Introduction
Materials and Methods
Plant material, treatments, and environmental conditions
Morphological observations
Fresh weight and water balance measurements
Measurements of O2-., H2O2 contents
MDA content assay
Glutathione assays
Assay of GR activity
Statistical analysis
Results
Effects of COS on vase life and water absorption of cut roses
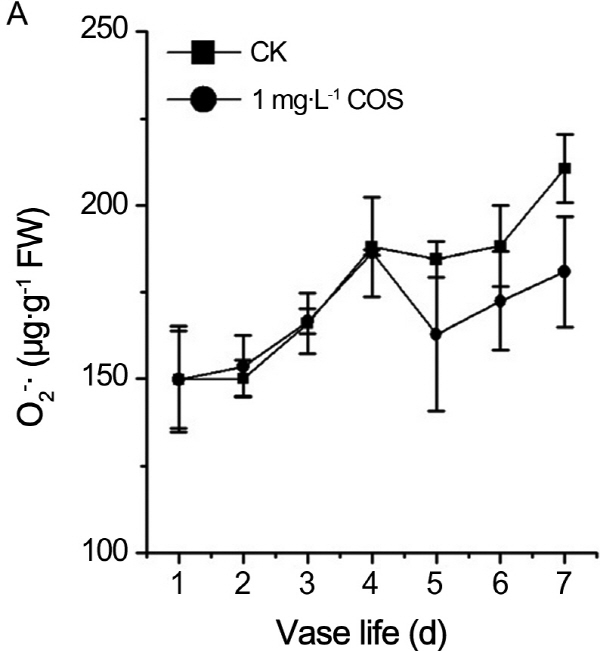
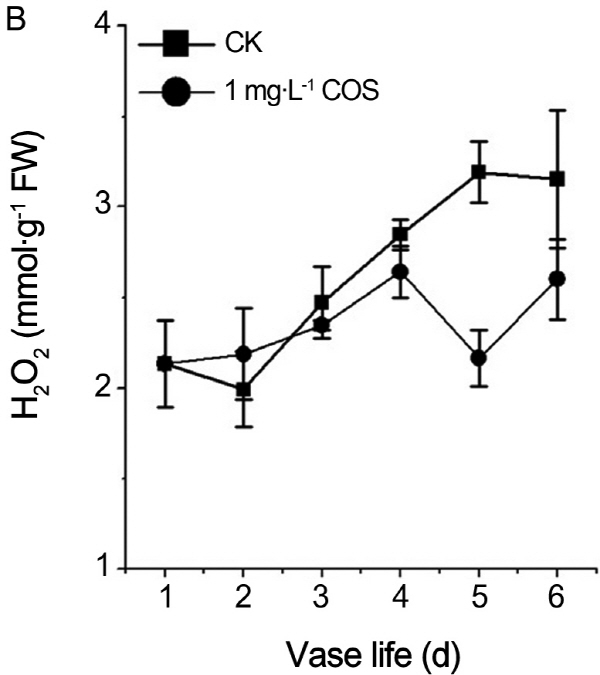
Effects of COS on contents of hydrogen peroxide and superoxide anion
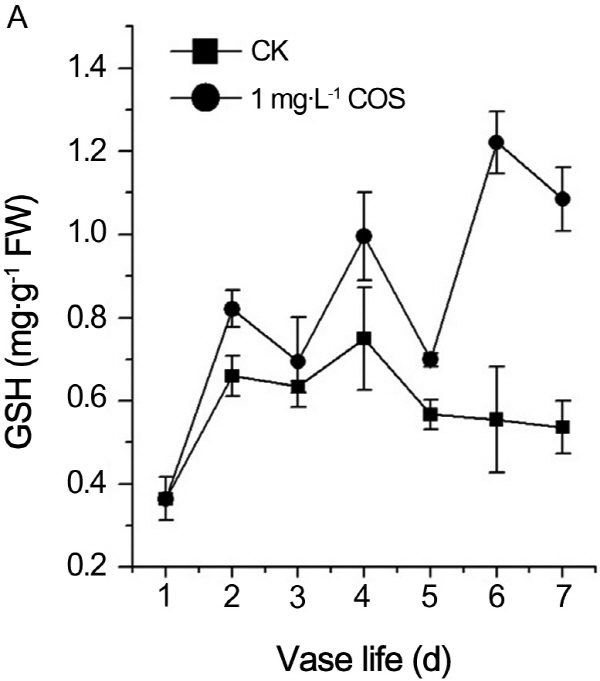
Effects of COS on contents of ROS nonenzymic scavenging molecules
Discussion
COS prolongs the vase life of cut roses
COS improves water absorption capacity of rose
The relationship of COS and ROS
Introduction
Reactive oxygen species (ROS) is the byproducts of normal aerobic metabolism and generated from plant organelles, such as chloroplasts, mitochondria and microbodies which have highly oxidizing activities or with sustained electron flows (Datet al., 2000). With the exception of the plant organelles, other main sources of ROS were peroxidases and oxidases in plants (Rinalducci et al., 2008). In mature petals of cut rose, the chloroplast is common to be disappeared, disintegrated or transformed to other components (Rogers, 2012). Therefore, ROS in mature petals usually derived from other two organelles or all kinds of peroxidases in petal cells.
It is confirmed that ROS is harmful for cellular functions and has vital roles in signaling events (Grennan, 2008). Many ROS, such as hydrogen peroxide (H2O2), superoxide anion (O2-.), and hydroxyl radical (OH.), will oxidize nearby biological macromolecules including DNA, proteins or lipids (Bokov et al., 2004; Rinalducci et al., 2008; Jing et al., 2011; Rogers, 2012) which can be damaging or even fatal for the cell (Mittler,2002). Malondialdehyde (MDA) is formed by oxidative damage of lipids which lead to membrane lipid mobility worse. Therefore, MDA is usually used as an index of plant oxidative stress (Scosndalios, 1994; Hodges et al., 1999). It was reported that the senescence of rose petals was accompanied with a burst of O2-. generation in cut rose (Kumar et al., 2007) and lilium (Abreu and Munné-Bosch, 2007). And rose petals at the later period of maturation were more sensitive to ROS owe to antioxidant defense capacity decrease (Kumar and Srivastava, 2008). Therefore, ROS may have vital role in senescence of rose petals (Rogers, 2012).
In order to sustain the relative stable circumstance in plant cell, the plant generally has both enzymatic and non- enzymatic mechanisms which can protect them against oxidative damage. Non-enzymatic molecule reduced glu-tathione (GSH) protects the cells of cut rose from ROS damage. In senescing rose petals, decline of GSH leads to shift the cellular redox toward more oxidized state (Kumar and Srivastava, 2008; Kumar et al., 2008). GSH can directly react with ROS and act as a substrate for enzymes scavenge ROS (Ghezzi et al., 2005; Szalai et al., 2009). In the enzymatic scavenging of ROS, glutathione reductase (GR) reduces oxidized glutathione (GSSG) to reduced GSH by use of NAD(P)H (Rogers, 2012).
Chitooligosaccharide (COS) is the degraded products of chitosan or chitin by several methods such as enzymatic and acidic hydrolysis. Many nonspecific enzymes, namely, cellulases, lipases and proteases, or specific enzymes chito-sanases, have been used to prepare COS (Thadathil and Velappan 2014). The 10 kDa or less COS have been found lots of biological activities owe to their structures and physicochemical properties (Xia et al., 2011), such as hypo-cholesterolemic (Kim and Rajapakse, 2005), antimicrobial (Jeon and Kim, 2000, 2001), immunity-enhancing and anticancer effects (Xia et al., 2011), accelerating calcium and iron absorption (Bravo-Osuna et al., 2007), antioxidant (Je et al., 2004; Mendis et al., 2007; Fang et al., 2013) and so on. However, relatively little is known about the effects and mechanism of COS on the vase life of cut roses. Therefore, in the paper, the results concerning the relationship between COS and ROS contents, non-enzymatic or enzymatic scavenging systems were reported in the cut roses “Red France”.
Material and Methods
Plant material, treatments, and environmental conditions
Cut roses (Rosa hybrida L.) “Red France” flowers were obtained from a local flowering plants market in Zhengzhou, Henan, China. The fresh vivid flowers were selected and immediately placed in tap water, then delivered to the laboratory within 2 h. After slant cutting the stems to a length of 30 cm under water, the flowers were treated with distilled water as control (CK), 1 mg・L-1 of COS as experimental group in 500 mL Erlenmeyer flask contained about 250 mL treated solutions. 1 mg・L-1 of COS was the best concentration through preliminary experiment and the molecular weight of COS was 2 kD and deacetylation of COS was 95%. Therefore, the flower were then placed in a climate-controlled room at 20-25°C, 30-40% relative humidity (RH), and 140 μmol・m-2・s-1 light intensity (LI) to simulate commercial vase life. The experiment was repeated three times and more than 30 flowers were placed to be observed and detected per treatment.
Morphological observations
Vase life was measured by the period from the first vase day to the last vase life when the flowers was lost ornamental value, as defined by pedicel bending, flower petal wilting and bluing, and general discoloration (Pompodakis and Joyce, 2003).
Fresh weight and water balance measurements
Fresh weight and water balance were both measured by electronic balance. Total weight of Erlenmeyer flask and flowers and solutions and weight of flowers were weighed every day respectively. The changes in fresh weight were presented as the percentage of initial weight. Otherwise, loss water was the difference of total weight in two continuous days likewise water absorption was the difference of weight of Erlenmeyer flask and solutions which total weight subtracts flower weight. So water balance was water absorption minus loss water.
Measurements of O2-., H2O2 contents
Superoxide anion content was determined according to the method developed by Elstner and Heupel (Oracz et al., 2007). One gram of outermost whorl petals was ground with liquid nitrogen. The O2-. was extracted with 5mL 50 mM of phosphate buffer (pH 7.8) containing 1 mM EDTA and 50 mg polyvinylpyrolidone (PVP-10). Extracts were filtered through two layers of cheesecloth and centrifuged at 12,000 × g for 15 min. The supernatant (1 mL) was first incubated at 25°C for 30 min in the presence of 1 mM hydroxylamine hydrochloride in 50 mM sodium phosphate buffer (pH 7.8). Then, 2 mL of this reaction mixture was incubated with 0.5 mL of 17 mM sulfanilamide and 0.5 mL of 7 mM 2-naphtylamine at 25°C for 30 min. The absorbance was measured at 530 nm after centrifugation at 13,000 g for 10 min. A calibration curve was established using sodium nitrite. The results are expressed as μg・g-1 FW.
To measure H2O2 content, rose petals (0.2 g FW) were ground in 1.4 mL of cold 5% trichloroacetic acid (TCA) (Patterson et al., 1984). The extracts were centrifuged at 12,000 g for 15 mins, the supernatants were used to detect H2O2 content using hydrogen peroxide colorimetric detection kits according to the instructions of manufacturer. (Nanjing Jiancheng Company of biological technology, China) (Zhang et al., 2009).
MDA content assay
To measure MDA content, 1 g petals was ground with liquid nitrogen using a mortar and pestle. 2-Thiobarbituric acid, the reactive substance of MDA was extracted with 5 mL 20% (w/v) TCA containing 0.5% TBA. Absorbance of the extracts was determined at 532 and 600 nm, respectively (Dhindsa and Thorpe, 1981), and MDA content was calculated using the extinction coefficient of 155 mM-1・cm-1.
Glutathione assays
Glutathione in its reduced (GSH) and oxidized (GSSG) form was determined according to Smith (1989). Petals (1 g FW) was homogenized in 5% (w/v) sulphosalicylic acid in an ice and then centrifuged at 10,000×g for 20 min. One 1 mL aliquot of the supernatant was removed and neutralized by adding 1.5 mL 0.5M potassium phosphate buffer (pH7.5). This sample was used for the determination of total glutathione (GSH+GSSG) content. Another 1 mL aliquot of neutralized supernatant was pretreated with 0.2 mL 2-vinylpyridine for 1.5 h at 25°C to mask the GSH and to permit determination of GSSG alone. Both samples were extracted twice with 5 mL 100% diethylether. The reaction mixture contained: 0.5 mL 0.1 M sodium phosphate buffer (pH 7.5) containing 5 mM EDTA, 0.2 mL 6 mM 5,5-dithiobis- (2-nitrobenzoic acid), 0.1 mL 2 mM NADPH, 0.1 mL (1 unit) of glutathione reductase type III (Sigma), and 0.1 mL of extract. The change in absorbance at 412 nm was followed at 25°C. A standard curve was prepared using a GSH standard.
Assay of GR activity
Glutathione reductase (GR) activity was measured according to Schaedle and Bassham (1977). The reaction mixture contained 50 mM Tris-HCl (pH 7.6), 0.15 mM NADPH, 0.5 mM oxidized glutathione (GSSG), 3 mM MgCl2, and up to 100 μL of homogenate (1 mg・mL-1 protein). NADPH oxidation was followed at 340 nm.
Protein content was determined according to the method of Bradford (1976) using bovine serum albumin a standard.
Statistical analysis
Measurements were carried out on three replicates with one flower being a replicate. Origin Version 8.0 (OriginLab Corporation, Northampton, USA) was used for analysis of data. Vase life and fresh weight changes were analyzed using analysis of variance (PROC ANOVA), and means separation was determined by t-test at p < 0.05 or p < 0.01 for each experiment. Other data are presented as means ± standard errors.
Results
Effects of COS on vase life and water absorption of cut roses
In order to checkout whether COS prolongs the vase life or alleviates the petal senescence of cut roses, 1 mg・L-1 of COS was applied to cut roses in the paper. From Table 1, we concluded that vase life of treatment by COS was 18.1 days and prolonged 6.4 days compared with control (11.7 days) (p < 0.01). In addition, The petals edge of cut roses treated by distilled water began to be perished and the pedicel of control inflorescence had been bended at the 9th vase day whereas those treated by COS was still bright-colored and strong (Fig. 1). Therefore, COS signifi-cantly prolonged the vase life and enhanced the morphous of petals and pedicle of cut rose.
To investigate the mechanism of COS on prolonging the vase life of cut roses, the value of water balance and change of fresh weight were calculated. From the Table 2, water balance of treatment by COS is minus at the fifth day and prolonged 2 days compared with control. That is mean that from the fifth day, water absorption of COS is less than water loss, namely, petals of cut flower began to be loss of some water owe to decrease of water absorption capacity. Therefore, COS remarkably improved water absorption capacity of cut rose. Correspondingly, fresh weight of control began to decrease at sixth day and one treated by COS still increase at eighth day (Table 3). Accordingly,COS increased the flower morphology of cut flowers and eventually prolonged the vase life of cut roses through improving the water absorption capacity of cut roses.
Effects of COS on contents of hydrogen peroxide and superoxide anion
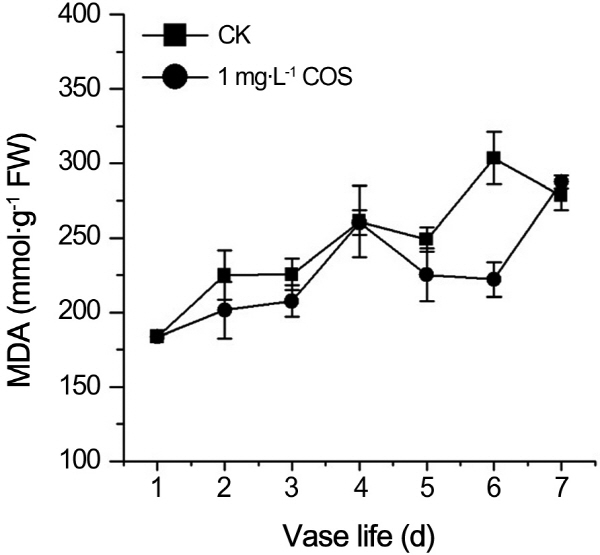
In order to verify whether prolongation vase life of cut rose by COS is related to ROS. Hydrogen peroxide and superoxide anion, as two kinds of ROS, had vital roles in process of plant senescence. Contents of O2-., H2O2 were recorded during the senescence process of cut roses. From Fig. 2, contents of O2-., H2O2 continued to rise no matter whether there is COS. O2-.content of treatment by COS was less than control at fifth and sixth day (Fig. 2A). Likewise, the H2O2 content was lower than control at 3th, 4th, and 6th day (Fig. 2B). MDA, as the products of peroxide of membrane lipid, is the index of plants damaged by ROS. As shown in Fig. 3, MDA content persistently increased in aging process of cut roses regardless of COS and MDA concentration of treatment by COS was less than one of control except the 7th day during petal aging process. Therefore, COS decreased ROS and MDA levels in the senescence process of cut roses.
Effects of COS on contents of ROS nonenzymic scavenging molecules
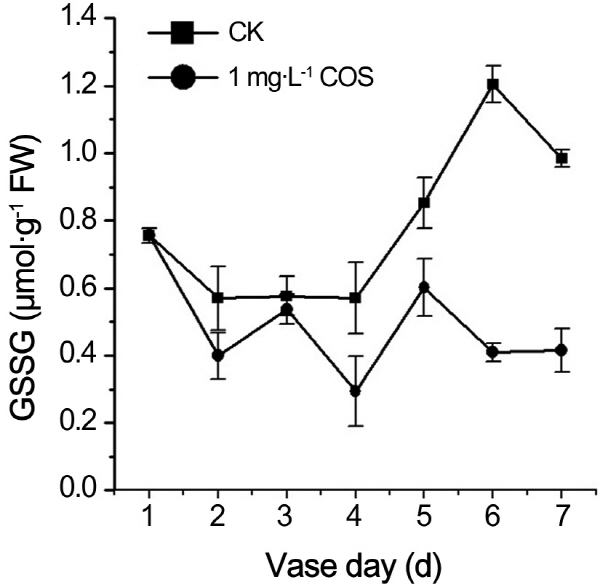
Influences of COS on ROS contents have shown as above. Content GSH, as non-enzymic ROS scavenging molecules, was measured. The results showed that GSH content of control increased firstly and started to decrease from the fourth day. On the contrary, fluctuant increase of GSH content in cut rose treated by COS were detected during senescence of cut rose (Fig. 4A). Same observations were reported by Kumar (2008 a, b) and Jing (2011). Corres-pondingly, oxidized glutathione of treatment by COS also changed slightly whereas GSSG content of control increased dramatically during the senescence of cut rose (Fig. 5). Therefore, COS shifted the redox homeostasis to reduction owe to increase o GSH contents.
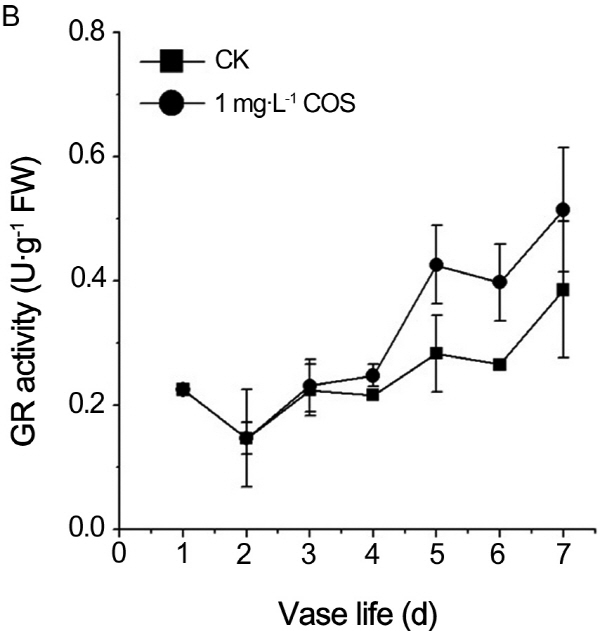
It is well known that GR reduce the oxidized GSSG to reduced GSH by use of NAD(P)H (Rogers, 2012). In order to search the reason of GSH increase and GSSG decrease in cut roses treated by COS, activity of GR in petals of cut rose was measured every day during the senescence period. As shown in Fig. 4B, GR activity continued to rise throughout the senescence period. GR activity of treatment by COS was higher than one of control during the whole aging process of cut roses. Accordingly, COS increased reduced GSH contents by activating GR activity during senescence process.
Discussion
COS prolongs the vase life of cut roses
COS was degraded from chitosan or chitin. Chitin is a major component of the shells of crustaceans, such as crab, shrimp and crawfish. Chitosan is a natural nontoxic biopolymer produced by the deacetylation of chitin. However, there is no obvious difference between chitosan and Chitin. Although COS, as a preservative, was applied in apple juice, and prolonged the juice storage period at 37°C from nine days to 70 days (Xia et al., 2011). In the paper, it is reported that COS prominently prolonged the vase life 6.4 days and markedly improved the ornamental quality of cut roses (Table 1 and Fig. 1). Therefore, COS may be as a harmless natural preservative of cur roses in the vase in the future.
COS improves water absorption capacity of rose
Though cut roses were incised from the whole plant, cut flowers were still living organism which needs to absorb a variety of nutrients, such as water, inorganic ion and so on. Most of all, loss of water lead to a lack of flower opening, premature petal wilting and bending of the pedicel (Xue et al., 2009). Vessels and sieve tube of cut roses were the vital tissues to water absorption. However, the liquid and water which transported by sieve tube and vessel were leakaged from the nick when cut roses were dissected from the whole flower tree. The site of the nick was very easy to multiply many kinds of bacteria and fungi owe to the liquid contained anorganic ion, small peptide, polysaccharide and so on. It’s well known that the COS is against the common bacteria, molds and yeasts (Joodi et al., 2011; Fang et al., 2013) connected with content of protonated amino group and relative molecular weight (Xia et al., 2011). Accordingly, COS improved the absorption of water in cut roses (Table 2) and increased the fresh weight (Table 3), flower morphology (Fig. 1) and eventually prolonged the vase life of cut roses 6.4 more days (Table 1) owe to sustaining the vessel and sieve tube unblocked as an antimicrobial.
The relationship of COS and ROS
ROS include both free radicals and hydrogen peroxide. Free radical, such as O2-., can oxidized nearby a range of macromolecules with varying specificity (Dat et al., 2000). H2O2, absent of unpaired electron, can diffuse across trans-membrane to other subcellular compartments where it may act as a signal and/or be removed. Our results showed that O2-. or H2O2 contents were notably decreased in petals of cut rose treated by COS during the petals senescence of cut rose (Fig. 2). It was reported that COS directly eliminated all kinds of ROS, such as O2-., H2O2, OH., and so on (Je et al., 2004; Mendis et al., 2007; Fang et al., 2013). Je et al. (2004) have shown that COS could quench various radicals by the action of nitrogen on C-2 position of COS. Moreover, it was reported that the scavenging mechanisms of chitosans are related to their hydrogen donating ability to free radicals to form stable molecules (Mendis et al., 2007; Fang et al., 2013). MDA, the products of lipid peroxidation, was also decreased markedly by COS (Fig. 3). Above results is consistant with the results that COS treatment ameliorated the damage of lipids of retinal cells (Fang et al., 2013) and huaman umbilical vein endothelial cells (Liu et al., 2009).
It has been suggested that increase in ROS accumulation associated with senescence may in fact be counteracted by the rise in antioxidants (van Doorn and Woltering, 2008). There are many antioxidant molecules, such as reduced GSH (Rogers, 2012). GSH directly reacts with ROS and functions as a cofactor of glutathione peroxidase. COS can markedly increase the reduced GSH content during the senescence process of petals (Fig. 4A). Fang et al. (2013) had the similar results that COS treatment increased the reduced GSH in retinal cells. Therefore, COS prolongs the vase life and amendments flower morphology of cut rose through directly or indirectly alleviating the oxidative stress in cut rose.