Introduction
Materials and Methods
Sample Preparation
Assay of Immune Activity
Assay of XO Inhibitory Activity
Assay of Reducing Power
Assay of Cytotoxicity
Data Analysis
Results and Discussion
Responses of Immune Activity Toward T and B Cells
XO Inhibitory Activity
Reducing Power
Cytotoxicity
Introduction
Recently, plants and plant-derived products have become a part of the healthcare system, with the increasing use of bioactive phytochemicals. Studies have shown that the traditional medicinal plants
have in vitro mutagenic, toxic, and carcinogenic properties (Li et al., 2007; Pulla et al., 2008). Therefore, it is important to explore medicinal plants for their cytotoxicity. The evaluation of cytotoxicity of plants is a major subject in pharmaceutical studies, particularly in the area of cancer research (Cuyacot et al., 2014).
Codonopsis lanceolata is a perennial flowering plant of the Campanulaceae family and is grown commercially in East Asia. The species is known to exert various pharmacological effects in humans. The roots of C. lanceolata mainly contain triterpenoid saponins, including codonolasides I-V and lancemaside A-G (Boo et al., 2016; Moon et al., 2018). The saponins exhibit anti-inflammatory effects for conditions such as bronchitis and cough, insomnia, and hypomnesia. Lancemaside A, which is the main constituent of C. lanceolata, has been reported to potently inhibit LPS-stimulated, TLR-4-linked NF-κB activation of the 293-hTLR4-hemagglutinin cells (Joh et al., 2010). In South Korea, C. lanceolata is widely used as a food, and the root extract has been used as a crude drug. However, its medicinal applications have not been explored thoroughly (Wang et al., 2011).
Adverse ecological conditions and stress factors result in immune dysfunction in humans. Synthetic, biotechnological, and natural medicinal products have been used to mitigate immunological disorders (Isaykina et al., 2008). Furthermore, medicinal plant extracts might also reduce the risk of oxidative stress and cell damage (Guizani et al., 2013). It has been reported that the increase in immune response improves the defense against various diseases, such as microbial infections and leukemia (Paul et al., 2014). Both cells and molecules of the immune system play important roles. Additionally, cellular dichotomy in adaptive immune responses is also reflected in functional division, where T cells serve as effector cells. Cell-mediated immune responses, such as delayed-type hypersensitivity, and B cells serve as helpers in the production of highly specific proteins (Janeway et al., 1999). Xanthine oxidase (XO) is an important enzyme that catalyzes the oxidation of hypoxanthine to xanthine to uric acid in human. This suggests that compounds that inhibit the production of superoxide anion might have a beneficial effect not only in the treatment of hyperuricaemia and gout, but also in the alleviation of inflammation (Cimanga et al., 2001). Antioxidant compounds in food play an important role in maintaining good health. Scientific evidence suggests that antioxidants reduce the risk of chronic diseases, including cancer and heart disease. Most of the antioxidant compounds in a typical diet are derived from plant sources, and they belong to various classes of compounds with various physical and chemical properties. Several studies have also investigated changes in functional materials during storage (Tilahun et al., 2017), which are important in selecting storage conditions. Therefore, the present study aimed to evaluate the XO inhibitory activity, reducing power, immune activity, and cytotoxicity of 30% ethanolic extracts of C. lanceolata roots stored at various temperatures and for different durations.
Materials and Methods
Sample Preparation
Three-year-old C. lanceolata roots grown in the Jeju region and harvested in autumn were purchased from a commercial farm. The roots of C. lanceolate, which were stored at different temperatures (5, 15, 25, 35, and 45°C) in a thermo-chamber and with various storage periods (0, 15, 30, 45, 60, 75, and 90 days), were freeze-dried and ground to a fine powder. Generally, growers sell the roots soon after harvest, but sometimes they may be stored for up to 3 months. We assumed 90 days (3 months) as the maximum storage period because there is a considerable difference in quality, depending on storage temperature and period. The C. lanceolata roots were stored at a relative humidity of 50% in our experiments. The powder was stored at -20°C until further analyses. The freeze-dried powder was mixed with 30% ethanol, and the filtrate was collected thrice with constant stirring of the mixture at 24-h intervals for 72 h. The filtrate was then concentrated under reduced pressure at 45°C using a vacuum rotary evaporator (IKA® RV 10 Basic Digital, IKA Co., Munich, Germany). The concentrated extract was stored at -20°C until further analysis.
Assay of Immune Activity
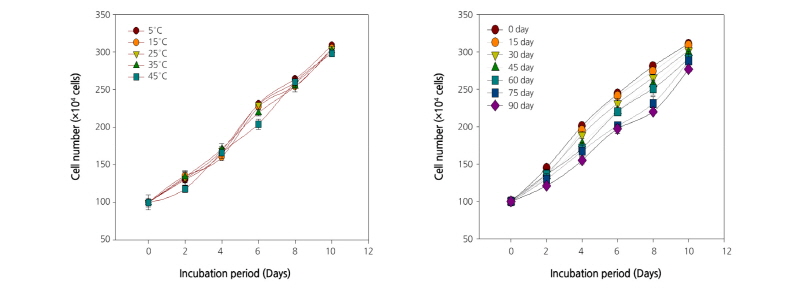
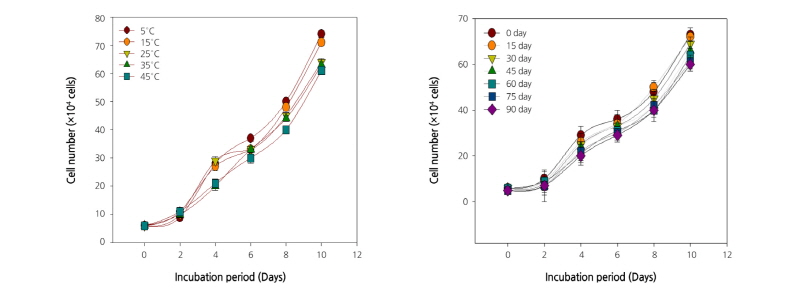
The extract immune enhancement effect was assayed according to the method of Lee et al. (2004) using T [CCRF-CEM (Human T cell lymphoblast-like cell line), KCLB (Korean Cell Line Bank) No. 10119], and B cells [RPMI (Roswell Park Memorial Institute) 8226, KCLB No. 10155]. T and B cell experiments were carried out to verify the effect on immune cell growth. The cells were incubated for 24 h in RPMI-1640 medium containing 10% fetal bovine serum (FBS) at 37°C with 5% carbon dioxide (CO2) in a humidified incubator (MCO-18 AC, Sanyo Co., Osaka, Japan). After incubation for 24 h, the purified cells were cultured with 0.5 µg·mL-1 extract for 1~10 days to a density of 2.5 × 104 cells/well in 24-well microtiter plates. After 10 days, the immune enhancement effect of the treatment was determined by counting the number of cells using a hemocytometer (Marienfield Superior Company, Baden-Wurttemberg, Germany) and then compared with that of untreated cells.
Assay of XO Inhibitory Activity
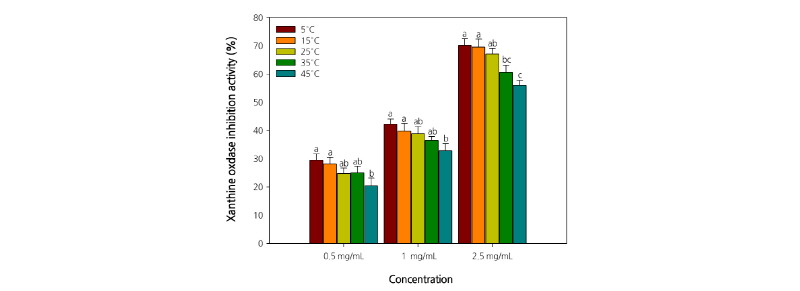
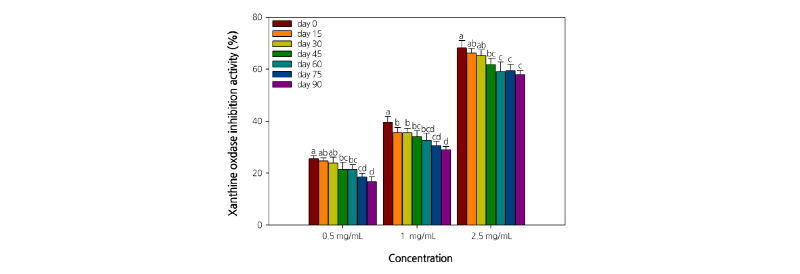
The XO inhibitory activity was measured spectrophotometrically by the method of Noro (1983) with the following modification. The assay mixture consisted of 1.0 mL of extract, 0.6 mL of 0.1 M potassium phosphate buffer (K2HPO4+ KH2PO4, pH 7.5), 0.2 mL of substrate solution dissolved in 2 mM xanthine, and 0.1 mL of XO solution (0.2 U·mL-1). After preincubation of the mixture at 37°C for 15 min, the reaction was initiated by the addition of 2.0 mL of substrate solution. This assay mixture was incubated at 37°C for 30 min. The reaction was stopped by the addition of 1 mL of 1 N hydrochloric acid (HCl), and the absorbance of the assay mixture at 292 nm was measured spectrophotometrically (Libra S22, Biochrom Co., Cambridge, England). Similarly, a blank was prepared, but the enzyme solution was added to the assay mixture after the addition of 1 N HCl. One unit of XO was defined as the amount of enzyme producing 1 µmol of uric acid per minute at 37°C. The XO inhibitory activity was expressed as the percentage of inhibition of XO in the above assay system. It was calculated as (1 - B/A) × 100, where A and B are the amount of uric acid in the reaction mixture and control, respectively.
Assay of Reducing Power
The reducing power of the extract was determined by the modified method of Oyaizu (1986). Various concentrations (1, 2.5, and 5 mg·mL-1) of 30% ethanolic root extract were mixed with 500 µL of sodium phosphate buffer (200 mM, pH 6.6) and 500 µL of 1% potassium ferricyanide. The mixture was then placed in a water bath for 20 min at 50°C. The resulting solution was cooled rapidly, mixed with 2.5 mL of 10% trichloroacetic acid, and centrifuged (Smart R17, Hanil Scientific Inc. Korea) at 3,000 rpm for 10 min. The supernatant (500 µL) was mixed with 500 µL of distilled water and 100 µL of 1% ferric chloride. After 10 min, the absorbance of the resultant mixture was measured at 700 nm. The higher the absorbance value, the stronger the reducing power.
Assay of Cytotoxicity
The cytotoxicity of C. lanceolata extracts was assayed using human cancer cell lines, including HeLa, Calu-6, and MCF-7 for human cervical carcinoma, pulmonary carcinoma, and breast adenocarcinoma, respectively. The cell lines were purchased from the Korean Cell Line Bank (KCLB) for the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The cells were plated on 96-well plates at a concentration of 3 × 104 cells/mL. The cells were incubated for 24 h in RPMI-1640 medium at 37°C with 5% CO2 in a humidified incubator and then treated with 2 µL of various concentrations (50, 100, 200, 400, and 800 µg·mL-1) of the extracts. During the cytotoxicity test, the concentration of the extract was set at a minimum of 50 µg·mL-1 to a maximum of 800 µg·mL-1. We determined that concentrations greater than 800 µg·mL-1 did not affect cytotoxicity. After incubation for 48 h, the cells were washed twice with phosphate buffer solution (PBS). The MTT solution (5 mg·mL-1) was dissolved in 1 mL of PBS, and 10 µL of this solution was added to each well. After a reaction period of 4 h, the solution in each well containing media, unbound MTT, and dead cells was removed by suction, and 100 µL of dimethyl sulfoxide (DMSO) was added to each well. The plates were shaken for 15 min using a plate shaker, and the absorbance was recorded using an enzyme-linked immunosorbent assay (ELISA) reader (Bio-Rad model 550, Hercules, CA, USA) at a wavelength of 540 nm. Cell viability was determined as the percentage of viability of treated cells compared with that of the untreated cell, and the values were then used to iteratively calculate the concentration of extract required to induce a 50% reduction (IC50) in the growth of each cell line.
Data Analysis
Statistical analysis was performed using Statistical Analysis System software (SAS version 9.1, SAS Institute Inc., Cary, NC, USA). The analysis of variance (ANOVA), followed by Duncan’s multiple range test (DMRT), was used to determine significant difference (p < 0.05) between the treatments.
Results and Discussion
Responses of Immune Activity Toward T and B Cells
The growth of human T and B cell lines treated with extracts of C. lanceolata roots stored at various temperatures and for different periods is shown in Figs. 1 and 2. In the present study, the growth of T and B cells increased gradually with incubation period. All the extracts of C. lanceolata promoted cell growth until day 10 of incubation. Overall, the growth of T and B cells increased when the root extracts were stored at a low temperature and for a short period. The induction of immune response using products obtained from medicinal plants, as a possible therapeutic measure, has become a subject of active scientific investigations. The T cells targeting tumor-associated antigens (TAAs) are readily detectable in blood, tumors, and tumor-draining lymph nodes, even at late stages of tumor. These tumor-specific T cells can be used to establish functional TAA-specific T-cell lines, which kill autologous tumor cells in vitro and in vivo (Curiel et al., 2003; Curiel et al., 2004). The target expressed by B-cell lymphomas can be exploited by the active immunotherapeutic approach (Timmerman et al., 2002). The results of the present study confirmed the immune cell growth-enhancing effect of root extracts of C. lanceolata. Furthermore, it was presumed that there was no significant change in the composition even when the roots were stored at a high temperature. The immune cell growth was promoted by the extract in a concentration-dependent manner and did not show a significant difference. The growth of both T and B cells increased significantly until day 10 of incubation, and then gradually decreased. The results reveal that the growth of both T and B cell depends on the incubation period.
XO Inhibitory Activity
The XO inhibitory activity of C. lanceolata root extract tended to decrease significantly with the increase in storage period and temperature. Furthermore, the XO inhibitory activity of the extracts increased in a concentration-dependent manner (Figs. 3 and 4). The extracts at a concentration of 2.5 mg·mL-1 exhibited an inhibition rate of >50%. In particular, the inhibition rate was the highest (70.2%) with the extract of C. lanceolata roots stored at 5°C. Furthermore, the XO inhibitory activity of the extract (2.5 mg·mL-1) of C. lanceolata roots stored for different periods was also relatively high, and almost similar, with 68.3%, 66.2%, and 65.3% on days 0, 15, and 30, respectively. The results revealed that the XO inhibitory activity of the extracts was the highest when the roots were stored at low temperatures (5°C-15°C) for <30 d. Superoxide anions are produced by several oxidative systems, especially the xanthine-XO system. XO is an important enzyme catalyzing the oxidation of hypoxanthine to xanthine to uric acid in human (Cimanga et al., 2001). During the last step of purine metabolism, XO catalyzes the oxidation of xanthine and hypoxanthine to uric acid. Uricosuric drugs (which increase the urinary excretion of uric acid) and XO inhibitors (which block the terminal step of uric acid biosynthesis) can lower plasma uric acid concentration and are generally employed to treat gout. The superoxide anion radicals generated by XO are involved in various pathological conditions, such as hepatitis, inflammation, ischemia-reperfusion, carcinogenesis, and aging. Thus, the search for novel XO inhibitors would be beneficial not only to treat gout, but also to combat various diseases (Nguyen et al., 2004). The results of the present study indicate that the root extract of C. lanceolata might be useful to treat hyperuricemia and gout, thus laying a foundation for further studies on C. lanceolata to isolate active constituents and develop drugs.
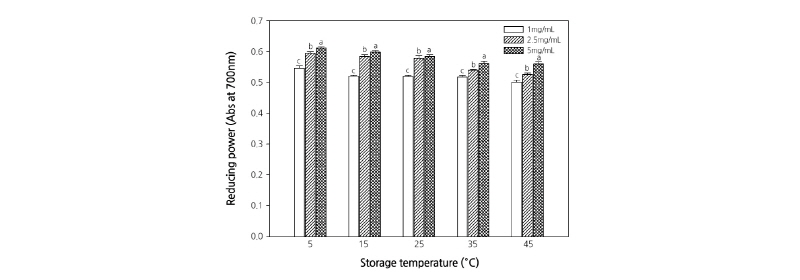
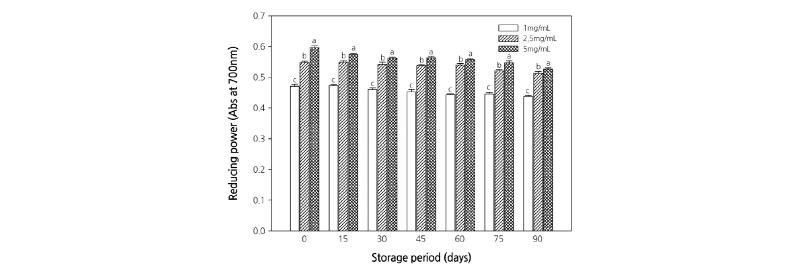
Reducing Power
Figs. 5 and 6 present the results of the reducing power assay. The reducing power of the extracts increased significantly in a concentration-dependent manner. However, when the roots were stored at temperatures >30°C, the reducing power of the root extract tended to decrease significantly. The reducing power of a compound is related to its electron transfer ability and may therefore serve as a significant indicator of its antioxidant activity. The reducing ability of a compound generally depends on the presence of reductones (Pin-Der-Duh, 1998), which exhibit antioxidative potential by breaking the free radical chain, donating a hydrogen atom (Gordon, 1990). The free radicals induce oxidative stress and alter the overall condition of the patients by paralyzing the immune system. It has been known that plant extracts containing phenolic and flavonoid compounds have antioxidant and antibacterial effects (Da-Silva et al., 2006; Majhenic et al., 2007; Pereira et al., 2007). The search for high-value natural sources might result in novel natural products with safer and better antioxidants that protect against oxidative damage. The results of the present study revealed the antioxidant properties of C. lanceolata, suggesting that C. lanceolata extract can be used as a natural reducing agent.
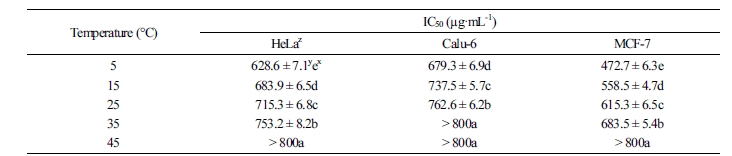
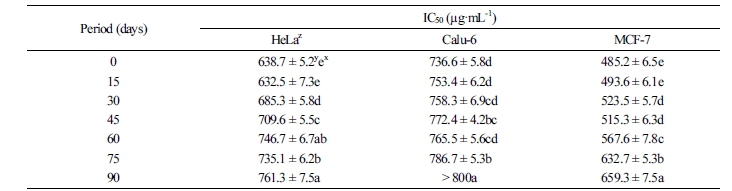
Cytotoxicity
The in vitro cytotoxicity of C. lanceolata on three human cancer cell lines was evaluated by the MTT assay. When cells were treated for 48 h with various concentrations (50, 100, 200, 400, and 800 µg·mL-1) of 30% ethanol extracts, the rate of cell survival progressively decreased in a dose-dependent manner. The results of the cytotoxicity assay are summarized in Tables 1 and 2. Overall, the cytotoxicity of C. lanceolata extracts differed significantly with storage temperature. The cytotoxic effect against the cancer cell lines was higher at storage temperatures below 25°C. In particular, the IC50 value of the extracts of roots stored at 5°C was the highest on the MCF-7 cell line (472.7 ± 6.3 µg·mL-1), followed by HeLa (628.6 ± 7.1 µg·mL-1) and Calu-6 cell lines (679.3 ± 6.9 µg·mL-1). On the contrary, the extracts of C. lanceolata roots stored at 45°C exhibited an IC50 value of >800 µg·mL-1 against the MCF-7, HeLa, and Calu-6 cell lines. The cytotoxicity of extracts of C. lanceolata roots stored for different periods was also the highest on the MCF-7 cell. The IC50 values of extracts of C. lanceolata roots stored for <15 d was the highest on the MCF-7 cell line (493.6 ± 6.1 µg·mL-1). Furthermore, significant activity was observed against HeLa (632.5 ± 7.3 µg·mL-1) and Calu-6 cell lines (753.4 ± 6.2 µg·mL-1). Overall, the cytotoxic activity of the three human cancer cell lines tended to decrease with increase in storage period. The persistent search for novel anticancer compounds in medicinal plants and traditional foods is a realistic and promising strategy. In plants, numerous compounds with anticancer properties have been identified, including alkaloids, phenylpropanoids, and terpenoids (Kintzios, 2006; Park et al., 2008; Yan-Wei et al., 2009; Vijayarathna and Sasidharan, 2012). Currently, there is a growing interest in herbal medicine worldwide, resulting in increased studies on the pharmacological properties of bioactive ingredients and their ability to treat various diseases (Lobo et al., 2009). It is well known that chemical and medicinal plant-based drugs might induce toxic effects. Based on the results of the present study, C. lanceolata can be used as a natural cytotoxic agent.
The variation in physiological functionalities during different storage periods at different temperatures may be associated with a reduction in phenolic compounds and enzymatic activity (data not shown). In general, the physiological functional substances (e.g., phenols and enzymes) of post-harvest crops are diminished due to high temperature and long-term storage. To further support our results, we plan to investigate various components that are affected by storage conditions. In particular, we will analyze Lansemaside A components, which are major functional substances in C. lanceolata.
In conclusion, our data on the physiological functionalities of C. lanceolata are useful for determining optimum storage conditions. Further in-depth research is necessary to support our results.