.90 ± 0.40 b |
| CaCl2 | 0.52 ± 0.03 a | 2.68 ± 0.05 a | 32.10 ± 0.35 a |
| GB | 0.56 ± 0.04 a | 2.70 ± 0.05 a | 32.15 ± 0.55 a |
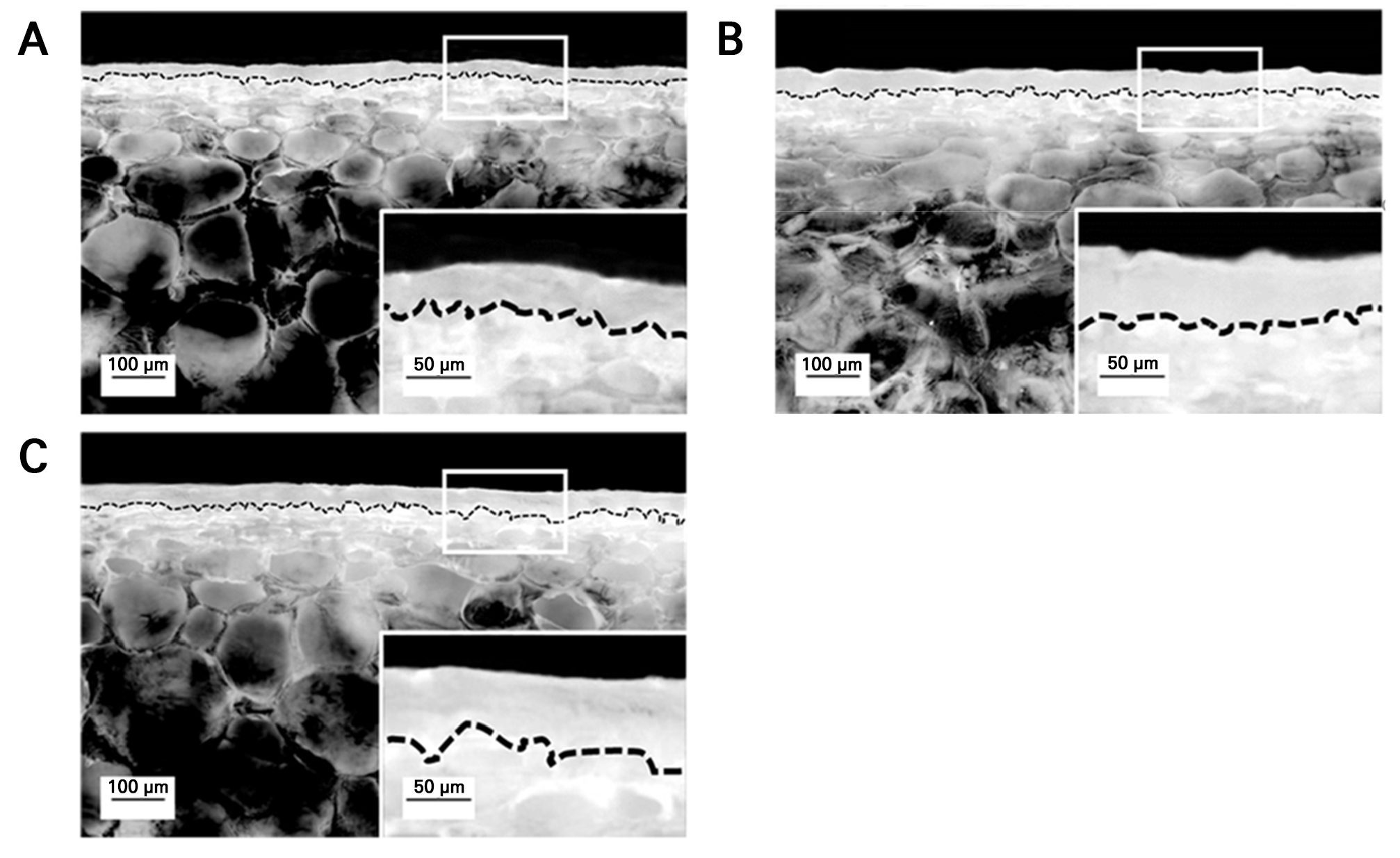
Fig. 3.
Optical microscopy images of the cuticle in the skin of ‘Picnic’ apples after pre-harvest foliar application of control (A), calcium chloride (B), and glycine betaine (C) treatments, observed at harvest. Samples were taken from the equatorial region of the apple skin. Black dotted lines represent the boundary between the cuticle and epidermal cells. Enlarged images (50 µm) of the cuticle from the highlighted regions (white squares) are shown in the upper-right corners of each panel.
Fruit quality characteristics at harvest
Fruit quality characteristics at harvest are summarized in Table 2. Pre-harvest CaCl2 and GB applications did not affect fruit weight or maturity indicators, such as flesh firmness, IEC, and SPI. However, fruits from both CaCl2 and GB treatments showed higher SSC compared to those from the control, while TA remained unaffected by the treatments (Table 2). In terms of fruit color, no significant differences were observed on the sun-shaded side, although redness (a*) was higher in the control. On the sun-exposed side, lightness (L*) was lower in the control compared to the CaCl2 treatment, but similar to the GB treatment. Additionally, no significant differences in b* values were observed among the treatments at harvest (Table 2).
Table 2.
Fruit quality characteristics of ‘Picnic’ apples observed at harvest following pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments
| Treatments |
Fruit weight (g) |
Flesh firmness (N) |
Internal ethylene concentration (µL·L‐1) |
Starch pattern index (1–8) |
Soluble solids content (°Brix) |
Titratable acidity (%) | |
| Control | 245.1 ± 5.05z ay | 66.83 ± 1.16 a | 0.33 ± 0.05 a | 6.33 ± 0.06 a | 14.74 ± 0.13 b | 0.37 ± 0.01 a | |
| CaCl2 | 249.7 ± 7.70 a | 67.20 ± 0.33 a | 0.49 ± 0.12 a | 6.33 ± 0.17 a | 15.29 ± 0.20 a | 0.39 ± 0.02 a | |
| GB | 239.0 ± 8.50 a | 66.19 ± 0.63 a | 0.47 ± 0.05 a | 6.17 ± 0.26 a | 15.27 ± 0.14 a | 0.40 ± 0.01 a | |
| Fruit color values on the sun-exposed side | Fruit color values on the sun-shaded side | ||||||
| L* | a* | b* | L* | a* | b* | ||
| Control | 42.18 ± 0.55 b | 31.42 ± 0.35 a | 11.20 ± 0.10 a | 58.30 ± 1.79 a | ‒2.99 ± 0.93 a | 15.20 ± 0.57 a | |
| CaCl2 | 44.72 ± 0.93 a | 29.71 ± 0.27 b | 11.89 ± 0.17 a | 62.15 ± 2.56 a | ‒3.31 ± 0.86 a | 16.96 ± 0.95 a | |
| GB | 43.32 ± 0.15 ab | 29.92 ± 0.46 b | 11.32 ± 0.42 a | 59.02 ± 2.41 a | ‒2.10 ± 0.96 a | 15.78 ± 1.07 a | |
Fruit quality characteristics during postharvest storage
The effects of foliar CaCl₂ and GB treatments on fruit quality during cold storage (0°C) were monitored for 3 months. IEC increased in all treatments from 2 to 3 months but showed no significant differences among treatments (Fig. 4A). Fruit weight loss also increased during storage but was not significantly different among treatments (Fig. 4B).
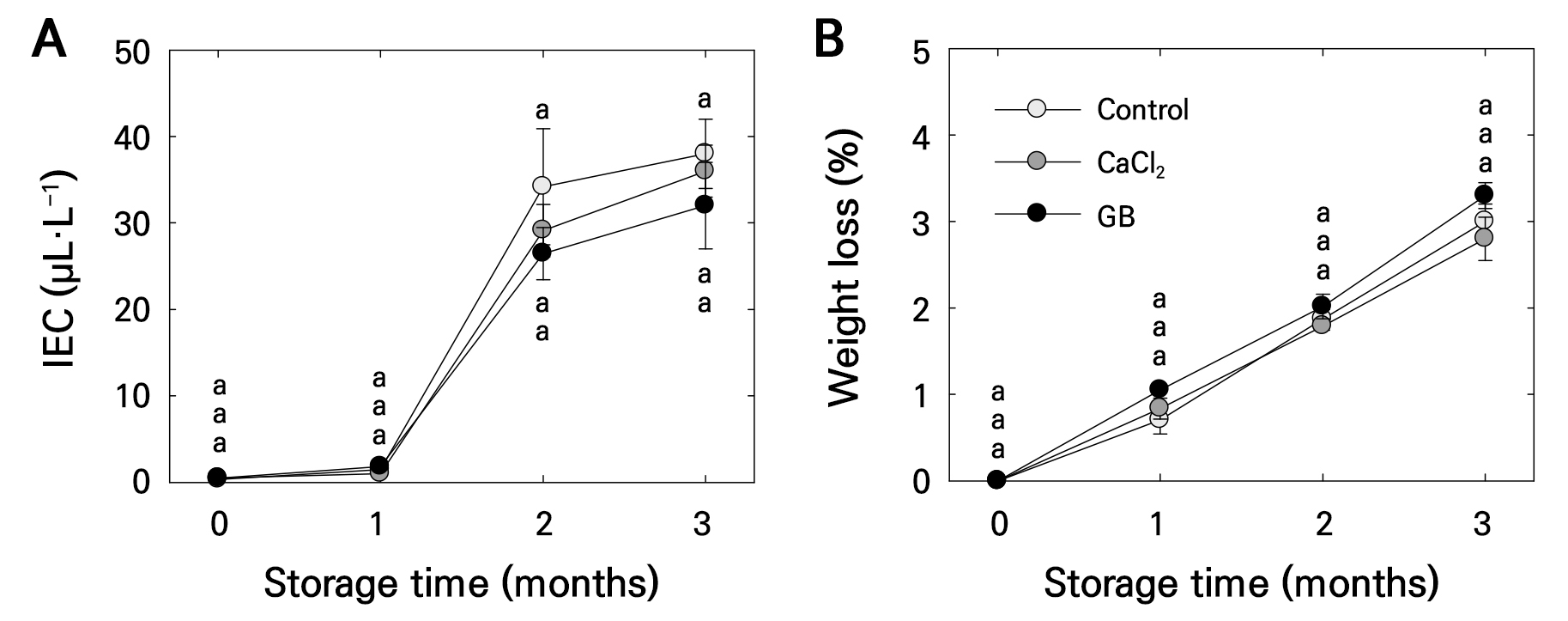
Fig. 4.
Internal ethylene concentration (IEC) (A) and fruit weight loss (B) in ‘Picnic’ apples after pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments, observed at harvest and during 3 months of postharvest storage at 0°C. Bars represent means ± standard errors (n = 15). Different lowercase letters indicate significant differences based on Tukey’s HSD test at p < 0.05.
Flesh firmness remained consistent across treatments for 2 months, but after 3 months, the control fruits exhibited lower firmness compared to GB-treated fruits (Fig. 5A). SPI increased during cold storage, but no significant differences were observed across treatments throughout the storage duration (Fig. 5B). TA was lower in the control than in CaCl2- and GB-treated fruits after 2 to 3 months of storage (Fig. 5C). SSC was higher in GB-treated fruits compared to the control and CaCl2 treatments after 2 to 3 months (Fig. 5D), with the control fruits also exhibiting higher SSC than CaCl2-treated fruits (Fig. 5D).
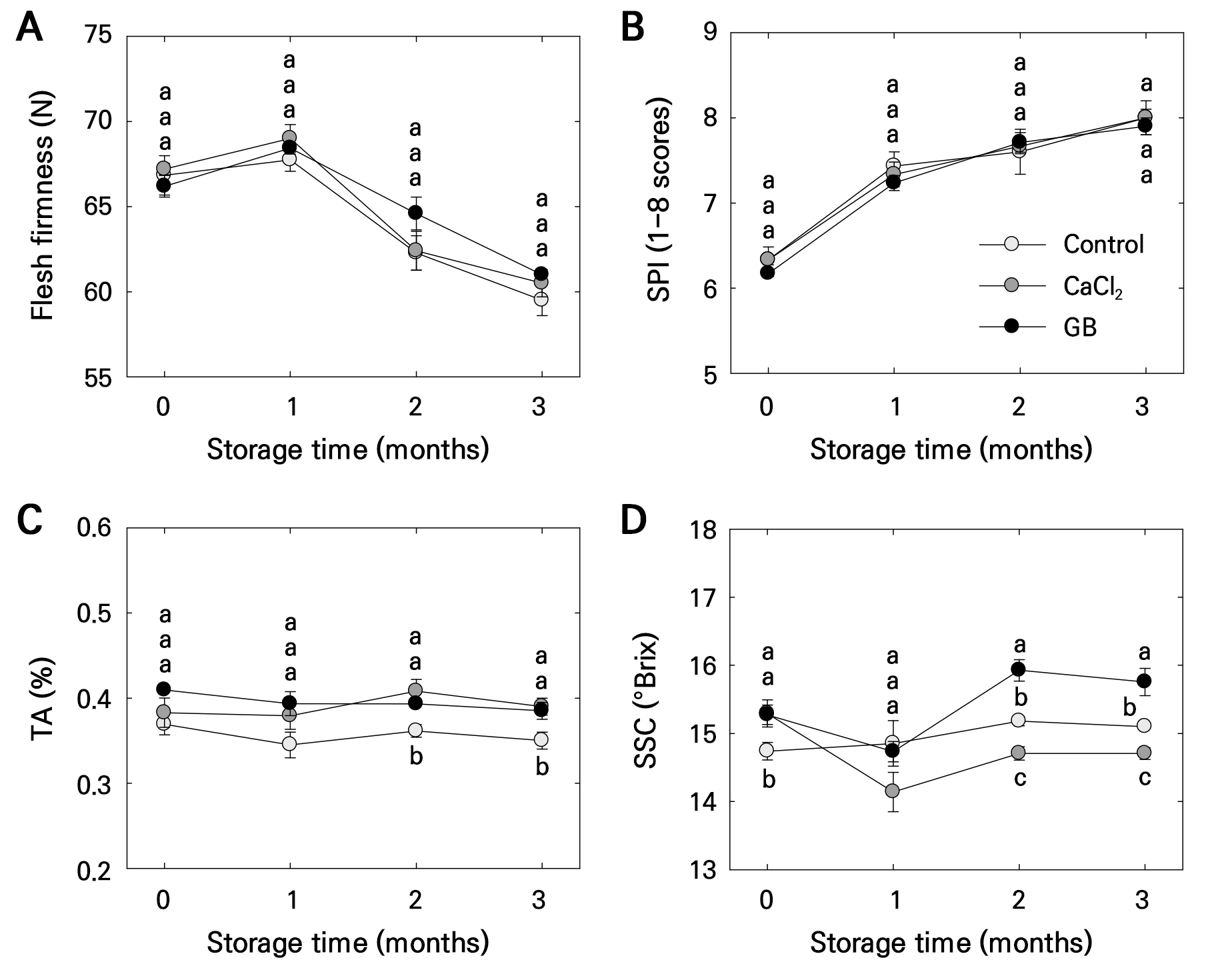
Fig. 5.
Flesh firmness (A), starch pattern index (SPI) (B), titratable acidity (TA) (C), and soluble solids content (SSC) (D) in ‘Picnic’ apples after pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments, observed at harvest and during 3 months of postharvest storage at 0°C. Bars represent means ± standard errors (n = 15). Different lowercase letters indicate significant differences based on Tukey’s HSD test at p < 0.05.
Changes in fruit color during storage were more pronounced on the sun-exposed side than the sun-shaded side (Fig. 6A–6F). No significant differences in L*, a*, and b* values on the sun-shaded side and in b* values on the sun-exposed side were observed across treatments (Fig. 6B and 6D–6F). However, the control exhibited lower L* (from 1 to 3 months) and higher a* (from 2 to 3 months) values on the sun-shaded side compared to the CaCl2 and GB treatments, respectively (Fig. 6A, 6C).
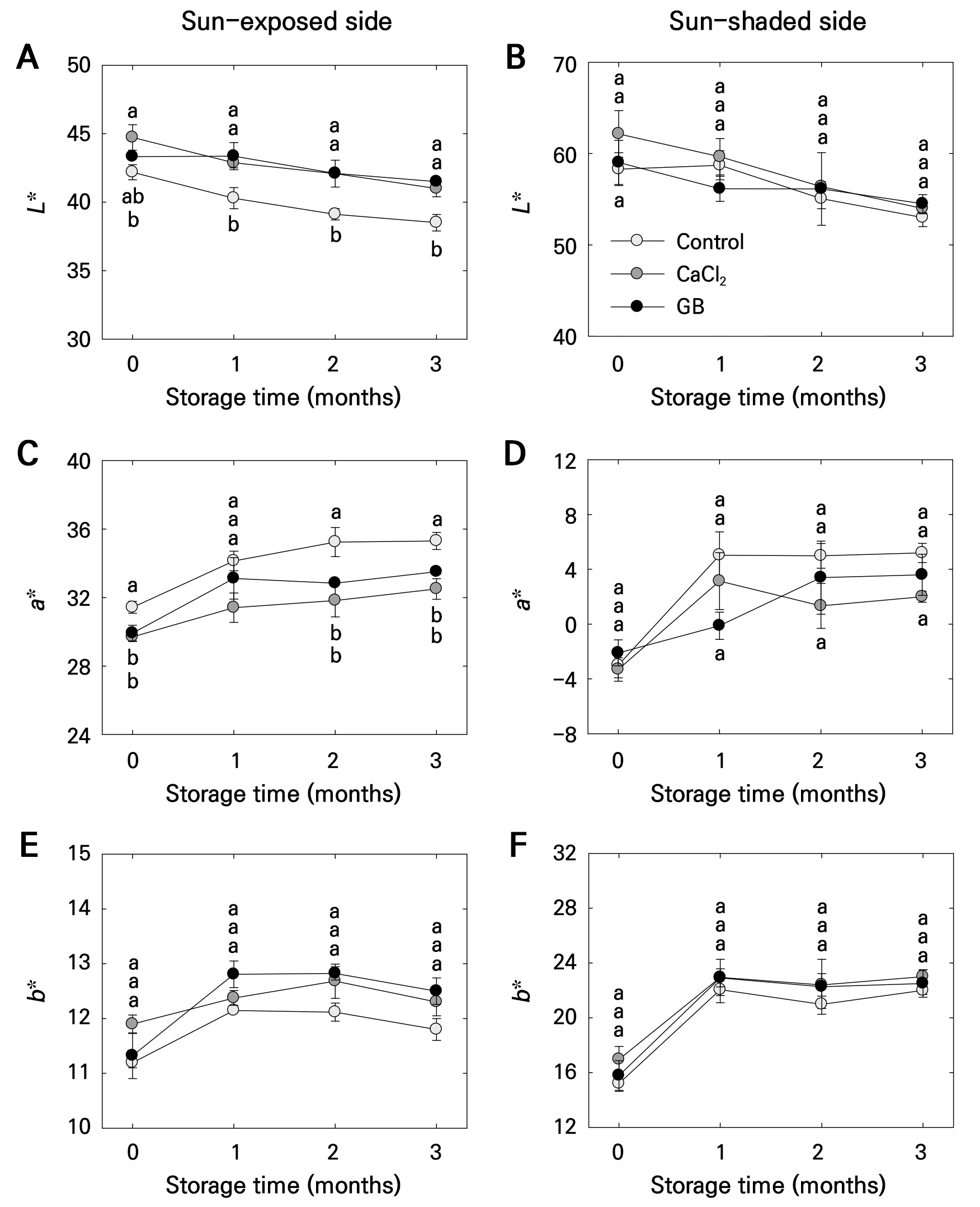
Fig. 6.
Fruit color values (L*, a*, and b*) of the sun-exposed (A, C, and E) and sun-shaded (B, D, and F) sides of ‘Picnic’ apples following pre-harvest foliar application of control, calcium chloride (CaCl2), and glycine betaine (GB) treatments, observed at harvest and during 3 months of postharvest storage at 0°C. Bars represent means ± standard errors (n = 15). Different lowercase letters indicate significant differences based on Tukey’s HSD test at p < 0.05.
Discussion
The production of high-quality apples is crucial for achieving better market value. However, fruit cracking, such as stem bowl cracking or splitting, can increase the risk of disease infection, significantly reducing apple quality, marketability, and storability. To address this issue, this study evaluated the potential of pre-harvest foliar applications of CaCl2 and GB, both known for enhancing cell wall integrity and stress tolerance, to mitigate stem bowl cracking in ‘Picnic’ apples and examine their effects on fruit quality during postharvest storage.
The incidence rate and severity of stem bowl cracking were significantly reduced in apples treated with CaCl2 and GB compared to the control at harvest. Calcium plays a key role in strengthening cell wall integrity, and lower calcium levels have been linked to increased susceptibility to cracking (Fan et al. 2023; Santos et al. 2023). Exogenous calcium application has been shown to reduce cracking at all stages of fruit development (Yu et al. 2020; Shi et al. 2024). Similarly, GB helps maintain cell membrane integrity by enhancing phenylalanine ammonia lyase (PAL) and antioxidant enzyme activities (Habibi et al. 2022a, 2022b), reducing lipid peroxidation and cellular damage (Wang et al. 2019). Therefore, the observed reduction in stem bowl cracking in the CaCl2- and GB-treated apples in the present study could be possibly attributed to these mechanisms.
Fruit cracking is closely associated with the cuticular metabolism of fruit skins (Lara et al. 2015; Santos et al. 2023). In the present study, both CaCl2 and GB treatments improved the morphological characteristics (area, weight, and thickness) of the cuticle at harvest. SEM analysis further confirmed that cuticle thickness was visually enhanced in treated fruits. Tessmer et al. (2023) reported that cuticle thickness increases as fruit matures. The cuticle and epidermal cells help regulate cuticular transpiration and protect fruits against biotic and abiotic stress (Konarska 2013). Lee et al. (2013) suggested that fruit cracking is linked to the enlargement of micro-cracks in the cuticle under stressful conditions. Therefore, increasing the cuticle thickness may help prevent fruit cracking in apples. Ortiz et al. (2011) reported that CaCl2 improves cell wall thickness and strength in apple fruits. Similarly, as mentioned earlier, GB enhances stress tolerance mechanisms in fruits by boosting antioxidant enzyme activity and reducing cell membrane damage (Shan et al. 2016; Habibi et al. 2022b). Therefore, the observed improvement in cuticular characteristics after foliar applications of CaCl2 and GB likely contributed to the reduction in fruit cracking in this study.
Despite the positive effects on stem bowl cracking, pre-harvest treatments had limited impact on other fruit quality parameters at harvest and during storage. The SPI scores, which assess the maturity of apples (Yoo et al. 2023), steadily increased across all treatments, indicating that the fruit ripened during storage. Doerflinger et al. (2015) reported a relationship between IEC and SPI scores. Additionally, SPI scores have been shown to significantly influence apple fruit quality parameters (Gonçalves et al. 2017). Consistent with previous studies, IEC increased while flesh firmness and TA decreased as SPI scores increased. However, no significant differences in SPI scores or flesh firmness were observed across treatments at both harvest and during storage.
Flesh firmness is closely associated with fruit ethylene production levels, and inhibiting ethylene production can delay the reduction of flesh firmness during storage (Payasi et al. 2009; Ireland et al. 2014). The lack of significant changes in flesh firmness in this study suggests that CaCl2 and GB treatments did not effectively inhibit ethylene production. Wang and Long (2015) reported that the maintenance of flesh firmness by CaCl2 varied depending on the cultivar and concentration applied, and lower concentrations of CaCl2 do not inhibit ethylene production during storage (Han et al. 2021). Similarly, it has been reported that a higher concentration of GB is more effective in maintaining flesh firmness in orange fruit during storage (Habibi et al. 2022b). Additionally, both foliar applications did not reduce weight loss during postharvest storage. Weight loss is associated with the respiration and transpiration rates of fruits (Fonseca et al. 2002). Since respiration is an ethylene-triggered process, inhibiting ethylene production can reduce the respiration rate in apples (Kwon et al. 2024). However, the results of this study indicate that pre-harvest foliar applications of CaCl2 and GB may not significantly affect the ethylene inhibition mechanism or the maturity levels of apples during postharvest storage.
Both foliar treatments showed minimal effects on SSC, TA, and fruit color. SSC increased in both treatments at harvest, with higher SSC observed in GB-treated fruits during storage. The reduction in TA was slower in both CaCl2 and GB treatments during storage, although no significant difference between treatments was observed at harvest. Both CaCl2 and GB treatments maintained a decrease in L* and an increase in a*, particularly on the sun-exposed side of the fruits during storage. The slower TA reduction can be attributed to the maintenance of organic acid levels during storage (Habibi et al. 2022b). On the other hand, the increase in red color (a* value) is attributed to the degradation of chlorophyll to anthocyanin in the fruit skin (Kapoor et al. 2022). Ortiz et al. (2011) reported that pre-harvest CaCl2 application resulted in higher TA levels but reduced SSC in apple fruits. Another study also reported a slower TA reduction in CaCl2-treated apple fruits during storage (Shirzadeh et al. 2011). Habibi et al. (2022b) reported that GB treatment slowed TA reduction and increased SSC during cold storage. Additionally, Shan et al. (2016) reported that GB treatment helped maintain fruit coloration under cold stress, consistent with the findings of the present study.
In conclusion, pre-harvest foliar applications of CaCl2 and GB effectively reduced the incidence and severity of stem bowl cracking at harvest by improving the morphological characteristics (thickness, area, and weight) of the cuticle in apple skins. Among the two treatments, GB was more effective than CaCl2 in reducing the incidence of stem bowl cracking, although neither treatment significantly affected the cuticle. Apart from a higher SSC value, both foliar treatments had no significant effect on other fruit quality parameters, including fruit weight, firmness, TA, SPI, and IEC, at harvest. During postharvest storage, both CaCl2 and GB treatments slowed the reduction of TA and helped maintain a* and L* values, but had minimal impact on other fruit quality parameters. This highlights the potential complexity of fruit maturation and the factors influencing these characteristics. Since calcium accumulation in fruit tissues is often linked to improve structural integrity and reduce fruit cracking, future studies should include the analysis of calcium contents to provide a more comprehensive understanding of its contribution on cracking reduction. Moreover, further studies should explore the combined effects of CaCl2 and GB foliar applications on fruit cracking and quality.



